Abstract
COVID-19 caused significant declines in colorectal cancer (CRC) screening. Health systems and clinics, faced with a new rapidly spreading infectious disease, adapted to maintain patient safety and address the effects of the pandemic on healthcare delivery. This study aimed to understand how CDC-funded Colorectal Cancer Control Program recipients and their partner health systems and clinics may have modified evidence-based intervention (EBI) implementation to promote CRC screening during the COVID-19 pandemic; to identify barriers and facilitators to implementing modifications; and to extract lessons that can be applied to support CRC screening, chronic disease management, and clinic resilience in the face of future public health crises. Nine recipients were selected to reflect the diversity inherent among all CRCCP recipients. Recipient and clinic partner staff answered unique sets of pre-interview questions to inform tailoring of interview guides that were developed using constructs from the Framework for Reporting Adaptations and Modifications to Evidence-based Implementation Strategies (FRAME-IS) and Consolidated Framework for Implementation Research (CFIR). The study team then interviewed recipient, health system, and clinic partner staff incorporating pre-interview responses to focus each conversation. We employed a rapid qualitative analysis approach then conducted virtual focus groups with recipient representatives to validate emergent themes. Three modifications that emerged from thematic analysis include: (1) offering mailed fecal immunochemical test (FIT) kits for CRC screening with mail or drop off return; (2) increasing the use of patient education and engagement strategies; and (3) increasing the use of or improving automated patient messaging systems. With improved tracking and automated reminder systems, mailed FIT kits paired with tailored patient education and clear instructions for completing the test could help primary care clinics catch up on the backlog of missed screenings during COVID-19. Future research can assess the effectiveness and cost-effectiveness of offering mailed FIT kits on maintaining or improving CRC screening, especially among people who are medically underserved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The COVID-19 pandemic led to temporary clinic closures[1]; diversion of healthcare resources, including staff, to address the pandemic [2]; postponement of non-urgent medical procedures including colorectal cancer (CRC) screenings [3,4,5]; social distancing practices to slow the spread of disease [6]; and a reluctance of patients to seek care for non-emergent conditions based on fear of infection [7], which collectively contributed to a decline in cancer screening rates [2, 5, 8,9,10]. Recent estimates indicate that screenings for breast, colon, and cervical cancer in the United States declined by as much as 80% to 90% in April 2020 compared to pre-pandemic levels [11, 12].
Within Federally Qualified Health Centers (FQHCs) that provide primary care services to people who are medically underserved, the percentage of patients ages 50–75 years who were up to date on CRC screening was well below the national average of 67.1% [13] prior to the pandemic, declining from 45.6% in 2019 to 40.1% in 2020 [13, 14]. Delays in CRC screening, including missed screening cycles, may lead to an increase in later-stage at diagnosis, increased CRC deaths, and greater CRC disparities in people who are medically underserved and disproportionately affected by COVID-19 [3, 5, 10, 15,16,17].
Maintaining CRC screening and catching up on the backlog of screenings missed during the initial waves of the COVID-19 pandemic can mitigate further disparities in CRC outcomes. The Centers for Disease Control and Prevention (CDC) has made investments in the Colorectal Cancer Control Program (CRCCP). The CRCCP is based on a health systems change model that promotes integrating public health and primary care to improve population health, specifically to mitigate disparities by increasing CRC screening in clinics with low screening rates that primarily serve people with lower incomes [18].
In July 2020, CDC funded a 5-year cooperative agreement (DP20-2002) with 35 CRCCP award recipients (hereafter referred to as recipients) that partner with health systems and primary care clinics (hereafter referred to as clinic partners) to implement evidence-based interventions (EBIs) recommended by the Community Preventive Services Task Force in The Community Guide [19] to increase uptake of CRC screening [20]. Participating clinic partners worked with recipients and other partners (e.g., primary care associations, health center control networks) to implement EBIs including provider assessment and feedback, provider reminders, patient reminders, and reducing structural barriers to screening based on clinic priorities and feasibility.
This study aimed to understand how CRCCP clinic partners may have modified EBI implementation to promote CRC screening during the COVID-19 pandemic; to identify barriers and facilitators to implementing modifications; and to extract lessons that can be applied to support CRC screening, chronic disease management, and clinic resilience in the face of future public health crises. This study also contributes to an emerging body of literature on health system resilience [21,22,23,24] by identifying factors that supported or hindered clinic partners’ ability to adapt priorities to address the effects of the COVID-19 pandemic on continuing CRC screening promotion efforts.
Methods
Guiding Theories
Constructs from the Framework for Reporting Adaptations and Modifications to Evidence-based Implementation Strategies (FRAME-IS) [25, 26] and Inner and Outer Setting domains of the Consolidated Framework for Implementation Research (CFIR) [27] were used to guide development of the evaluation questions and data collection tools. Given that there is no singular definition or construct for measuring resilience in health systems [24, 28, 29], we used Mallak’s definition of resilience [30]. Table 1 presents definitions of each theory’s constructs or domains that informed our study and example evaluation questions aimed at understanding how each construct influenced clinic partners’ modifications to EBI implementation. Additional File 1 includes all evaluation questions.
Site Selection and Recruitment
We used purposive sampling to select nine recipients—Alaska Native Tribal Health Consortium (ANTHC), Georgia Center for Oncology Research and Education (Georgia CORE), Iowa Department of Public Health, New York Department of Public Health, Rhode Island Department of Public Health, University of Arkansas System, University of Chicago Medicine, University of Missouri, and University of Texas Health Science Center at Houston—for participation in this study.
Recipients were selected to reflect the diversity inherent among all CRCCP recipients. Factors considered included priority population(s) served, recipient geographic region, previous CRCCP funding, and the type of recipient organization (Table 2). The selected recipients are all participants in the CRCCP Learning Collaborative, a project designed to develop and apply a standardized approach to evaluate the implementation, effectiveness, cost, and cost-effectiveness of multi-component interventions to inform future scale-up of these interventions. Selected recipients were willing to participate in the study and identified one of their partner health systems and a clinic partner within that health system to participate in the study.
Data Collection and Analysis
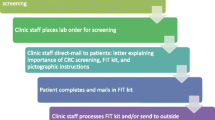
Our iterative approach to data collection and analysis began with having one representative from each of the nine recipient sites and one representative from each recipient’s selected clinic partner answer unique sets of pre-interview questions to inform tailoring of interview guides. No more than nine participants responded to each set of pre-interview questions. We then interviewed recipient, health system, and clinic partner staff, incorporating pre-interview responses to focus each conversation. We selected these participant types to gather input from multiple perspectives. Finally, we conducted focus groups with recipient representatives to validate preliminary findings from the interviews (Fig. 1). Institutional Review Board approval was not required for this data collection because it did not constitute human subjects research.
Pre-interview Questions
Recipient representatives from all nine programs and their participating clinics responded to pre-interview questions to provide contextual information relevant to our discussions, including (1) confirmation of the recipient’s selection of EBIs for clinic partners to implement, (2) confirmation of the EBIs implemented at the clinic partner site included in the study, and (3) the extent to which recipients and clinic partners agreed or disagreed with several statements describing the impact of the COVID-19 pandemic on EBI implementation. We used unique sets of pre-interview questions for recipient and clinic partner respondent types; we did not administer pre-interview questions to health system respondents to minimize respondent burden, and because we anticipated clinic staff would be best suited to respond to our line of questioning. A summary of pre-interview questions and responses are provided in Additional File 2. We used the responses to tailor interview guides for recipient, health system, and clinic partner staff.
Key Informant Interviews
We developed unique interview guides for each of the three participant roles: recipient, partner health system, and clinic partner. No more than nine participants of each type were interviewed. Interview guide topics included modifications to EBI implementation at the clinic level because of COVID-19 (including the effect of testing and vaccination efforts and patient safety concerns); factors that influenced clinics’ ability to continue implementing CRC EBIs; and the kinds of support clinics needed from the recipient and health system to implement CRC EBIs. Sample interview questions are provided in Additional File 3.
For each selected recipient, we first interviewed the recipient-level participant, followed by the health system participant and the clinic participant. In total, 27 individuals participated in semi-structured virtual interviews conducted via Zoom between August 2021 and March 2022. All participants gave verbal consent to participate in interviews and for audio recording. Recordings were transcribed for analysis.
A lead note-taker documented detailed notes during each interview in an Excel spreadsheet that mapped participant responses to interview guide topics. A team of three analysts abstracted interview data from the Excel spreadsheet for each of the three respondent groups, referring to interview transcripts as necessary for clarity or examples, and then met to refine a list of preliminary themes mentioned by at least three participants to validate with recipient representatives.
Focus Groups
Following the completion of key informant interviews and preliminary analysis of detailed interview notes, the study team conducted two focus groups with three and six recipient representatives, respectively. The purpose of the focus groups was to (1) get feedback on emergent themes reflecting common modifications that partner clinics made to their EBI implementation processes to continue their CRC screening promotion efforts, and (2) probe on barriers and facilitators to implementing these modifications.
During each focus group, a lead moderator presented the emergent themes, invited participants to respond to a poll to document their level of agreement with each theme, and led participants in a discussion to refine each theme and identify barriers and facilitators to implementing each modification.
A lead note-taker and co-moderator documented detailed notes during each focus group in an Excel spreadsheet that mapped focus group participant responses to preliminary themes, including the level of agreement with each theme, factors that facilitated implementation of the modification, and challenges encountered when implementing the modification. Two analysts conducted a primary and secondary review of detailed notes from each focus group to document participants’ examples of agreement and disagreement with each modification, and to identify common facilitators and challenges related to each modification. A third analyst conducted a final review and adjudication of validated themes, which are presented in the results. Validated themes were supported by the majority of recipient respondents (five or more). In some cases, we included examples within each validated theme that reflected the specific experiences of only one focus group respondent when the respondent’s example was particularly compelling.
Results
We identified five initial modifications that emerged from thematic analysis of interview data:
-
Offering mailed fecal immunochemical test (FIT) kits for CRC screening with mail or drop off return.
-
Increasing the use of patient education and engagement strategies.
-
Increasing the use of or improving automated patient messaging systems.
-
Adapting clinic workflows and patient flow in clinics.
-
Reducing financial barriers to CRC screening.
Figure 2 shows focus group participant level of agreement with the extent to which each modification was reflected in their clinic partner activities. Of these five initial themes, three were validated among focus group respondents. Most respondents agreed that their clinic partners increased use of mailed FIT kits with mail or drop off return options and focused on patient education and engagement to promote CRC screening in response to COVID-19. About half of respondents agreed that their clinic partners improved or increased use of automated patient messaging systems to promote CRC screening. However, few respondents agreed that their partners were adapting clinic or patient flow or reducing financial barriers as modifications specific to increasing CRC screening in response to COVID-19. For example, respondents described clinic workflows that were adapted to increase safety and encourage patients to resume seeking in-person health care when necessary but said that this was not directly focused on promotion of CRC screening. Further, respondents noted that although reducing financial barriers for CRC screening such as covering the cost of colonoscopies for uninsured patients and the cost of transportation to colonoscopy appointments is important, clinics were already doing this and this was not directly tied to COVID-19. We therefore describe and present illustrative quotes for the three validated themes only: offering mailed FIT kits with mail or drop-off return, increasing the use of patient education and engagement strategies, and increasing the use of, or improving automated patient messaging systems.
Offering Mailed FIT Kits with Mail or Drop-Off Return
Increased use of mailed FIT kits with mail or drop-off return to encourage patients to engage in CRC screening emerged as a theme across sites during interviews. Interview respondents discussed increasing use of mailed FIT or other stool-based tests over referrals for screening colonoscopy; using mailed FIT kits, some with labels filled in or self-addressed envelopes to facilitate return; and offering contactless FIT drop-off options.
“… a lot of people aren’t coming in. So the effort that we are doing this fall is to mail [FIT kits] to people who are eligible, based on the data in our electronic health record (EHR). So this is a couple thousand people … where we would rather take the risk of having FIT tests get tossed in the trash. We’re going to accept that cost because it’s worth it to us. If we got 500 of them back, that would be fantastic.”—Health System Interview Respondent
“… I would say that the mailing of the FIT test probably, that’s [something] that we had an increase of because of COVID so the patient wouldn’t have to come into the clinic necessarily to get the FIT test. We’ve been able to mail them out and then the patient completes it and mails it back.”—Clinic Interview Respondent
“With the pandemic, again, it was more FIT kits because... there weren’t GI [gastrointestinal] offices that were open [and conducting non-urgent medical procedures during the initial wave of the pandemic] so that wasn’t going to be feasible for patients…[FIT] was something we were already doing, but [the pandemic] compounded it more to be … better for patients.”—Health System Interview Respondent
In discussing the interview data around this theme, most focus group respondents agreed that increased use of mailed FIT kits with mail or drop-off return options was a modification that their clinic partners adopted to promote CRC screening in response to COVID-19. Respondents shared that although FIT kits were already included as an option for CRC screening, mailed FIT kits were prioritized to reduce potential exposure to COVID-19 and because patients were not able or willing to come in person to clinic sites.
“[Some] clinics have really made significant improvements in terms of mailing kits prior to patient appointments or using some automated messaging to remind patients to send them in. So really enhancing what they’ve done.”—Recipient Focus Group Respondent
“Again, when we had surveyed our federally qualified health centers, a hundred percent of them came back and stated that they did increase FIT screening during COVID.”—Recipient Focus Group Respondent
Respondents noted challenges and facilitators to use of mailed FIT kits. Challenges included insufficient capacity in existing systems to track distribution and return of FIT kits and reluctance from clinicians to support use of a screening tool other than colonoscopy. Respondents also described the challenge of receiving incomplete or incorrectly completed FIT tests due to patient misunderstanding around instructions to complete FIT testing which could have occurred because of cultural/linguistic barriers or a breakdown in communication when patients do not receive instructions in person.
“Especially during COVID, I’ve thought that an at-home test would be phenomenally well appreciated and people interested in it. And certainly patients have been interested, but providers have been very reluctant… feeling that’s an inferior test compared to colonoscopy and that people should really wait for colonoscopy.”—Recipient Focus Group Respondent
“We had huge success with prioritizing FIT kits … but the tracking was something that really had to be prioritized as well, because they were consistently giving FIT kits out and having no idea how many were given, how many were completed.”—Recipient Focus Group Respondent
“...one of the issues that we saw with increasing the mailed FIT was just culturally or linguistically appropriate instructions. Even if patients were called, were spoken to over the phone or even if they did like a video telehealth meeting there, something gets lost in translation there.”—Recipient Focus Group Respondent
For the reluctance to support use of CRC screening tests other than colonoscopy, recipients described the importance of support from clinic champions and framing of messaging to clinic staff to get them on board with using mailed FIT kits to reach more patients, particularly during a time when clinics were facing high rates of staff turnover and patients were not able or willing to come into the clinic. One recipient focus group respondent shared that clinician education was equally as important as patient education in promoting the importance of CRC screening during the pandemic. They explained that high rates of clinic staff turnover necessitated development and dissemination of effective onboarding materials to train new clinical staff on COVID-19 protocols, including processes for disseminating and collecting completed FIT kits, and to reinforce clinic commitment to promotion of CRC screening despite competing demands.
“For us, it was the clinic champions… the ones who were very much into the data and saw that we have all these people due for screening … and so they as leaders said, ‘Okay, we’re going to have to use FITs as well because we’re not going to get everyone screened with colonoscopy alone.’ And for us, that was what really helped propel the conversation.”—Recipient Focus Group Respondent
“In some of our clinic areas, [patients can wait up to] a year for a colonoscopy. And even the clinicians who think it’s the gold standard we’ve been able to get them to the point of ‘the best test is the one that gets done.’ So it’s been a spectrum, but [communication to providers about the benefits of FIT] has helped us get some buy-in to these other screening options.”—Recipient Focus Group Respondent
“We have developed a virtual education platform for providers, as well as staff. This was directly spurred on by COVID, because we decided it was best to be able have a mechanism [for staff education] as staff turned over.”—Recipient Focus Group Respondent
Increasing the Use of Patient Education and Engagement Strategies
Respondents reported increasing use of patient education and engagement strategies as a key clinic partner modification. Interview respondents discussed having patient navigators and clinic staff use motivational interviewing to acknowledge patients’ fears and talk through safety measures in place to make in-person visits to clinics and CRC screening safe. They also discussed designing infographic materials to reassure people that clinics were operating with patient safety as a priority.
“We try to relate this [patient safety information] to the patients as much as possible through verbal conversations. We also put information on our social media sites, our website, emails, on our patient portals. We try to get it out there as much as possible. We have signage in our clinics, letting people know all of this.”—Clinic Interview Respondent
In responding to interview data supporting this theme, most focus group respondents agreed that patient education and engagement was central to their clinic partners’ efforts to promote CRC screening while responding to COVID-19.
“[Some] clinics realized that they could be using patient education more so than they had been. [Saying] ‘I don’t know why we weren’t doing this all this time. It just makes sense.’ So we saw strong success with it.”—Recipient Focus Group Respondent
Respondents shared specific examples of activities intended to educate and engage patients around CRC screening including the use of small media (i.e., materials tailored to patient health literacy levels, t-shirts worn by clinicians to promote screening, infographics to convey FIT test instructions and follow-up steps, posters tailored to specific racial or ethnic groups with QR codes that link to CRC screening education placed in COVID-19 vaccination waiting areas, or direct mailings to patients) and one-on-one patient education via phone calls.
“We developed [CRC screening] education materials like posters tailored to race ethnicity and in multiple languages with the QR code. And we distributed these to all our [health] systems because we know patients are waiting for 15 minutes after [their] COVID vaccine. We broke [messages] down by race ethnicity.”—Recipient Focus Group Respondent
Respondents noted challenges and facilitators to promoting patient education and engagement around CRC screening while responding to COVID-19. Challenges identified included staffing shortages and crafting messages that would not be overlooked or exacerbate patients’ feelings of being overwhelmed during the pandemic. Facilitators to effective patient education and engagement noted by respondents included staff enthusiasm for patient education and particularly the opportunity for care teams to focus on something other than COVID-19.
“I would say the zeal of the team members that are doing patient education was a huge...Sometimes when they meet with us, they’re like, “Oh, this is a breath of fresh air. We’re so tired of working with COVID. This is a chance to focus on something and get invigorated about something else than COVID.”—Recipient Focus Group Respondent
Respondents also noted using electronic medical record (EMR) data to identify patients with upcoming appointments who are due for screening so that patient navigators could conduct educational outreach and offer to mail a FIT kit ahead of the patient’s upcoming appointment.
Increasing the Use of or Improving Automated Patient Messaging Systems
During interviews, respondents discussed benefiting from increasing use of automated patient messaging systems while responding to COVID-19 to disseminate CRC screening reminders and establishing or streamlining FIT kit standing orders to reduce burden on heavily strained clinic staff.
“Initially we were trying to do a lot of manual reminders. But over time during COVID we’ve, as everybody else has, had staffing issues. We’ve lost a lot of staff, have had staff out sick, we just don’t have enough staff. So we’ve had to transition to more automated reminder approaches.”—Health System Interview Respondent
“One of the systems that we work with… started using this messaging system … to notify patients of their COVID results. When we started having conversations about EBIs with them, they were quick to jump on it [saying], ‘You know what? We already do this. We can use a system like that to remind patients to return the kits.’”—Recipient Interview Respondent
“We put a standing order in place where ... [patient navigator] can reach out to a patient to see if they’re due for screening and mail the FIT to them. So they only need to come in once to drop it off, and then we have a drop box outside the clinic. And I think that really helped during the pandemic because [patients] didn’t need to come inside the clinic.”—Clinic Interview Respondent
A majority of focus group respondents agreed that increased reliance on automated patient messaging systems to reach patients was a modification implemented by their clinic partners to promote CRC screening during COVID-19. Respondents shared that the push to overhaul systems to accommodate patient communication around COVID-19 testing and vaccination had a positive impact on automation of reminders for CRC screening. Clinics described efforts to streamline automation through bundled orders (i.e., combining multiple patients in a single FIT kit order that automatically includes referrals for patient navigation) that reduced the number of “clicks” necessary for clinicians to refer patients for CRC screening and broadening access of order privileges to patient navigators or other non-clinicians as a means to reduce barriers to getting FIT kits in patients’ hands.
“I think the automated systems is an absolutely huge piece to our clinics ... colorectal cancer just being one part of what they’re doing, if we can create a system that is as automated as possible, that cuts down the amount of work that your staff members have to do to get this done, I think that’s a huge win.”—Recipient Focus Group Respondent
“I would just say that I know most major institutions have had to add an entire layer of the [automation] to operate COVID protocols...You saw a lot of gaps. You saw a lot of loops that weren’t closed…it was very noticeable that complete overhauls of systems had to be completed in order for the work to get done.”—Recipient Focus Group Respondent
“There was one workflow that our clinic liaison implemented to make it easier to order [referrals]. They decreased it from 30 clicks down to one or two, and that would include not only the FIT kit, but the link to the [patient] navigators.”—Recipient Focus Group Respondent
Some respondents expressed support for streamlining and automation but said that their clinic partners were overwhelmed with COVID-19 and did not have the staff capacity or resources to automate systems that were not already in place. Others noted that outdated systems and lack of funding were barriers to updating or improving system automation.
“Outdated electronic medical records (EMRs) or being able to provide funding for someone’s time in order to make these improvements that’s really what we hear a lot of the time in response to updating or changing these automated reminder systems.”—Recipient Focus Group Respondent
Discussion
Healthcare systems were grappling with the first wave of the COVID-19 pandemic when the current round of CRCCP funding (DP20-2002) was awarded in July 2020. Given significant healthcare infrastructure challenges [2], clinics had to focus on COVID-19 and patient safety concerns thus directing attention away from non-urgent medical procedures, including CRC screening. In response, some CRCCP clinic partners modified their practices to try and maintain CRC screening. This study aimed to understand how health systems and clinics pivoted during this public health crisis to continue providing preventive services including CRC screening. Lessons learned can be used by the CRCCP, other chronic disease programs, and primary care clinic teams to support clinic resilience to continue promoting CRC and other cancer screening during infectious disease outbreaks or acute public health emergencies that may place significant burden on systems of care [23, 32,33,34].
Using an iterative approach to qualitative data collection and analysis, our study yielded three validated themes that describe how clinics modified CRC screening promotion efforts during the COVID-19 pandemic: (1) offering mailed FIT kits for CRC screening with mail or drop-off return, (2) increasing the use of patient education and engagement strategies, and (3) increasing the use of or improving automated patient messaging systems.
Among CRC screening test options, colonoscopy is the most frequently used CRC screening test and stool-based tests are the second most frequently used [35, 36]. FIT is an annual stool-based test that can be completed at home [37] and the kits have traditionally been offered to patients during in-person primary care visits. Mailed FIT outreach programs have been shown to increase CRC screening rates and to be cost-effective [38,39,40,41], but were not widely adopted in primary care clinics prior to the COVID-19 pandemic [42]. This public health emergency served as a catalyst for primary care clinics to adopt mailed FIT outreach as a means to continue promoting CRC screening while addressing patient hesitancy to attend in-person appointments, reducing patient risk for COVID-19 exposure and addressing the issue of postponed or missed non-urgent procedures (including screening colonoscopies) during periods of high infection rates [10, 15, 16, 43]. Consistent with previous studies [42], we found that mailed FIT outreach is challenging without adequate staff capacity and systems for tracking FIT kit dissemination and return and follow-up to ensure patients with positive test results complete a timely colonoscopy. Developing stand-alone tracking databases and/or increasing use of automated systems embedded within clinic EMRs may improve FIT kit tracking and facilitate dissemination of patient reminders to complete and return their kits [42]. Participants also reported reluctance among some clinicians to embrace increased use of FIT kits vs. continued promotion of screening colonoscopies, even temporarily [44,45,46] as a challenge. In these cases, participants noted that clinic champions were essential to fostering clinician and leadership buy-in for using mailed FIT kits as well as supporting improved tracking systems that would reduce burden on staff. Despite these challenges, mailed FIT outreach holds promise as an effective, sustainable, and scalable approach to catching up on missed CRC screenings, and is likely to remain an adopted CRC screening option [42, 47,48,49,50,51]. Health systems and clinics may consider adopting a plan for incorporating mailed FIT outreach into their programs to facilitate continued CRC screening in the event of a future public health emergency and to provide patients with another option for screening, which has been shown to improve CRC screening rates [52].
Increased patient education and engagement emerged as a modification to address challenges around receipt of completed FIT kits that did not adhere to requirements and were, therefore, unusable because they were missing the collection date or received more than 14 days after the sample was collected. A recent study conducted within a safety-net based health system estimated that nearly one in five (19.8%) samples returned from mailed FIT outreach were unusable, primarily due to an undocumented collection date [53]. During the COVID-19 pandemic, receipt of unusable FIT kits may have placed additional burden on already overwhelmed staff to follow up with patients and request that they complete another FIT kit. In response to this challenge, clinic staff bolstered their patient education protocols to increase clarity among patients about how to correctly complete and return kits. The recently published Mailed FIT Implementation Guide (2021) recommends additional strategies to minimize receipt of unusable tests including: sending patients advance notification via an introductory letter, email, phone call, or text message to let them know why they need CRC screening and that they will receive a mailed FIT that will need to be completed and returned within a specified timeframe (e.g., 7 days); and mailing FIT kits with customized print and graphic instructions, a return envelope, and tailored FIT labels that include the patient’s medical record number, date of birth, and the collection date field highlighted in yellow to ensure patients provide the collection date [54].
Health systems and clinics also used automated patient messaging systems that were put in place to deliver messaging to support CRC screening (e.g., return of FIT kits), a modification that reduced burden on overwhelmed staff. Although institutionalizing EBIs is an established mechanism for sustainment [55, 56] our study indicates that automation of EBIs may be even more beneficial in helping healthcare systems deal with the challenges that public health emergencies may introduce. Automated reminders may be more effective when paired with culturally appropriate patient education and follow-up from a patient navigator or community health worker. Additionally, clinicians can be made aware of, and trained on automated systems, which can be difficult when faced with emergent priorities or during periods of high staff turnover. Web-based training for clinicians on automated systems and follow-up procedures could be a solution for this challenge as well.
Lessons Learned Regarding Clinic Resilience During Public Health Emergencies
Findings from this study revealed factors that can support maintenance of CRC screening promotion efforts during future public health emergencies, such as COVID-19. First, documentation of CRC screening protocols within clinics facilities may smooth onboarding of clinicians, particularly considering rapid staff turnover that can occur during a public health crisis. Specifically, documenting processes and educating clinicians on FIT kit dissemination, patient reminders to complete and return FIT kits, and tracking of dissemination and completion; using automated patient messaging systems to support patient reminders; and delivering patient education can help mitigate disruptions in CRC screening promotion efforts. Second, consistent with previous research, including of the CRCCP [18, 57, 58], clinic champions can help promote CRC screening and maintain clinic staff buy-in to modify existing practices despite competing priorities. Third, support from clinic leadership is essential for continuing CRC screening promotion efforts and exploring innovative solutions to emergent challenges, especially when faced with limited clinic resources and staff capacity [42, 59]. Finally, fostering inclusion, belonging, and a shared purpose among clinic staff to deliver high-quality care to the community they serve can facilitate adoption and implementation of modified processes.
Study Limitations
This study had at least three limitations. First, we engaged representatives from select CRCCP recipients and their health system and clinic partners. Therefore, findings from this study reflect perspectives from a small number of respondents and are not generalizable to all CRCCP recipients or their health system and clinic partners. Second, we employed a qualitative analysis approach that relied primarily on interview notes to identify emergent themes. Although this approach was beneficial for expediting analysis and was strengthened with the engagement of recipient representatives to validate our findings, we did not conduct line-by-line coding of interview and focus group transcripts and therefore may have missed some examples related to each modification. Third, we did not collect or assess CRC screening outcomes and therefore are unable to draw conclusions about the impact each modification had on CRC screening rates within the participating health systems and clinics.
Conclusion
COVID-19 caused significant declines in cancer screening, including for CRC. Faced with this new, rapidly spreading infectious disease, many health systems and clinics had to quickly adapt to address the pandemic and maintain patient safety. Our study found that promoting the use of fecal CRC screening tests by mailing FIT kits and providing mail or drop-off return options was a solution that supported continuation of CRC screening during a time when patients were not able or willing to visit clinic facilities. With improved tracking and automated reminder systems, mailed FIT kits paired with tailored patient education and clear instructions for completing the test may help primary care clinics catch up on the backlog of missed screenings during the COVID-19 pandemic. Future research could assess the effectiveness and cost-effectiveness of offering mailed FIT kits on maintaining or improving CRC screening rates, including among people who are medically underserved, during public health crises.
Future research could also explore whether and how these modifications were sustained during the COVID-19 pandemic to inform efforts for future public health emergencies. And finally, it may be important to monitor whether and how health systems and clinics are ensuring timely follow-up colonoscopies for positive FIT tests, particularly among FQHC patients where coordination with external gastroenterologists remains a challenge in part because of insufficient health system capacity [3, 60].
Data Availability
The qualitative data generated or analyzed during the current study are not publicly available because they were generated in interviews conducted by the research team, with the expectation that participant identity would be kept confidential.
Code Availability
Not applicable.
References
Health Resources Services Administration, Bureau of Primary Health Care. Health Center COVID-19 Survey. Retrieved March 30, 2022, from https://data.hrsa.gov/tools/data-reporting/program-data/national
Cancino, R. S., Su, Z., Mesa, R., Tomlinson, G. E., & Wang, J. (2020). The impact of COVID-19 on cancer screening: Challenges and opportunities. JMIR Cancer, 6(2), e21697. https://doi.org/10.2196/21697
Balzora, S., Issaka, R. B., Anyane-Yeboa, A., Gray, D. M., & May, F. P. (2020). Impact of COVID-19 on colorectal cancer disparities and the way forward. Gastrointestinal Endoscopy, 92(4), 946–950. https://doi.org/10.1016/j.gie.2020.06.042
Issaka, R. B., & Somsouk, M. (2020). Colorectal cancer screening and prevention in the COVID-19 era. JAMA Health Forum, 1(5), e200588–e200588. https://doi.org/10.1001/jamahealthforum.2020.0588
Harber, I., Zeidan, D., & Aslam, M. N. (2021). Colorectal cancer screening: Impact of COVID-19 pandemic and possible consequences. Life (Basel, Switzerland), 11(12), 1297. https://doi.org/10.3390/life11121297
Hess, R., Mao, J. Z., Rho, K., Khan, A., Levy, E., Pollina, J., & Mullin, J. P. (2020). Embracing disruptive change during the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. World Neurosurgery, 139, 726.
Fisher-Borne, M., Isher-Witt, J., Comstock, S., & Perkins, R. B. (2021). Understanding COVID-19 impact on cervical, breast, and colorectal cancer screening among federally qualified healthcare centers participating in “Back on track with screening” quality improvement projects. Preventive Medicine, 151, 106681. https://doi.org/10.1016/j.ypmed.2021.106681
Chen, R. C., Haynes, K., Du, S., Barron, J., & Katz, A. J. (2021). Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncology, 7(6), 878–884. https://doi.org/10.1001/jamaoncol.2021.0884
DeGroff, A., Miller, J., Sharma, K., Sun, J., Helsel, W., Kammerer, W., Rockwell, T., Sheu, A., Melillo, S., Uhd, J., Kenney, K., Wong, F., Saraiya, M., & Richardson, L. C. (2021). COVID-19 impact on screening test volume through the National Breast and Cervical Cancer Early Detection Program, January–June 2020, in the United States. Preventive Medicine, 151, 106559. https://doi.org/10.1016/j.ypmed.2021.106559
Kadakuntla, A., Wang, T., Medgyesy, K., Rrapi, E., Litynski, J., Adynski, G., & Tadros, M. (2021). Colorectal cancer screening in the COVID-19 era. World Journal of Gastrointestinal Oncology, 13(4), 238–251. https://doi.org/10.4251/wjgo.v13.i4.238
Epic Health Research Network. (2020). Delayed cancer screenings. Cosmos Study. Retrieved May 24, 2022, from https://epicresearch.org/articles/delays-in-preventive-cancer-screenings-during-covid-19-pandemic
Martin, K., Kurowski, D., Given, P., Kennedy, K., & Clayton, E. (2021). The impact of COVID-19 on the use of preventive health care. HCCI’s original reports powered by #HCCIdata. Retrieved May 24, 2022, from https://healthcostinstitute.org/hcci-research/the-impact-of-covid-19-on-the-use-of-preventive-health-care
Centers for Disease Control and Prevention, National Center for Health Statistics. (2022). Colorectal cancer screening. National Health Interview Survey, 1987–2019. Retrieved May 24, 2022, from https://progressreport.cancer.gov/detection/colorectal_cancer#:~:text=In%202019%2C%2067.1%25%20of%20adults,on%20the%20most%20recent%20guidelines
Health Resources & Services Administration. (n.d.). National Health Center Program Uniform Data System (UDS) Awardee Data. Health Center Program UDS Data: Data Overview. Retrieved May 24, 2022, from https://data.hrsa.gov/tools/data-reporting/program-data/national
Nodora, J. N., Gupta, S., Howard, N., Motadel, K., Propst, T., Rodriguez, J., Schultz, J., Velasquez, S., Castañeda, S. F., Rabin, B., & Martínez, M. E. (2020). The COVID-19 pandemic: Identifying adaptive solutions for colorectal cancer screening in underserved communities. JNCI Journal of the National Cancer Institute, 113(8), 962–968. https://doi.org/10.1093/jnci/djaa117
Kopel, J., Ristic, B., Brower, G. L., & Goyal, H. (2022). Global impact of COVID-19 on colorectal cancer screening: Current insights and future directions. Medicina (Kaunas, Lithuania), 58(1), 100. https://doi.org/10.3390/medicina58010100
Karmakar, M., Lantz, P. M., & Tipirneni, R. (2021). Association of social and demographic factors with COVID-19 incidence and death rates in the US. JAMA Network Open, 4(1), e2036462–e2036462.
DeGroff, A., Sharma, K., Satsangi, A., Kenney, K., Joseph, D., Ross, K., Leadbetter, S., Helsel, W., Kammerer, W., Firth, R., Rockwell, T., Short, W., Tangka, F., Wong, F., & Richardson, L. (2018). Increasing colorectal cancer screening in health care systems using evidence-based interventions. Preventing Chronic Disease, 15, E100. https://doi.org/10.5888/pcd15.180029
The Community Guide (2019). Cancer. Retrieved May 24, 2022, from https://www.thecommunityguide.org/topic/cancer
Centers for Disease Control and Prevention, Colorectal Cancer Control Program (CRCCP). About us. Cancer Home. Retrieved May 24, 2022, from https://www.cdc.gov/cancer/crccp/index.htm
Kruk, M. E., Myers, M., Varpilah, S. T., & Dahn, B. T. (2015). What is a resilient health system? Lessons from Ebola. The Lancet, 385(9980), 1910–1912. https://doi.org/10.1016/S0140-6736(15)60755-3
Meyer, D., Bishai, D., Ravi, S. J., Rashid, H., Mahmood, S. S., Toner, E., & Nuzzo, J. B. (2020). A checklist to improve health system resilience to infectious disease outbreaks and natural hazards. BMJ Global Health, 5(8), e002429. https://doi.org/10.1136/bmjgh-2020-002429
Thomas, S., Sagan, A., Larkin, J., Cylus, J., Figueras, J., & Karanikolos, M. (2020). Strengthening health systems resilience: Key concepts and strategies: European observatory on health systems and policies, Copenhagen (Denmark).
Turenne, C. P., Gautier, L., Degroote, S., Guillard, E., Chabrol, F., & Ridde, V. (2019). Conceptual analysis of health systems resilience: A scoping review. Social Science & Medicine, 232, 168–180. https://doi.org/10.1016/j.socscimed.2019.04.020
Miller, C. J., Barnett, M. L., Baumann, A. A., Gutner, C. A., & Wiltsey-Stirman, S. (2021). The FRAME-IS: A framework for documenting modifications to implementation strategies in healthcare. Implementation Science, 16(1), 36. https://doi.org/10.1186/s13012-021-01105-3
Wiltsey Stirman, S., Baumann, A. A., & Miller, C. J. (2019). The FRAME: An expanded framework for reporting adaptations and modifications to evidence-based interventions. Implementation Science, 14(1), 58. https://doi.org/10.1186/s13012-019-0898-y
Damschroder, L. J., Aron, D. C., Keith, R. E., Kirsh, S. R., Alexander, J. A., & Lowery, J. C. (2009). Fostering implementation of health services research findings into practice: A consolidated framework for advancing implementation science. Implementation Science, 4(1), 50. https://doi.org/10.1186/1748-5908-4-50
Fridell, M., Edwin, S., von Schreeb, J., & Saulnier, D. D. (2020). Health system resilience: What are we talking about? A scoping review mapping characteristics and keywords. International Journal of Health Policy and Management, 9(1), 6–16. https://doi.org/10.15171/ijhpm.2019.71
Barasa, E., Mbau, R., & Gilson, L. (2018). What is resilience and how can it be nurtured? A systematic review of empirical literature on organizational resilience. International Journal of Health Policy and Management, 7(6), 491–503. https://doi.org/10.15171/ijhpm.2018.06
Mallak, L. A. (1998). Measuring resilience in health care provider organizations. Health Manpower Management, 24(4–5), 148–152. https://doi.org/10.1108/09552069810215755
Coronado, G. D., Schneider, J. L., Green, B. B., Coury, J. K., Schwartz, M. R., Kulkarni-Sharma, Y., & Baldwin, L. M. (2020). Health plan adaptations to a mailed outreach program for colorectal cancer screening among Medicaid and Medicare enrollees: The BeneFIT study. Implementation Science, 15(1), 77–77. https://doi.org/10.1186/s13012-020-01037-4
Carroll, R., Duea, S. R., & Prentice, C. R. (2022). Implications for health system resilience: Quantifying the impact of the COVID-19-related stay at home orders on cancer screenings and diagnoses in southeastern North Carolina, USA. Preventive Medicine, 158, 107010. https://doi.org/10.1016/j.ypmed.2022.107010
Steben, M., Norris, T., Rosberger, Z., & Action, H. P. V. G. (2020). COVID-19 won’t be the last (or worst) pandemic: It’s time to build resilience into our cervical cancer elimination goals. Journal of Obstetrics and Gynaecology Canada, 42(10), 1195–1196. https://doi.org/10.1016/j.jogc.2020.08.006
Forcellini, D. (2022). A resilience-based (RB) methodology to assess resilience of health system infrastructures to epidemic crisis. Applied Sciences, 12(6), 3032.
Adam, E. E., White, M. C., & Shapiro, J. A. (2022). Prevalence of colorectal cancer screening test use by test type and age among older adults in the United States. Journal of the American Geriatrics Society. https://doi.org/10.1111/jgs.17890
Shapiro, J. A., Soman, A. V., Berkowitz, Z., Fedewa, S. A., Sabatino, S. A., de Moor, J. S., Clarke, T. C., Doria-Rose, V. P., Breslau, E. S., Jemal, A., & Nadel, M. R. (2021). Screening for colorectal cancer in the United States: Correlates and time trends by type of test. Cancer Epidemiology, Biomarkers & Prevention, 30(8), 1554–1565. https://doi.org/10.1158/1055-9965.EPI-20-1809
U.S. Preventive Services Task Force. (2021). Final recommendation statement. Colorectal cancer: Screening. Retrieved from https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
Jager, M., Demb, J., Asghar, A., Selby, K., Mello, E. M., Heskett, K. M., Lieberman, A. J., Geng, Z., Bharti, B., Singh, S., & Gupta, S. (2019). Mailed outreach is superior to usual care alone for colorectal cancer screening in the USA: A systematic review and meta-analysis. Digestive Diseases and Sciences, 64(9), 2489–2496. https://doi.org/10.1007/s10620-019-05587-6
Dougherty, M. K., Brenner, A. T., Crockett, S. D., Gupta, S., Wheeler, S. B., Coker-Schwimmer, M., Cubillos, L., Malo, T., & Reuland, D. S. (2018). Evaluation of interventions intended to increase colorectal cancer screening rates in the United States: A systematic review and meta-analysis. JAMA Internal Medicine, 178(12), 1645–1658. https://doi.org/10.1001/jamainternmed.2018.4637
Somsouk, M., Rachocki, C., Mannalithara, A., Garcia, D., Laleau, V., Grimes, B., Issaka, R. B., Chen, E., Vittinghoff, E., Shapiro, J. A., & Ladabaum, U. (2020). Effectiveness and cost of organized outreach for colorectal cancer screening: A randomized, controlled trial. Journal of the National Cancer Institute, 112(3), 305–313. https://doi.org/10.1093/jnci/djz110
Pignone, M., Lanier, B., Kluz, N., Valencia, V., Chang, P., & Olmstead, T. (2021). Effectiveness and cost-effectiveness of mailed FIT in a safety net clinic population. Journal of General Internal Medicine, 36(11), 3441–3447. https://doi.org/10.1007/s11606-021-06691-y
Gupta, S., Coronado, G. D., Argenbright, K., Brenner, A. T., Castañeda, S. F., Dominitz, J. A., Green, B., Issaka, R. B., Levin, T. R., Reuland, D. S., Richardson, L. C., Robertson, D. J., Singal, A. G., & Pignone, M. (2020). Mailed fecal immunochemical test outreach for colorectal cancer screening: Summary of a centers for disease control and prevention-sponsored summit. CA: A Cancer Journal for Clinicians, 70(4), 283–298. https://doi.org/10.3322/caac.21615
Shah, S. K., & McElfish, P. A. (2022). Cancer screening recommendations during the COVID-19 pandemic: Scoping review. JMIR Cancer, 8(1), e34392–e34392. https://doi.org/10.2196/34392
Zapka, J., Klabunde, C. N., Taplin, S., Yuan, G., Ransohoff, D., & Kobrin, S. (2012). Screening colonoscopy in the US: Attitudes and practices of primary care physicians. Journal of General Internal Medicine, 27(9), 1150–1158. https://doi.org/10.1007/s11606-012-2051-3
Guerra, C. E., Schwartz, J. S., Armstrong, K., Brown, J. S., Halbert, C. H., & Shea, J. A. (2007). Barriers of and facilitators to physician recommendation of colorectal cancer screening. Journal of General Internal Medicine, 22(12), 1681–1688. https://doi.org/10.1007/s11606-007-0396-9
Subramanian, S., Bobashev, G., & Morris, R. J. (2010). When budgets are tight, there are better options than colonoscopies for colorectal cancer screening. Health Affairs (Millwood), 29(9), 1734–1740. https://doi.org/10.1377/hlthaff.2008.0898
Coronado, G. D., Petrik, A. F., Vollmer, W. M., Taplin, S. H., Keast, E. M., Fields, S., & Green, B. B. (2018). Effectiveness of a mailed colorectal cancer screening outreach program in community health clinics: The STOP CRC cluster randomized clinical trial. JAMA Internal Medicine, 178(9), 1174–1181. https://doi.org/10.1001/jamainternmed.2018.3629
Issaka, R. B., Akinsoto, N. O., Strait, E., Chaudhari, V., Flum, D. R., & Inadomi, J. M. (2020). Effectiveness of a mailed fecal immunochemical test outreach: A medicare advantage pilot study. Therapeutic Advances in Gastroenterology, 13, 1756284820945388. https://doi.org/10.1177/1756284820945388
Zhong, G. C., Sun, W. P., Wan, L., Hu, J. J., & Hao, F. B. (2020). Efficacy and cost-effectiveness of fecal immunochemical test versus colonoscopy in colorectal cancer screening: A systematic review and meta-analysis. Gastrointestinal Endoscopy, 91(3), 684-697 e615. https://doi.org/10.1016/j.gie.2019.11.035
Wheeler, S. B., O’Leary, M. C., Rhode, J., Yang, J. Y., Drechsel, R., Plescia, M., Reuland, D. S., & Brenner, A. T. (2020). Comparative cost-effectiveness of mailed fecal immunochemical testing (FIT)-based interventions for increasing colorectal cancer screening in the Medicaid population. Cancer, 126(18), 4197–4208. https://doi.org/10.1002/cncr.32992
Gorin, S. N. S., Jimbo, M., Heizelman, R., Harmes, K. M., & Harper, D. M. (2021). The future of cancer screening after COVID-19 may be at home. Cancer, 127(4), 498–503. https://doi.org/10.1002/cncr.33274
Inadomi, J. M., Vijan, S., Janz, N. K., Fagerlin, A., Thomas, J. P., Lin, Y. V., Muñoz, R., Lau, C., Somsouk, M., El-Nachef, N., & Hayward, R. A. (2012). Adherence to colorectal cancer screening: A randomized clinical trial of competing strategies. Archives of Internal Medicine, 172(7), 575–582. https://doi.org/10.1001/archinternmed.2012.332
Wang, A., Rachocki, C., Shapiro, J. A., Issaka, R. B., & Somsouk, M. (2019). Low literacy level instructions and reminder calls improve patient handling of fecal immunochemical test samples. Clinical Gastroenterology and Hepatology, 17(9), 1822–1828. https://doi.org/10.1016/j.cgh.2018.11.050
National Association of Chronic Disease Directors, Kaiser Permanente Center for Health Research, Centers for Disease Control and Prevention (CDC). (2021). Mailed FIT implementation guide. Retrieved from https://chronicdisease.org/wp-content/uploads/2021/11/60851-Mailed-FIT-Guide-V28.pdf
Pluye, P., Potvin, L., & Denis, J. (2004). Making public health programs last: Conceptualizing sustainability. Evaluation and Program Planning, 27, 121–133. https://doi.org/10.1016/j.evalprogplan.2004.01.001
Buchanan, D., Fitzgerald, F., Ketley, D., Gollop, R., Jones, J., Saint Lamont, S., Neath, A., & Whitby, E. (2005). No going back: A review of the literature on sustaining organizational change. International Journal of Management Reviews, 7(3), 189–205. https://doi.org/10.1111/j.1468-2370.2005.00111.x
Miech, E. J., Rattray, N. A., Flanagan, M. E., Damschroder, L., Schmid, A. A., & Damush, T. M. (2018). Inside help: An integrative review of champions in healthcare-related implementation. SAGE Open Medicine, 6, 2050312118773261. https://doi.org/10.1177/2050312118773261
Schlueter, D., DeGroff, A., Soloe, C., Arena, L., Melillo, S., Tangka, F., & Hoover, S. (2022). Factors that support sustainability of health systems change to increase colorectal cancer screening in primary care clinics: A longitudinal qualitative study. Health Promotion Practice. https://doi.org/10.1177/15248399221091999
Baldwin, L. M., Schneider, J. L., Schwartz, M., Rivelli, J. S., Green, B. B., Petrik, A. F., & Coronado, G. D. (2020). First-year implementation of mailed FIT colorectal cancer screening programs in two medicaid/medicare health insurance plans: Qualitative learnings from health plan quality improvement staff and leaders. BMC Health Services Research, 20(1), 132. https://doi.org/10.1186/s12913-019-4868-5
Martin, J., Halm, E. A., Tiro, J. A., Merchant, Z., Balasubramanian, B. A., McCallister, K., Sanders, J. M., Ahn, C., Bishop, W. P., & Singal, A. G. (2017). Reasons for lack of diagnostic colonoscopy after positive result on fecal immunochemical test in a safety-net health system. The American Journal of Medicine, 130(1), 93.e91–93.e97. https://doi.org/10.1016/j.amjmed.2016.07.028
Acknowledgements
We would like to express appreciation to all the individuals who participated in the interviews for this project, including staff from the Alaska Native Tribal Health Consortium, the University of Arkansas for Medical Sciences, the Georgia Center for Oncology Research and Education, the University of Chicago Medical Center, the Iowa Department of Public Health, Curators of the University of Missouri, the New York State Department of Health, the Rhode Island Department of Health, the University of Texas Health Science Center at Houston, HCC Network, Albany Area Primary Health Care, Southeast Alaska Regional Health Consortium, 1st Choice Healthcare, Comprehensive Community Health Centers, Sun River Health, Friend Family Health Center, and All Care Health Center.
Funding
Funding support was provided by the Centers for Disease Control and Prevention (CDC) (Contract No. GS00F354CA Task 7) to RTI International. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the CDC.
Author information
Authors and Affiliations
Contributions
All authors contributed to the development of the research and evaluation questions. CS and LA conducted and developed the interview guide, with feedback from SS, SH, AD, FT, DS and SM. CS, LA, and SH conducted the interviews. LA, CS, and KF-B conducted the qualitative coding and performed the thematic analysis. LA drafted and revised the manuscript based on comments from the coauthors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Ethical Approval
The RTI Institutional Review Board determined that this study did not constitute human subjects research and therefore did not require review.
Consent to Participate
Verbal consent to participate was obtained from all individuals who participated in interviews.
Consent for Publication
Consent for a de-identified summary of findings was obtained from individuals who participated in the interviews.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arena, L., Soloe, C., Schlueter, D. et al. Modifications in Primary Care Clinics to Continue Colorectal Cancer Screening Promotion During the COVID-19 Pandemic. J Community Health 48, 113–126 (2023). https://doi.org/10.1007/s10900-022-01154-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10900-022-01154-9