Abstract
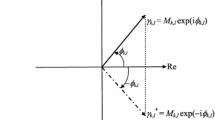
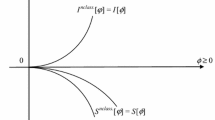
Information-theoretic description of the electron probabilities and currents in molecules is extended to cover the complex amplitudes (wave functions) of quantum mechanics. The classical information measures of Fisher and Shannon, due to the probability/density distributions themselves, are supplemented by the nonclassical terms generated by the wave-function phase or the associated probability current. The previous one-electron development in such an entropic perspective on the molecular electronic structure is extended to cover N-electron states by adopting the Harriman-type framework of equidensity orbitals. This analysis emphasizes the phase part of electronic states, which generates the probability-current density and the associated non-classical entropy contributions, which allow one to distinguish the information content of states generating the same electron density and differing in their current composition. A complementary character of the Fisher and Shannon information measures is explored in the associated vertical (density-constrained) information principles, for determining the equilibrium state corresponding to the fixed ground-state electron density. It is argued that the lowest “thermodynamic” state generally differs from the true ground state of the system, by exhibiting the space-dependent phase and hence also the non-vanishing probability current, linked to the system electron distribution.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fisher R.A.: Proc. Camb. Phil. Soc. 22, 700 (1925)
Shannon C.E.: Bell System Tech. J. 27, 379, 623 (1948)
Frieden B.R.: Physics from Fisher Information: A Unification. Cambridge University Press, Cambridge (1998)
Hall M.J.W.: Phys. Rev. A 62, 012107 (2000)
Nagy A.: Chem. Phys. Lett. 449, 212 (2007)
Nalewajski R.F.: Int. J. Quantum Chem. 108, 2230 (2008)
R.F. Nalewajski, in Foundations of Chemistry: Quantum Theory of Atoms and Molecules, ed. by C. Matta (in press)
R.F. Nalewajski, Entropic representation in the theory of molecular electronic structure. J. Math. Chem. (in press)
Nalewajski R.F.: Information Theory of Molecular Systems. Elsevier, Amsterdam (2006)
Levy M.: Proc. Natl. Acad. Sci. USA 76, 6062 (1979)
Hohenberg P., Kohn W.: Phys. Rev. 136B, 864 (1964)
Kohn W., Sham L.J.: Phys. Rev. 140A, 1133 (1965)
R.G. Parr, W. Yang, Density Functional Theory of Atoms and Molecules (Oxford, New York, 1989)
Dreizler R.M., Gross E.K.U.: Density Functional Theory: An Introduction to the Quantum Many-Body Problem. Springer, Berlin (1990)
R.F. Nalewajski, ed. Density Functional Theory I–IV, Topics in Current Chemistry, vols. 180–183 (Springer, Heidelberg, 1996)
Callen H.B.: Thermodynamics: An Introduction to the Physical Theories of the Equilibrium Thermostatics and Irreversible Thermodynamics. Wiley, New York (1960)
Harriman J.E.: Phys. Rev. A 24, 680 (1981)
Macke W.: Ann. Phys. Leipzig 17, 1 (1955)
Gilbert T.L.: Phys. Rev. B 12, 2111 (1975)
March N.H.: Phys. Rev. A 26, 1845 (1982)
G. Zumbach, K. Maschke, Phys. Rev. A 28, 544. Erratum, Phys. Rev. A 29, 1585 (1984)
R.F. Nalewajski, in Recent Advances in Density Functional Methods, part III, ed. by V. Barrone, A. Bencini, P. Fantucci (World Scientific, Singapore 2002), p. 257
Nagy A., Liu S.B.: Phys. Lett. A 372, 1654 (2008)
von Weizsäcker C.F.: Z. Phys. 96, 431 (1935)
R.F. Nalewajski, Ann. Phys. Leipzig (submitted)
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Nalewajski, R.F. Use of Harriman’s construction in determining molecular equilibrium states. J Math Chem 51, 369–381 (2013). https://doi.org/10.1007/s10910-012-0088-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10910-012-0088-5