Abstract
In this paper, an n-step, linear and unbranched pathway with Michaelis–Menten kinetics is solved in a quasi-analytical way. The method, based on the optimal control theory, calculates the optimal enzyme concentrations while minimizing the operation time. In the computation of the solution, the Lambert W-function plays a fundamental role, due to the presence of a non-linear kinetic model. Our method allows us to obtain the generalized solution and to perform the sensitivity analysis of the catalytic parameters.






Similar content being viewed by others
References
E. Klipp, R. Heinrich, H.G. Holzhutter, Prediction of temporal gene expression. Metabolic optimization by re-distribution of enzyme activities. Eur. J. Biochem. 269(22), 5406–5413 (2002)
M. Bartl, P. Li, S. Schuster, Modelling the optimal timing in metabolic pathway activation—-use of Pontryagin’s maximum principle and role of the golden section. Biosystems 101, 67–77 (2010)
L. Bayon, J.M. Grau, M.M. Ruiz, P.M. Suarez, Optimal control of a linear unbranched chemical process with steps: the quasi-analytical solution. J. Math. Chem. 52(4), 1036–1049 (2014)
L. Bayon, J.A. Otero, M.M. Ruiz, P.M. Suarez, C. Tasis, Sensitivity analysis of a linear unbranched chemical process with n steps. J. Math. Chem. 53(3), 925–940 (2015)
D. Oyarzun, B. Ingalls, R. Middleton, D. Kalamatianos, Sequential activation of metabolic pathways: a dynamic optimization approach. Bull. Math. Biol. 71(8), 1851–1872 (2009)
R. Heinrich, S.M. Rapoport, T.A. Rapoport, Metabolic regulation and mathematical models. Prog. Biophys. Mol. Biol. 32, 1–82 (1977)
R. Heinrich, E. Klipp, Control analysis of unbrached enzymatic chains in states of maximal activity. J. Theor. Biol. 182, 243–252 (1996)
A. Zaslaver, A. Mayo, R. Rosenberg, P. Bashkin, H. Sberro, M. Tsalyuk, M. Surette, U. Alon, Just-in-time transcription program in metabolic pathways. Nat. Genet. 36(5), 486–491 (2004)
E. Melendez-Hevia, N.V. Torres, J. Sicilia, H. Kacser, Control analysis of transition times in metabolic systems. Biochem. J. 265, 195–202 (1990)
G. Curien, M.L. Cardenas, A. Cornish-Bowden, Analytical kinetic modeling: a practical procedure. Methods Mol. Biol. 1090, 261–280 (2014)
L.A. Segel, M. Slemrod, The quasi-steady state assumption: a case study in perturbation. SIAM Rev. 31(3), 446–477 (1989)
S. Schnell, C. Mendoza, A closed form solution for time-dependent enzyme kinetics. J. Theor. Biol. 187(2), 207–212 (1997)
M.N. Berberan-Santos, A general treatment of Henri–Michaelis–Menten enzyme kinetics: exact series solution and approximate analytical solutions. MATCH Commun. Math. Comput. Chem. 63(2), 283–318 (2010)
M. Schleeger, J. Heberle, S. Kakorin, Simplifying the analysis of enzyme kinetics of cytochrome c oxidase by the Lambert-W function. Open J. Biophys. 2, 117–129 (2012)
R.M. Corless, G.H. Gonnet, D.E.G. Hare, D.J. Jeffrey, D.E. Knuth, On the Lambert W function. Adv. Comput. Math. 5, 329–359 (1996)
L.S. Pontryagin, V.G. Boltayanskii, R.V. Gamkrelidze, E.F. Mishchenko, The mathematical theory of optimal processes (Wiley, Hoboken, 1962)
T. Turanyi, Sensitivity analysis of complex kinetic systems. Tools and applications. J. Math. Chem. 5(3), 203–248 (1990)
A. Saltelli, M. Ratto, S. Tarantola, F. Campolongo, Sensitivity analysis for chemical models. Chem. Rev. 105(7), 2811–2828 (2005)
M. Komorowski, M. Costa, D. Rand, M. Stumpf, Sensitivity, robustness, and identifiability in stochastic chemical kinetics models. Proc. Natl. Acad. Sci. USA 108(21), 8645–8650 (2011)
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Proof of Theorem 2.
We now prove that the solution obtained using Pontryagin’s Minimum Principle is effectively a solution of our problem. In (22), we have \(F=1\), \(B=1\) and the Hamiltonian H is:
which is autonomous, so that \(H_{t}\equiv 0\Rightarrow H(t)=ct\). This condition together with (iv) implies that \(H(t)=0\). Now the optimality condition (ii) leads to:
with \(\lambda _{n+1}=0\). According to the optimality condition (i), we have:
It is known from (33) that the control \(u_{i}\) is activated when the switching function \(\mu _{i}\) reaches its maximum. Moreover, when this happens, the coefficient \(\mu _{i}\) must be positive, because otherwise \(u_{i}=0\). Hence, it follows that \(\lambda _{i}\) is decreasing. From (33):
We obtain the optimal solution constructively by intervals, starting at \(t=0\) and concatenating the results. This procedure will prove essential in order to obtain a simple solution to the problem. We shall also see that using the following condition in (22):
we are not going to require either the final condition (i) \(\lambda _{i}^{*}(t_{f}^{*})=0\) for the costate variables, or the transversality condition (iv) \(H(t)=0\). As a matter of fact, we shall see that it will not be necessary to compute \(\lambda _{i}^{*}\), so that we shall not compute H(t) either.
(1) Interval: \(\left[ 0,t_{1}\right] \).
We reason by contradiction. Assume that \(u_{1}=0\). From (19):
and the product would not be produced. Hence, we have \(u_{1}=1\) and from condition (36) we get:
Once the optimal values for the enzymes are computed, we can solve now (19):

In Appendix 2 we give the details of the solution. In order to generalize the formula, it is interesting to use the following notation: we denote by \(x_{ji}(t)\) the concentration of j-metabolite in the i-interval \([t_{i-1},t_{i}],\) \(i=1,\ldots ,n\), with \(x_{10}(t_{0})=1\). So, we have:
From (34), the following holds:
From (33) and (19), after some elementary computations and substituting \(\dot{\lambda }_{1}\) and \(\dot{x}_{1}\) by their values, we get
On the other hand:
So that
(2) Interval: \(\left[ t_{1},t_{2}\right] .\)
With a reasoning analogue to the one used for the first interval:
and:

which gives:
and performing the adequate substitutions, one proves that:
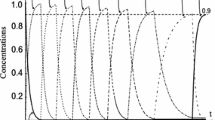
In Fig. 7, the behavior of the switching functions is shown.
The values for each successive interval are similarly obtained, by concatenating the solutions. For the sake of simplicity, we present only the solution for the last one.
(n) Interval: \([t_{n-1},t_{f}]\).
In this case:
and
Once the optimum values for \(x_{i}^{*}\) and \(u_{i}^{*}\) have been obtained, it is still required to compute the values of the following unknowns: the switching times \(t_{1},t_{2},\dots ,t_{n-1}\) and the operation time \(t_{f}\). In order to do so, we use the restriction (21) which we have not used yet. The simplest way is to apply the Lagrange multipliers to the augmented functional:
where the values of the concentrations \(x_{1n}(t_{f}),x_{2n}(t_{f} ),\ldots ,x_{nn}(t_{f})\) are given by (24), (25) and (26), and in which one sees that the unknowns \(t_{1},t_{2},\dots ,t_{n-1}\) appear. We need to solve the non-linear system:
which can be done with any computer algebra software. This is the only part of the solution which is not carried out analytically, whence our calling it “quasi-analytical.” Now the problem is completely solved.
Appendix 2: Solution of the state equations
In order to shed some light on the solution of the state equations, we carry it out completely for the case of the interval \([0,t_{1}]\). First, we solve the differential equation for \(x_{1}(t)\):
Integrating:
Imposing the initial condition \(x_{1}(0)=1\), we get \(C=1\), so that
By exponentiation:
Dividing by \(K_{m1}\):
And from the definition of the Lambert W-function
we get:
In order to obtain the closed form expression for \(x_{2}(t)\), instead of integrating
it is much easier to realize that
from which follows, immediately, that
so that we get the recurrence relation:
And one proceeds similarly for the remaining intervals.
Rights and permissions
About this article
Cite this article
Bayón, L., Otero, J.A., Suárez, P.M. et al. Solving linear unbranched pathways with Michaelis–Menten kinetics using the Lambert W-function. J Math Chem 54, 1351–1369 (2016). https://doi.org/10.1007/s10910-015-0579-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10910-015-0579-2