Abstract
Changes to the environmental landscapes from the Eocene to Holocene have influenced the evolution of Cercopithecoidea from arboreal origins in wet, forested regions in the Early Oligocene Fayum to semi-terrestrial lifestyles in drier Neogene landscapes and social systems of larger group living. These eco-behavioural transitions likely accompanied changes in behaviour, brain function, and associated skull morphology. The temporal lobe of the brain, an association cortex, is in close proximity to the middle cranial fossa (MCF) allowing prediction of temporal lobe volume (TLV) and investigation of cerebral reorganisation. We used micro-computed tomography (µCT) cranial scans (n = 135) generated into 3D virtual crania with seven MCF metrics predicting TLV from a multiple regression of 11 extant anthropoid taxa. We studied eight extinct taxa Proteopithecus sylviae and Catopithecus browni from the Late Eocene, Early Oligocene Apidium phiomense, Parapithecus grangeri and Aegyptopithecus zeuxis, Middle Miocene Victoriapithecus macinnesi, Pliocene Dinopithecus ingens, Pleistocene Papio angusticeps, and extant cercopithecines Cercocebus atys, Macaca mulatta and Papio anubis. PGLS regressions examined relative TLV to brain size between extinct and extant taxa. We tested differences in slopes and intercepts between extinct and extant cercopithecoids with statistically significant differences in slopes but not the intercepts, with stem-anthropoids having relative smaller TLV for brain size compared to extant cercopithecoids. Potential drivers for temporal lobe evolution include paleoenvironmental shifts from Eocene tropical rainforests to Plio-Pleistocene savannas. Socio-behavioural implications include change from arboreal to semi-terrestrial lifestyles, higher visual acuity, larger group sizes and greater cognitive complexity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The diversification of Cercopithecoidea occurred in the context of the climate-driven environmental changes between the Eocene and Holocene. The ancestral members of this superfamily lived in the wet, forested regions of the Late Eocene to Early Oligocene Fayum, perhaps a similar environment to modern-day Uganda, and postcranial evidence suggests most of them were arboreal quadrupeds inhabiting a swampy, densely vegetated paleolandscape with high rainfall (Simons 1995; Kay et al. 1997). By the Neogene, the paleoenvironment of the Fayum and Northern Africa in general had become drier and more open with forests giving way to sparse open woodlands inhabited by cercopithecoids like Victoriapithecus with semi-terrestrial locomotion (Benefit 1999; Jablonski 2002). The African paleoenvironment continued a trend towards more open woodland and grassy savanna (DuPont and Leroy 1995), where cercopithecoids like Dinopithecus were habitual terrestrial quadrupeds that likely lived in large baboon-like troops during the Plio-Pleistocene and Holocene (Elton 2012).
Adapting to more open landscapes and living in larger troops likely required behavioural adaptations associated with increased inter and intra-group competition and greater social complexity, which has been argued to be associated with larger absolute brain size (Benefit 1999). The relatively large brain in primates functions in reproductive and life history strategies associated with learning and cognitive and memory skills in species with prolonged growth and low reproductive turnover but complex social organisation (Shea 2006; Dunbar 2009). For stem-anthropoids inhabiting dense tropical rainforests such as those that existed during the Eocene-Oligocene transition, higher visual acuity and the ability to distinguish between closely spaced visual stimuli, coupled with colour vision that would have allowed the discrimination of red, orange ,and yellow fruits and between those which were ripe and unripe, would likely have been important brain functions (Dominy 2004; Kirk and Kay 2004). This shift away from forest habitats to larger, open-habitat group structures would have changed foraging and anti-predator strategies perhaps in ways that are found in among extant terrestrial primates that form troops the more open landscapes of Africa today (Cheney and Wrangham 1987; McKee and Keyser 1994; Gilbert et al. 2018). Extant cercopithecoids are known to rely extensively on visual communication such as determining colour changes related to sexual communication (Changizi et al. 2006; Moreira et al. 2022) and changes in facial expressions (Peterson et al. 2018), which were all at the expense of auditory and olfactory communication (Strier 2017). The Social Brain Hypothesis posits that the association between increased brain size and larger troop is a causal one, driven by more the complex social behaviours that some cercopithecoids would have started adopting in the Miocene (Barton 2006; Kudo and Dunbar 2001).
Many functions that can logically be linked to these behavioural transitions—memory, object recognition, perception of places and paths—are coordinated through the temporal lobe of the brain (e.g., Murray et al. 2007; Bryant and Pruess 2018; Schuurman and Bruner 2023). The temporal lobe is one of the least studied regions of the primate brain despite its importance in early anthropoid and human evolution, although there are now a few studies in non-human primate outgroups that are relevant to assessing evolution of the temporal lobe in hominin evolution (Rilling 2014; Braunsdorf 2021). Compared to other mammals, primates have greater visual acuity than most other mammals, and anthropoids have greater visual acuity relative to other primates (Kirk and Kay 2004; Veilleux and Kirk 2014). The temporal lobe in anthropoid primates also has an important role in processing visual, auditory and memory-related tasks (Murray et al. 2007). Visual acuity is related to specialised organisation of ganglion cells in the retina and information processed via two pathways, ventral and dorsal, in the middle temporal and inferior temporal cortexes (Pruess 2006). The dorsal pathway neurons are sensitive to object motion, while the ventral pathway neurons are sensitive to object form (Pruess 2006).
Despite the logic that both brain size and proportions of regions like the temporal lobe are likely to have been affected by the environmental transitions of the Cenozoic, examination of absolute or relative brain size in extinct and extant cercopithecoids remains has been pursued by only a few studies (Barton 2006; Grabwoksi 2016; Sansalone et al. 2020). The quantification of cerebral proportions relative to brain size is indicative of cerebral reorganisation, and extinct taxa have the potential to pinpoint when and where in evolution such reorganiation occurred and in what environmental context, allowing a understanding of when behavioural changes may have occurred alongside morphological adaptations. Relative changes to olfactory bulb size and frontal lobe size have been noted alongside increases in absolute brain size during cercopithecoid evolution (Simons 1995, 1997, 2001; Bush et al. 2004; Gonzales 2015; Harrington 2016). Notably, the identification of cerebral reorganisation in extinct catarrhines have functional, evolutionary, and phylogenetic significance for the important neuroanatomical changes in the Cercopithecoidea.
Whether the increase in brain size in crown cercopithecoids was associated with the Cenozoic environmental transition and whether it was accompanied by a relative increase or decrease in the temporal lobe remains largely unknown. However, the proposition that the opening of Neogene environmental and concomitant changes in cercopithecoid behaviours was accompanied by temporal lobe reorganization can be tested using both extinct and Recent taxa using the bony middle cranial fossa as a proxy. The Functional Craniology Hypothesis of Moss and Young (1960) suggests that a reciprocal association exists between brain and skull form, where the temporal lobe is in close spatial proximity to the middle cranial fossa (MCF) of the basicranium (Bastir et al. 2008). This association has allowed MCF metrics to predict temporal lobe volume (TLV) in extant and extinct anthropoids with reasonable accuracy (Rilling and Seligman 2002; Pearson et al. 2020, 2021).
The aim of this paper is to determine whether there was an increase in temporal lobe size relative to brain size between extinct and extant cercopithecoids and whether the potential temporal lobe size changes coincided with the paleoenvironmental transitions and whether there is any association with the emergence of anthropoid behavioural ecology. To examine these aims, we studied eight key extinct species from the Cercopithecoidea: Proteopithecus sylviae and Catopithecus browni from the Late Eocene of the Fayum in Egypt; Parapithecus grangeri, Apidium phiomense, and Aegyptopithecus zeuxis from the Early Oligocene of the Fayum; East African Middle Miocene Victoriapithecus macinnesi; South African Pliocene Dinopithecus ingens,; and Pleistocene Papio angusticeps. We also studied three extant cercopithecines: Cercocebus atys, Macaca mulatta, and Papio anubis. Evolutionary changes to relative temporal lobe size among stem anthropoids and extant cercopithecines may pinpoint the timing of cerebral reorganisation in the Cercopithecoidea and the role of phylogenetic and functional drivers in temporal lobe evolution.
Materials and methods
Two different imaging modalities were used in the initial stages of this study, micro-computed tomography (µCT) scans of ex vivo extinct and extant anthropoid crania (n = 142) and in vivo T1-structural magnetic resonance imaging (MRI) of the human and non-human primate brain (n = 78). The total sample (n = 222) represents 17 anthropoid species including eight extinct catarrhine species (Table 1). Nonhuman primate MRI scans were accessed from the National Chimpanzee Brain Resource with necessary animal ethics protocol approvals (Rilling and Insel 1999) and human MRI scans accessed from the Open Access Series of Imaging Studies (OASIS) with appropriate human ethics protocol approvals (Marcus et al. 2007). All processing of MRI scans was conducted in Brainsuite 17a (Shattuck and Leahy 2002).
Fossil specimens
In this study, we analysed miro-computed tomography scans of the fossil specimens from collections housed at Duke Lemur Centre Division of Fossil Primates (DPC): DPC- 18651, DPC-9867, DPC-11388, DPC-14095, DPC-1208, and DPC-5401; The Cairo Geological Museum (CGM): CGM-40327 and CGM-85785; The Ditsong National Museum of Natural History (DNMNH): DNMNH:SK:554, CO100, CO135, and SK554; and the National Museums of Kenya (KNM): KNM-MB-29100.
Data collection
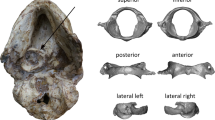
To predict right temporal lobe volume (TLV) in extant and extinct catarrhines, seven linear metrics were digitally measured (in mm) using Checkpoint (Stratovan 2021) on the right middle cranial fossa (MCF) surface in extant anthropoids (Fig. 1). TLV was predicted in the extinct taxa and compared to the predicted TLV in Three extant cercopithecoid species.
The seven metrics used to predict right temporal lobe volume, shown on the right middle cranial fossa (MCF). See Online Resource 1 for anatomical description of metrics used
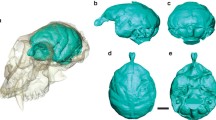
Endocranial volume in extant taxa was calculated (in cm3) through the generation of virtual endocasts from the µCT scans in Slicer 4.0 (Fedorov et al. 2012). For extinct taxa, endocranial volumes (in cm3) were taken from published literature (Online Resource 2, Table 1). For comparison with predicted TLV, the endocranial hemisphere volume (EHV) was calculated as the endocranial volume/2.
Statistical analysis
Preliminary analysis examined whether endocranial volume (EHV) was a suitable proxy for cerebral hemisphere volume. A phylogenetic generalized least-squares (PGLS) regression tested the correlation of extant anthropoid right cerebral hemisphere volume (in cm3) and right temporal lobe volume (in cm3) calculated by voxel from the MRI. The temporal lobe data were taken from Pearson et al. (2020), where a more detailed description of how we defined temporal lobe boundaries can be found. Intraobserver error was assessed by an analysis of variance, where measurement error was calculated as the proportion of the mean-squared differences between replicates relative to the total between-group variation (see Bailey and Byrnes 1990). For the seven MCF metrics, measurement error ranged from ≤ 1–4%. These were not considered error values of note and analysis proceeded.
A second PGLS regression examined the correlation between endocranial hemisphere volume (EHV) (in cm3), calculated from the endocasts, and predicted temporal lobe volume (in cm3), calculated from right MCF metrics. Both PGLS regressions indicated the predicted TLV and endocast EHV were suitable proxies for cerebral volumes (Online Resource 3).
All MCF variables were first converted from linear measurements (in mm) to natural logarithmic units. A multiple regression of MCF variables provided an equation to predict TLV in extant and extinct taxa. The cube-root was taken for TLV and EHV (in cm3), then converted to natural logarithmic units to linearise the scaling relationships for further analysis (Simpson et al. 2003).
PGLS regressions tested the correlation between TLV and EHV in extant and extinct taxa with PGLS regressions followed Martins and Hansen (1997). A consensus anthropoid tree was downloaded from 10kTrees version 3 (Arnold et al. 2010) containing Papio anubis, Cercocebus atys and Macaca mulatta with their molecular divergence dates. We then modified the consensus anthropoid tree in Mesquite version 3.70 (Maddison and Maddison 2021) to include extinct taxa with the generally accepted geological ages and taxonomic affinities used to estimate the phylogeny (Fig. 2).
Blomberg’s K (kappa) statistic was used to assess the phylogenetic correlations of the traits. K measures the proportion of the tip variance that matches the expectation of Brownian motion relative to the total variance and thus ranges from 0 to 1 (Blomberg, Garland et al. 2003). Values of K close to 0 indicate little congruence between the trait and the phylogenetic tree as expected under Brownian motion, whereas values closer to 1 indicate a strong congruence.
To examine if TLV was relatively larger or smaller than EHV in extinct taxa compared to extant taxa, further PGLS regressions tested whether the values for the slopes and intercepts were as high or higher than those observed by chance using random sampling (5,000 iterations).
All statistical analyses, PGLS regressions, K calculations, and trait- mapping were performed using Phylogenetics for Mathematica version 6.5 (Polly 2019) and performed in Mathematica version 13.1 (Wolfram 2021). For full Mathematica script, see Pearson (2023).
Results
We examined whether there was an increase or decrease in relative TLV to EHV in extinct taxa compared to extant cercopithecoids through trait-mapping, PGLS regressions with random sampling of regression slopes and intercepts between extant cercopithecines and stem-anthropoids.
Predicted temporal lobe volume
A multiple regression of naturally log-transformed TLV and seven MCF variables provided a statistically significant correlation (adjusted r2 = 0. 98, p < 0.01) in extant anthropoids (Table 2). Natural log-transformed variables for TLV and EHV were analysed with a multiple regression with the resultant prediction equation used to predict extant and extinct TLV (Table 3).
Evolution of brain size and temporal lobe size in the Cercopithecoidea
To examine the potential effects of phylogeny on EHV and TLV in the Cercopithecoidea, a PGLS regression of naturally log-transformed TLV and EHV produced a statistically significant correlation (r2 = 0.71, p ≤ 0.00107) in both extinct and extant taxa (Table 4; Fig. 3). Trait mapping indicated a stronger influence of phylogeny on EHV (k = 3.67) compared to a lesser influence on TLV (k = 0.60) in both extant and extinct taxa (Table 4; Fig. 4).
Relative temporal lobe size to brain size in the Cercopithecoidea
The difference in relative TLV to EHV in the Cercopithecoidea was conducted using two groups of taxa: (1) the clade of Miocene and later taxa, including the 3 extant cercopithecine species and 3 extinct cercopithecoids, and (2) the pre-Miocene stem-group of 5 extinct anthropoid species. PGLS regressions tested whether the slope and intercept values in the two groups were more different than expected by chance using a non-parametric bootstrapping test. The difference in slopes using random sampling with 5,000 iterations was statistically significant (p = 0.0014), while the difference in intercepts using random sampling with 5, 0000 iterations was not statistically significant (p = 0.27) between the two groups (Table 5; Fig. 5).
Comparison of random sampling in slopes (a) and intercepts (b) of stem-anthropoids and extinct and extant cercopithecoids from phylogenetic generalised least squares regression. The red bar represents the slope and intercept values in Table 5
Ultimately, these findings indicate temporal lobe size was larger relative to brain size in the extant cercopithecoid clade compared to the stem-anthropoids (Fig. 6). We note that the Late Eocene taxon Catopithecus and Early Oligocene taxa Apidium and Parapithecus lie well below the regression line as fit to the entire catarrhine clade. Differences in the slope for the stem-anthropoids was near zero and slightly negative which suggests that as brain size got larger there was either no relationship to temporal lobe size or that the temporal lobe size became proportionally smaller. In the crown-clade the slope was near 1.0, which suggests isometric scaling with body size: as brain got larger, the temporal lobe scaled 1-to-1 with it.
Phylogenetic generalised least squares regression depicting the regression line of TLV on EHV and comparison of slopes between stem-anthropoids (in blue) and crown-clade cercopithecoids (in orange) showing that relative TLV to EHV changed most noticeably between the stem-anthropoids and the crown-clade in the Late Eocene and Early Oligocene
Discussion
Brain evolution in the Cercopithecoidea and paleoenvironment
In general, our findings agree with Radinsky (1974) and Falk (1978), who documented an absolute brain size increase in cercopithecoid evolution (Fig. 4b). Our results also suggest a striking shift in the relationship between an increase in temporal lobe size relative to brain size between the stem and crown groups. Because of the links of the temporal lobe to visual acuity, memory, object recognition, and spatial navigation, and the roles that these play in extant cercopithecoid behaviours, it seems plausible that the marked transition between stem and crown scaling of the lobe is related to changes in individual and social behaviours that were associated with the environmental transitions from the Paleogene to the Neogene.
The changes in the paleoenvironment were also likely associated with changes in absolute brain size and body mass. A period of cooling and drying during the Oligocene likely required stem-anthropoids to expand into newer environments, where changes from an arboreal to semi-terrestrial locomotion would likely have been associated with shifts in body mass and diet (Scott 1995; Fleagle 2013) Among extant cercopithecines, species with larger body masses are unable to forage in the thinner upper canopy branches, which is where smaller-bodied cercopithecines have used niche partitioning (Cheney and Wrangham 1987). During the Miocene the continuing drier climate resulted in an expansion of sub-Saharan grassland savannahs and a shift from dense rainforests to more open woodlands favouring semi-terrestrial locomotion rather than obligate arboreal with postcranial and dentition of the extinct cercopithecine Victoriapithecus resembling extant taxa which favour semi-terrestrial locomotion also a diet of hard fruit and seeds (Benefit and McCrossin 2002).
Today, terrestrial primate taxa often have larger group sizes and a larger body mass. Studies of Papio, with its large body mass and strong sexual dimorphism, show that it has high levels of intragroup aggression and diets that are reliant during the African dry season on poor vegetation consisting of grasses and subterranean foods, supplemented in other seasons with fruit, leaves and occasionally meat consumed by males (Melnick and Pearl 1987). In contrast, Macaca, which inhabits tropical rainforest and woodland environments that are dietarily appropriate for a much smaller and absence sexually dimorphic body size and intragroup aggression. The semi-arboreal Cercocebus inhabits wetland, tropical woodland forest to open woodlands with smaller body mass and is reliant on the consumption of fruit and leaves (Melnick and Pearl 1987). These comparisons suggest that extinct taxa associated with tropical and rainforest vegetation might have consumed nutrients that would be suitable for larger brain size compared to the lower quality foods of savannas and grasslands. However, we found that extant taxa Cercocebus and Macaca, which inhabit forests and consume fruit and leaves, had smaller relative brain sizes compared to the savannah taxon Papio. Clutton-Brock and Harvey (1980) argued that diet and intra-family brain size are associated more with diet, where folivores had comparatively smaller brain size than frugivores and a lesser association between range size. In a more recent study by Chambers et al. (2021), diet was a better indicator of brain size than range size or social structure, which is at odds with the expectation of the Social Brain Hypothesis (Chambers et al. 2021). However, both studies suggested that diet variation and consistent access to resources might play a more significant role in primate brain size than social system.
In the Paleocene, early primate and euprimate taxa are thought to have been small-bodied and nocturnal and to have had limited colour perception (Dominy 2004). Their reliance on prehensile tail or grasping hands and feet to navigate the upper canopies were coupled with keen olfaction to better exploit access to fruit, but in the Eocene, stem anthropoids inhabited forests, exploiting fleshy fruit from palms, figs, and more lipid-rich laurels (Dominy 2004). Extant anthropoid primates are mostly diurnal with a few exceptions such as the nocturnal platyrrhine Aotus, but the emergence of colour vision is a staple argument in the divergence of anthropoids from the strepsirrhine-like primates ( Fleagle 2013; Strier 2017). Climate cooling in the late Paleogene resulted in increased seasonality in Africa and the loss of the palm forests; this required stem anthropoids to consume of proteinaceous leaves instead of fruit, as observed in the Egyptian Fayum deposits where Early Eocene anthropoids show a shift from fruit and insect diet to Early Oligocene preference for fruit and leaves (Dominy 2004). The the development of trichromatic vision has likely implications for cercopithecoid temporal lobe evolution where the ability to discern young reddish leaves against a background of mature foliage likely selected for those taxa with higher visual acuity which were more able to discern young seasonal leaves with greater nutritional value (Dominy 2004).
Cercopithecine behaviour and implications for temporal lobe size
Our findings indicated an isometric scaling for brain size likely associated with an increase in body size throughout cercopithecoid evolution as previously noted by Radinsky (1974) and Falk (1978). Potential drivers for an increase in brain size in Middle Miocene and Plio-Pleistocene taxa include the emergence of sexual dimorphism in Aegyptopithecus (Simons et al. 2007), suggesting increased intragroup social aggression, which is commonly found in extant cercopithecines with larger troop sizes such as savanna baboons (Cheney and Wrangham 1987; Melnick and Pearl 1987; Meguerditchiana et al. 2002).
The temporal lobe in humans is a highly multimodal association cortex associated with language processing, memory tasks, and auditory and visual processing (Rilling 2014). In non-human primates, which are less studied than humans, distinguishing auditory, memory, visual processing has been more difficult (Braunsdorf et al. 2021). A comparison between Homo sapiens and Pan troglodytes temporal cortex noted homologous language regions hidden beneath a sulcal fold in chimpanzees but visible on the sulcal surface in modern humans (Rilling et al. 2012). Complex vocal communication is not restricted to modern humans, but our species does rely heavily on a language system to the detriment of visual communication. In human and nonhuman primates, visual signalling and the processing of visual communication occurs in the temporal cortex and is associated with facial recognition and memory (Pruess 2006). Thus, in the early emergence of stem anthropoids inhabiting dense tropical rainforest, the ability to distinguish and memorise the faces, facial expressions, and appearance of group members versus other groups would have become increasingly relied upon, especially with the reduction in olfactory reliance for visual senses (Fleagle 2013).
The evolutionary implications for catarrhine reliance on visual communication is related to visual acuity, or the ability to distinguish between closely spaced visual stimuli. Diurnal anthropoids have significantly higher visual acuity than other mammals (Kirk and Kay 2004). In dense tropical rainforests during the Eocene, higher visual acuity combined with colour vision would allow discrimination of red, orange, and yellow fruits (Dominy 2004).
In dense rainforests, low-frequency calls travel further than higher-frequency calls at the same amplitude (Seyfarth 1987; Strier 2017). The use of greater vocal communication to pinpoint other group members may have been traded for greater reliance on visual acuity. For example, Late Eocene Cateopithcus and Proteopithecus had temporal lobes which are proportionally much larger and smaller, respectively. These differences in relative temporal lobe size could be associated with behavioural attributes, where greater reliance on navigating upper canopies versus lower canopy subsistence could result in a shift towards higher visual acuity, greater memory reliance, and cognitive complexity rather than lower visual acuity but greater auditory reliance. The Late Oligocene stem anthropoid Parapithecus was likely diurnal with visual acuity as high as extant primates (Kirk and Kay 2004). Our findings suggest Parapithecus had a small temporal lobe relative to brain size, which raises questions about whether increased temporal lobe function and higher cognitive processes were directly related to an increase in cortex size or, more likely, whether Parapithecus represents a transitional shift in cerebral reorganisation in the stem group of the Cercopithecoidea.
We found extant Papio had a slightly larger relative temporal lobe for brain size. Since baboons are terrestrial quadrupeds that inhabit open grassland and savannah environments, we hypothesize that larger group size, more vocal and visual communication such as the visual pathways processing in the inferior temporal cortex associated with facial recognition (Pruess 2006), behavioural cues including lip-curling and eye-lid flips commonly observed aggression displays among baboons (Strier 2017) likely require greater cognitive demands on the temporal lobe. Our findings imply the presence of interconnections between paleoenvironment and eco-behavioural adaptations in stem anthropoids and throughout extinct cercopithecine evolution.
Relative temporal lobe size in the Cercopithecoidea
Our findings indicate differences in relative temporal lobe size to brain size between extinct and extant cercopithecines and stem anthropoids. The evolution of the stem anthropoid temporal cortex may be influenced by visual and auditory communication. The smaller-bodied Late Eocene taxa were habitually arboreal quadrupeds and leapers inhabiting dense canopies where the demands of a higher functioning temporal cortex was selected in favour of relatively large olfactory bulbs for brain size (Simons 1995).
We did not find a consistent observable trend in relative temporal lobe size among the stem anthropoids, but Late Eocene Proteopithecus had smaller relative temporal lobes to brain size compared to the contemporaneous Catopithecus, which had proportionally much larger temporal lobes for brain size. Early Oligocene Apidium and Parapithecus both had smaller relative temporal lobes compared to the sexually dimorphic Aegyptopithecus, which had temporal lobes proportional to brain size. In the extant and extinct cercopithecines, temporal lobe scaled proportionally with brain size, likely associated with increases in body mass as present in the Middle Miocene extinct cercopithecoid Victoriapithecus, Pliocene extinct cercopithecine Dinopithecus, Early Pleistocene Papio, and extant cercopithecines Macaca, Cercocebus and Papio.
Conclusion
Our findings about relative temporal lobe size show that the scaling relationship between temporal lobe and brain size changed between the stem cercopithecines of the Late Eocene and Oligocene compared to the crown group in the Late Miocene and Quaternary. The timing of the transition suggests that cerebral reorganisation occurred at several key stages during the evolution of the Cercopithecoidea but was most noticeable in the Late Eocene and Early Oligocene during the initial opening of paleoenvironments.
Through the application of virtual imaging methods and phylogenetic comparative methods, this study expands on previous work conducted on absolute cercopithecoid brain evolution. In general, we found in agreement with previous findings that absolute brain size increased throughout cercopithecoid evolution. However, we note caution as there is continued debate about the phylogenetic relationships among stem-anthropoid taxa, and because there is a range of phylogenetic, behavioural and neurological differences in the taxa we have studied.
This study provides the first quantification of evolutionary changes to relative brain size and temporal lobe size in the Cercopithecoidea. The inclusion of comparative neuroanatomy and cranial anatomy, phylogenetic comparative methods and robust statistical analyses allow further understanding of relative brain size and temporal lobe evolution in the Cercopithecoidea. Potential drivers for temporal lobe evolution include changes to the processing of visual and communication pathways in response to fluctuating in paleoenvironment and socioecology.
Data availability
The data supporting this study are available via Dryad: https://doi.org/10.5061/dryad.xd2547dn3.
References
Arnold C, Matthews LJ, Nunn, CL (2010). The 10kTrees website: A new online resource for primate phylogeny. Evol Anthro 19:114–118
Bailey RC, Byrnes J (1990) A new, old method for assessing measurement error in both univariate and multivariate morphometric studies. Syst Zool 39(2):124–130
Barton RA (2006) Primate brain evolution: Integrating comparative, neurophysiological, and ethological data. Evol Anthropol 15:224 – 236
Bastir M, Rosas A, Lieberman D, O’Higgins P (2008) Middle cranial fossa anatomy and the origin of modern humans. Anat Rec 291:130–140
Benefit BR (1999) Victoriapithecus: The key to Old World monkey and catarrhine origins. Evol Anthropol 155–174
Benefit BR, McCrossin M (2002) The Victoriapithecidae: Cercopithecoidea. In: Hartwig WC (ed) The Primate Fossil Record. Cambridge University Press, Cambridge, UK, pp 241–253
Blomberg SP, Garland, TJ, Ives, AR (2003). Testing for phylogenetic signal in comparative data: Behavioral traits are more labile. Evol 57(4):717–745
Braunsdorf M, Blazquez-Freches G, RoumazeillesL, Eichert N, Schurzad M, Uithol S, Bryant KL, Mars RB (2021) Does the temporal cortex make us human? A review of structural and functional diversity of the primate temporal lobe. Neurosci Biobehav Rev 131:400–410
Bryant KL, Preuss TM (2018) A comparative perspective on the human temporal lobe. In: Bruner E, Ogihara N, Tanabe HC (eds) Digital Endocasts: From Skulls to Brains. Springer, Tokyo, pp 239–258
Bush EC, Simons EL, Allman JM (2004) High-resolution computed tomography study of the cranium of a fossil anthropoid primate, Parapithecus grangeri: new insights into the evolutionary history of primate sensory systems. Anat Record 281:1083–1087
Chambers HR, Heldstab SA, O’Hara SJ (2021) Why big brains? A comparison of models for both primate and carnivore brain size evolution. PLoS One 16(12):e0261185
Changizi MA, Qiong Z, Shinsuke S (2006) Bare skin, blood and the evolution of primate colour vision. Biol Lett 2217–221
Cheney DL, Wrangham RW (1987) Predation. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate Societies. University of Chicago Press, Chicago, London, pp 227–239
Clutton-Brock TH, Harvey PH (1980) Primates, brains and ecology. J Zool 190:309–323
Dominy NJ (2004) Color as an indicator of food quality to anthropoid primates: ecological evidence and an evolutionary scenario. In: Ross CF, Kay R (eds) Anthropoid Origins: New Visions. Kluwer Academic/Plenum Publishers, New York, pp 615–644
Dunbar RIM (2009) The social brain hypothesis and its implications for social evolution. Ann Hum Biol 36(5):562–572
DuPont LM, Leroy SAG (1995) Steps toward drier climatic conditions in Northwestern Africa during the Upper Pliocene. In: Vbra ES, Denton GH, Partridge TC, Burckle LH (eds) Paleoclimate and Evolution, with Emphasis on Human Origins. Yale University Press, pp 289–298
Elton S (2012) Impacts of environmental change and community ecology on the composition and diversity of the southern African monkey fauna from the Plio-Pleistocene to the present. In: Reynolds S, Gallagher A (eds) African Genesis: Perspectives on the Hominin Evolution. Cambridge University Press, Cambridge, UK, pp 471–486
Falk D (1978) Brain evolution in Old World monkeys. Am J Phys Anthrop 48:315–320
Fedorov A et al (2012) 3D Slicer as an image computing platform for the quantitative imaging network. JMRI 30(9):1323–41
Fleagle JG (2013) Primate Adaptation and Evolution, 3rd Ed. Academic Press, San Diego, Waltham, London
Gilbert CC, Frost SR, Kelsey D, Pugh KD, Anderson M, Delson E (2018) Evolution of the modern baboon (Papio hamadryas): A reassessment of the African Plio-Pleistocene record. J Hum Evol 122: 38–69
Gonzales LA, Benefit BR, McCrossin ML, Spoor F (2015) Cerebral complexity preceded enlarged brain size and reduced olfactory bulbs in old world monkeys. Nature Comm 6:75–80
Grabwoksi M (2016) Bigger brains led to bigger bodies? The correlated evolution of human brain and body size. Curr Anthropol 57(2):174–196
Harirngton A, Silcox MT, Yapuncich GS, Boyer DM, Bloch JI (2016) First virtual endocasts of adapiform primates. J Hum Evol 99:52–78
Jablonski, NG (2002) Fossil Old World monkeys: The late Neogene radiation. In: Hartwig WC (ed) The Primate Fossil Record. Cambridge University Press, Cambridge, UK, pp 255–300
Kay RF, Ross C, Blythe, WA (1997) Anthropoid origins. Science 275(5301):797–804
Kirk C, Kay RF (2004) Evolution of high visual acuity in the Anthropoidea. In: Ross CF, Kay R (eds) Anthropoid Origins: New Visions. Kluwer Academic/Plenum Publishers, New York, pp 539–602
Kudo H, Dunbar RIM (2001) Neocortex size and social network size in primates. Anim Behav 62(4):711–722
Maddison WP, Maddison DR (2021) Mesquite: a modular system for evolutionary analysis Version 3.70. Available at http://www.mesquiteproject.org
Marcus DS, Wang TH, Parker J, Csernansky JG, Morris JC, & Buckner RL (2007) Open access series of imaging studies (OASIS): Cross-sectional MRI data in young, middle aged, nondemented, and demented older adults. J Cogn Neurosci 19(9):1498–1507
Martins EP, Hansen TF (1997) Phylogenies and the comparative method: A general approach to incorporating phylogenetic information into the analysis of interspecific data. Am Nat 149:646–667
McKee JK, Keyser AW (1994) Craniodental remains of Papio angusticeps from the Haasgat Cave site, South Africa. Int J Primatol 15:823–841
Meguerditchiana A, Marie D, Margiotoudi K, Roth M, Nazarian A J-L, Claidière N (2002) Baboons (Papio anubis) living in larger social groups have bigger brains. Evol Hum Behav 42:30–34
Melnick DJ, Pearl MC (1987) Cercopithecines in multimale groups: genetic diversity and population structure. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate Societies. University of Chicago Press, Chicago, London pp 121–134
Moreira LAA, Watsa M, Erkenswick G, Higham JP, Melin AD (2022) Evaluating genital skin color as a putative sexual signal in wild saddleback (Leontocebus weddelli) and emperor (Saguinus imperator) tamarins. Am J Primatol 85(2):e23456. https://doi.org/10.1002/ajp.23456
Moss ML, Young RW (1960) A functional approach to craniology. Am J Phys Anthrop 18:281–292
Murray EA, Bussey TJ, Saksida LM (2007) Visual perception and memory: a new view of the medial temporal lobe function in primates and rodents. Ann Rev Neurosci 30:99–122
Pearson A (2023) Cercopithecoidea Mathematica Notebook PDF. Dryad Dataset https://doi.org/10.5061/dryad.xd2547dn3
Pearson A, Polly PD, Bruner E (2020) Is the middle cranial fossa a reliable predictor of temporal lobe volume in extant and fossil anthropoids? Am J Phys Anthropol 172:698–713
Pearson A, Polly PD, Bruner E (2021) Temporal lobe evolution in Javanese Homo erectus and African Homo ergaster: Inferences from the cranial base. Quat Int 603:5–21
Peterson A, Abella EF, Grine FE, Teaford MF, Ungar PS (2018) Microwear textures of Australopithecus africanus and Paranthropus robustus molars in relation to paleoenvironment and diet. J Hum Evol 119:42–63
Polly PD (2019) Phylogenetics for Mathematica Version 6.5. Department of Earth and Atmospheric Sciences, Bloomington, Indiana, Indiana University. Available at http://pollylab.indiana.edu/software.html
Pruess TM (2006) Evolutionary specializations of primate brain systems. In: Ravosa MT, Dagosto M (eds) Primate Origins: Adaptations and Evolution. Springer Science + Business Media, LLC, New York, NY, USA, pp 623–675
Radinsky L (1974) The fossil evidence of anthropoid brain evolution. Am J Phys Anthrop 41:15–28
Rilling JK (2014) Comparative primate neurobiology and the evolution of brain language systems. Curr Opin Neurol 28:10–14
Rilling JK, Insel TR (1999) The primate neocortex in comparative perspective using magnetic resonance imaging. J Hum Evol 37(2):191–223
Rilling JK, Seligman RA (2002) A quantitative morphometric comparative analysis of the primate temporal lobe. J Hum Evol 42(5):505–533
Rilling JK, Glasser MF, Jbabadi S, Andersson J, Preuss TM (2012) Continuity, divergence, and the evolution of brain language pathways. Front Evol Neurosci https://doi.org/10.3389/fnevo.2011.00011
Sansalone G et al (2020) Variation in the strength of allometry drives rates of evolution in primate brain shape. Proc R Soc B https://doi.org/10.1098/rspb.2020.0807
Schuurman T, Bruner E (2023) A comprehensive anatomical network analysis of human brain topology. J Anatomy https://doi.org/10.1111/joa.13828
Scott L (1995) Pollen evidence for vegetational and climatic change in southern Africa during the Neogene and Quaternary In: Vbra ES, Denton GH, Partridge TC, Burckle LH (eds) Paleoclimate and Evolution, with Emphasis on Human Origins. Yale University Press, New Haven, CT, pp 65–76
Seyfarth R (1987) Vocal communication and its relation to language. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate Societies. University of Chicago Press, Chicago, pp 440–451
Shattuck DW, Leahy RM (2002). BrainSuite: An automated cortical surface identification tool. Med. Image Anal 6(2):129–142
Shea, BT (2006) Start small and live slow: Encephalization, body Size, and life history strategies in primate origins and evolution. In: Ravosa MT, Dagosto M (eds) Primate Origins: Adaptations and Evolution. Springer Science + Business Media, LLC, New York, NY, USA, pp 583–623
Simons E (1995) Egyptian Oligocene primates: a review. Yearb Phys Anthropol 38:199–238
Simons E (1997) Preliminary description of the cranium of Proteopithecus sylviae, an Egyptian late Eocene anthropoidean primate. Proc Natl Acad Sci U S A 94:14970–14975
Simons E (2001) The cranium of Parapithecus grangeri, an Egyptian Oligocene anthropoidean primate. Proc Natl Acad Sci U S A 98(14):7892–7897
Simons EL, Seiffert ER, Ryan TM, Attia Y (2007) A remarkable female cranium of the early Oligocene anthropoid Aegyptopithecus zeuxis (Catarrhini, Propliopithecidae). Proc Natl Acad Sci U S A 104(21):8731–8736
Simpson GG, Roe A, Lewontin RC (2003) Quantitative Zoology (Revised ed.) Dover Publications, New York
Stratovan (2021) Stratovan Checkpoint. UC, Davis, California: Stratovan Corporation
Strier KB (2017) Primate Behavioral Ecology, 5th edn. Routledge, Oxon, New York
Veilleux, CC, Kirk CE (2014) Visual acuity in mammals: Effects of eye size and ecology. Brain Behav Evol 83(1):43–53
Wolfram (2021) Wolfram Mathematica Version 13.0. Wolfram Research, Inc. Champaign, Illinois
Acknowledgements
We thank Colin Groves for his invaluable support in the initial conception of this research and the following museums and researchers for access to digital resources. The Smithsonian Division of Mammals (Dr. Kristofer Helgen) and Human Origins Program (Dr. Matt Tocheri) for the scans of USNM specimens used in this research (http://humanorigins.si.edu/evidence/3d-collection/primate). These scans were acquired through the generous support of the Smithsonian 2.0 Fund and the Smithsonian’s Collections Care and Preservation Fund; Museum National d’histoire Naturelle (MNHN); Digital Morphology Museum (DMM) the Primate Research Institute, Kyoto University (KUPRI); Anatomisches Institut, Universität Leipzig (Germany); University of Pennsylvania Museum of Archeology and Anthropology and the Open Research Scan Archive (ORSA) care of J. Monge and P. T. Schoenemann (NSF proposal # 0447271); The National Chimpanzee Brain Resource care of J. Rilling; OASIS: Cross-Sectional: Principal Investigators: D. Marcus, R, Buckner, J, Csernansky J. Morris (#P50 AG05681, #P01 AG03991, #P01 AG026276, #R01 AG021910, #P20 MH071616, #U24 RR021382). The following digital access is acknowledged from Morphosource (www.MorphoSource.org) for fossil specimens DPC- 18651, DPC-9867, DPC-11388 and DPC-14095, CGM-40327, CGM- CGM-85785, DPC-1208, DPC-5401 care of Alan Walker (funded by Pennsylvania State University); CO100, CO135 and SK554 from the Ditsong National Museums of Natural History; KNM-MB-29100 imaging fossil specimen care of National Musuems of Kenya (Nairobi, Kenya), the Max Planck Institute for Evolutionary Anthropology (Leipzig, Germany), Fred Spoor and Lauren Gonzales.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
AP conducted data collection, wrote the draft manuscript and revisions. PDP conducted the analysis and figures and wrote the draft manuscript and revisions. Both authors reviewed the manuscript.
Corresponding author
Ethics declarations
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pearson, A., Polly, P.D. Temporal lobe evolution in extant and extinct Cercopithecoidea. J Mammal Evol 30, 683–694 (2023). https://doi.org/10.1007/s10914-023-09664-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10914-023-09664-6