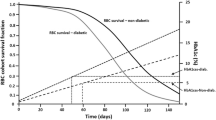
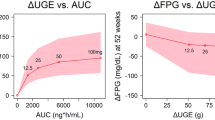
Abstract
Effective long-term treatment of Type 2 Diabetes Mellitus (T2DM) implies modification of the disease processes that cause this progressive disorder. This paper proposes a mechanism-based approach to disease progression modeling of T2DM that aims to provide the ability to describe and quantify the effects of treatment on the time-course of the progressive loss of β-cell function and insulin-sensitivity underlying T2DM. It develops a population pharmacodynamic model that incorporates mechanism-based representations of the homeostatic feedback relationships between fasting levels of plasma glucose (FPG) and fasting serum insulin (FSI), and the physiological feed-forward relationship between FPG and glycosylated hemoglobin A 1c (HbA 1c). This model was developed on data from two parallel one-year studies comparing the effects of pioglitazone relative to metformin or sulfonylurea treatment in 2408 treatment-naïve T2DM patients. It was found that the model provided accurate descriptions of the time-courses of FPG and HbA 1c for different treatment arms. It allowed the identification of the long-term effects of different treatments on loss of β-cell function and insulin-sensitivity, independently from their immediate anti-hyperglycemic effects modeled at their specific sites of action. Hence it avoided the confounding of these effects that is inherent in point estimates of β-cell function and insulin-sensitivity such as the widely used HOMA-%B and HOMA-%S. It was also found that metformin therapy did not result in a reduction in FSI levels in conjunction with reduced FPG levels, as expected for an insulin-sensitizer, whereas pioglitazone therapy did. It is concluded that, although its current implementation leaves room for further improvement, the mechanism-based approach presented here constitutes a promising conceptual advance in the study of T2DM disease progression and disease modification.
Similar content being viewed by others
References
De Fronzo R.A. (1987). Lilly Lecture- The triumvirate: beta cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 37:667–687
DeFronzo R.A. (1999). Pharmacologic therapy for type 2 diabetes mellitus. Ann. Intern. Med. 131:281–303
Matthaei S., Stumvoll M., Kellerer M., Häring H.-U. (2000). Pathophysiology and pharmacological treatment of insulin resistance. Endocr. Rev. 21:585–618
Inzucchi S.E. (2002). Oral antihyperglycemic therapy for type 2 diabetes: scientific review. JAMA 287:360–372
Turner R.C., Matthews D.R. (1984). Insulin secretion in Type I and Type II diabetes. Front. Diabetes 4:36–54
UKPDS33. (1998). Intensive blood glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes. UK Prospective Diabetes Study Group. Lancet 352:837–853
UKPDS34. (1998). Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes. UK Prospective Diabetes Study Group. Lancet 352:854–865
Matthews D.R., Hosker J.P., Rudenski A.S., Naylor B.A., Turner R.C. (1985). Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
Chan P.L., Holford N.H. (2001). Drug treatment effects on disease progression. Annu. Rev. Pharmacol. Toxicol. 41:625–659
Holford N.H., Peace K.E. (1992). Methodologic aspects of a population pharmacodynamic model for cognitive effects in Alzheimer patients treated with tacrine. Proc. Natl. Acad. Sci. USA 89:11466–11470
Frey N., Laveille C., Paraire M., Francillard M., Holford N.H., Jochemsen R. (2003). Population PKPD modelling of the long-term hypoglycaemic effect of gliclazide given as a once-a-day modified release (MR) formulation. Br. J. Clin. Pharmacol. 55:147–157
Post T.M., Freijer J.I., DeJongh J., Danhof M. (2005). Disease system analysis: basic disease progression models in degenerative disease. Pharm. Res. 22:1038–1049
Wallace T.M., Levy J.C., Matthews D.R. (2004). Use and abuse of HOMA modeling. Diabetes Care 27:1487–1495
Schernthaner G., Matthews D.R., Charbonnel B.H., Hanefeld M., Brunetti P., and the QUARTET Study Group. Effectiveness and safety of pioglitazone versus metformin in patients with type 2 diabetes mellitus: a double-blind, randomized trial. J. Clin. Endocrinol. Metab. 89:6068–6076 (2004).
Charbonnel B.H., Matthews D.R., Schernthaner G., Hanefeld M., Brunetti P. (2004). A long-term comparison of pioglitazone and gliclazide in patients with Type 2 diabetes mellitus: a randomised, double-blind, parallel-group comparison trial. Diabetic Med. 22:399–405
Olefsky J.M. (2000). Treatment of insulin resistance with peroxisome proliferator-activated receptor γ agonists. J. Clin. Invest. 106:467–472
Aronoff S., Rosenblatt S., Braithwaite S., Egan J.W., Mathisen A.L., Schneider R.L. (2000). Pioglitazone hydrochloride monotherapy improves glycemic control in the treatment of patients with type 2 diabetes. Diabetes Care 23:1605–1611
Dayneka N.L., Garg V., Jusko W.J. (1993). Comparison of four basic models of indirect pharmacodynamic responses. J. Pharmacokinet. Biopharm. 21:457–478
Boeckmann A.J., Sheiner L.B., Beal S.L. (1994) NONMEM Users Guide—Part V. University of California, San Francisco
Yano Y., Beal S.L., Sheiner L.B. (2001). Evaluating pharmacokinetic/pharmacodynamic models using the posterior predictive check. J. Pharmacokinet. Pharmacodyn. 28:171–192
DeFronzo R.A., Goodman A.M. (1995). Effectiveness of metformin in patients with non-insulin-dependent diabetes mellitus. The Multicenter Metformin Study Group. N. Engl. J. Med. 333:541–549
Danhof M., Alvan G., Dahl S.G., Kuhlmann J., Paintaud G. (2005). Mechanism-based pharmacokinetic-pharmacodynamic modelling – a new classification of biomarkers. Pharm. Res. 22:1432–1437
American Diabetes Association. (2005). Standards of medical care in diabetes. Diabetes Care 28(Suppl 1): S4–S36
Tan M.H., Baksi A., Krahulec B., Kubalski P., Stankiewicz A., Urquhart R., Edwards G., Johns D. (2005). Comparison of pioglitazone and gliclazide in sustaining glycemic control over 2 years in patients with type 2 diabetes. Diabetes Care 28:544–550
Zhou G., Myers R., Li Y., Chen Y., Shen X., Fenyk-Melody J., Wu M., Ventre J., Doebber T., Fujii N., Musi N., Hirshman M.F., Goodyear L.J., Moller D.E. (2001). Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108(8):1167–1174
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Winter, W.d., DeJongh, J., Post, T. et al. A Mechanism-based Disease Progression Model for Comparison of Long-term Effects of Pioglitazone, Metformin and Gliclazide on Disease Processes Underlying Type 2 Diabetes Mellitus. J Pharmacokinet Pharmacodyn 33, 313–343 (2006). https://doi.org/10.1007/s10928-006-9008-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-006-9008-2