Abstract
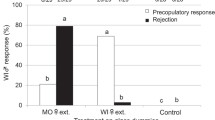
Herbivorous insects frequently exhibit sexual dimorphism in body size and a variety of other traits. Such differences often lead to distinct behaviors, which may present themselves in insect responses to conspecifics, plant defenses, and feeding. Based on a previous study of a congeneric species, we hypothesized that sexual dimorphism may yield differences in how the host-specific red milkweed beetle Tetraopes tetrophthalmus (Coleoptera: Cerambycidae) deactivates milkweed defenses, a behavior in which beetles cut latex-delivering veins prior to feeding. We also predicted that beetles would display differences in their subsequent feeding patterns and attraction of conspecifics. Although beetles were size dimorphic (females were larger than males), we did not find consistent differences in the rates of vein cutting or initiation of feeding between males and females. Females did, however, cut more milkweed veins per leaf, and attracted more conspecifics than males. Sex differences in size and other traits were thus associated with differential attraction, but not strongly associated with the overall deactivation of plant defense.


Similar content being viewed by others
Data Availability
All data from this study will be accessible after publication from: https://doi.org/10.6084/m9.figshare.25037171.
References
Agrawal AA (2004) Resistance and susceptibility of milkweed: competition, root herbivory, and plant genetic variation. Ecology 85:2118–2133. https://doi.org/10.1890/03-4084
Agrawal AA (2017) Monarchs and milkweed: a migrating butterfly, a poisonous plant, and their remarkable story of Coevolution. Princeton University Press, Princeton
Agrawal AA, Konno K (2009) Latex: a model for understanding mechanisms, ecology, and evolution of plant defense against herbivory. Annu Rev Ecol Evol Syst 40:311–331. https://doi.org/10.1146/annurev.ecolsys.110308.120307
Alexander RD (1957) Sound production and communication in insects. Ohio J Sci 57:101–113
Bengtsson JM (2008) Aggregation in non-social insects: an evolutionary analysis. Anim Behav Cogn 1–7. https://doi.org/10.1007/978-3-319-47829-6_433-1
Blomquist GJ, Bagnéres A (2010) Insect hydrocarbons: Biology, biochemistry, and chemical ecology. Cambridge, England
Boone CK, Keefover-Ring K, Mapes AC, Adams AS, Bohlmann J, Raffa KF (2011) Responses of bark beetle associated bacteria to host monoterpenes and their relationship to insect life histories. J Chem Ecol 37:808–817. https://doi.org/10.1007/s10886-013-0313-0
Brzozowski LJ, Gardner J, Hoffmann MP, Kessler A, Agrawal AA, Mazourek M (2020) Attack and aggregation of a major squash pest: parsing the role of plant chemistry and beetle pheromones across spatial scales. J Appl Ecol 57:1442–1451. https://doi.org/10.1111/1365-2664.13689
Dailey PJ, Graves RC, Kingsolver JM (1978) Survey of Coleoptera collected on the common milkweed, Asclepias syriaca, at one site in Ohio. Coleopt Bull 32:223–229
Dussourd DE (1999) Behavioral sabotage of plant defense: do vein cuts and trenches reduce exposure to exudate? J Insect Behav 12:501–515. https://doi.org/10.1023/A:1020966807633
Dussourd DE (2015) Theroa zethus caterpillars use acid secretion of anti-predator gland to deactivate plant defense. Plos One. https://doi.org/10.1371/journal.pone.0141924
Dussourd DE (2017) Behavioral sabotage of plant defenses by insect folivores. Annu Rev Entomol 62:15–34. https://doi.org/10.1146/annurev-ento-031616-035030
Dussourd DE, Eisner T (1987) Vein-cutting behavior: insect counterploy to the latex defense of plants. Science 237:898–901. https://doi.org/10.1126/science.3616620
Ferreira P, Rodrigues D (2015) Sabotaging behavior and decision-making in larvae of the Queen Butterfly Danaus gilippus. J Insect Behav 28:460–472. https://doi.org/10.1007/s10905-015-9514-4
Gontijo LM (2013) Female beetles facilitate leaf feeding for males on toxic plants. Ecol Entomol 38:272–277. https://doi.org/10.1111/een.12015
Lawrence WS (1982) Sexual dimorphism in between and within patch movements of a monophagous insect: Tetraopes (Coleoptera: Cerambycidae). Oecologia 53:245–250. https://doi.org/10.1007/BF00300006
Matter SF (2009) Abundance of an herbivorous beetle: factors affecting dispersal and local reproduction. Am Nat 162:19–28. https://doi.org/10.1674/0003-0031-162.1.19
Mori E, Mazza G, Lovari S (2017) Sexual dimorphism. Encycl Anim Cognit Behav 1–7. https://doi.org/10.1007/978-3-319-47829-6_433-1
Reagel PF, Ginzel MD, Hanks LM (2002) Aggregation and mate location in the red milkweed beetle (Coleoptera: Cerambycidae). J Insect Behav 15:811–830. https://doi.org/10.1023/A:1021127624215
Reisenman CE, Lorenzo Figueiras AN, Giurfa M, Lazzari CR (2000) Interaction of visual and olfactory cues in the aggregation behaviour of the haematophagous bug Triatoma infestans. J Comp Physiol A 186:961–968. https://doi.org/10.1007/s003590000149
Rice ME (1998) Natural history observations on Tetraopes and other Cerambycidae (Coleoptera) from the Great Plains ecosystem. J Kans Entomol Soc 61:412–419
Silliman CA (2014) Factors contributing to variation in cuticular hydrocarbon profiles of two species of longhorned beetles (Coleoptera: Cerambycidae). Dissertation, University of Illinois at Urbana-Champaign
Verrell P, Strand N, Hanson E (2001) Sexual dimorphism of mate location in the long-toed salamander Ambystoma macrodactylum columbianum. J Ethol 107:677–683. https://doi.org/10.1046/j.1439-0310.2001.00691.x
Vinterstare J, Ekelund Ugge GM, Hulthén K, Hegg A, Brönmark C, Nilsson PA, Zellmer UR, Lee M, Pärssinen V, Sha Y, Björnerås C, Zhang H, Gollnisch R, Herzog SD, Hansson L, Škerlep M, Hu N, Johansson E, Langerhans RB (2021) Predation risk and the evolution of a vertebrate stress response: parallel evolution of stress reactivity and sexual dimorphism. J Evol Biol 34:1554–1567. https://doi.org/10.1111/jeb.13918
Acknowledgements
We thank Amy Hastings for assistance in beetle collection and National Science Foundation grant IOS-2209762 to AAA for financial support.
Funding
This work was supported by National Science Foundation grant IOS-2209762 to AAA.
Author information
Authors and Affiliations
Contributions
Anurag Agrawal and Emma Craig conceptualized the study and designed the experiments. Emma Craig wrote the first draft of the manuscript and all authors participated in additional writing and editing. The bulk of field work and data collection was done by Emma Craig (Experiment 1 - Trials 1, 3, 4 and Experiment 2) and Max Goldman (Experiment 1 - Trial 2), and data was analyzed by Anurag Agrawal.
Corresponding author
Ethics declarations
The authors have no financial or non-financial interests to disclose.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Craig, E.J., Goldman, M.B. & Agrawal, A.A. Sexual Dimorphism, Deactivation of Plant Defense, and Attraction of Conspecifics in the Four-Eyed Red Milkweed Beetle (Tetraopes tetrophthalmus). J Insect Behav (2024). https://doi.org/10.1007/s10905-024-09847-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10905-024-09847-w