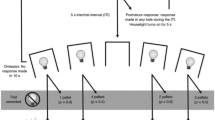
Abstract
Similar to drugs of abuse, random-ratio reward schedules are highly motivating and, in humans, are thought to foster gambling addiction. Animal gambling models, however, have not yet demonstrated the compulsivity so characteristic of drug addiction. Three criteria have been used to evaluate addiction-like behavior in drug models: (1) response inhibition when reward is not available, (2) persistence under a progressive ratio schedule, in which the response-to-reward ratio is stretched, and (3) persistence in spite of punishment. We tested whether prolonged exposure (6 weeks) to a gambling-like reward schedule would induce addiction-like symptoms in rats. In two studies, separate groups were trained to respond to either random- or fixed-ratio schedules for food reward. We found that rats trained on random-ratio schedules showed higher response rates and dramatically shorter pauses after rewards. Tests of addiction-like behavior, however, were largely negative. Response rates were not different during cued no-reward periods nor when reward was coupled with punishment. We also found no group differences when food was devalued nor in reinstatement of reward-seeking after a 1-week delay. The sole exception to this pattern was that rats in the second experiment showed greater persistence on a progressive ratio test. After experiment two, subjects were also orally administered pramipexole, which caused increased perseveration during progressive ratio testing, especially in the random ratio group. While, it is possible that longer training or more appetitive rewards might have led to addiction-like behavior, our results, on the surface, suggest that random-ratio schedules are motivating but not addictive.






Similar content being viewed by others
References
Alexander, B. K., Coambs, R. B., & Hadaway, P. F. (1978). The effect of housing and gender on morphine self-administration in rats. Psychopharmacology (Berlin), 58(2), 175–179.
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Arlington, VA: American Psychiatric Publishing.
Avena, N. M., Rada, P., & Hoebel, B. G. (2008). Evidence for sugar addiction: Behavioral and neurochemical effects of intermittent, excessive sugar intake. Neuroscience and Biobehavioral Reviews, 32(1), 20–39. https://doi.org/10.1016/j.neubiorev.2007.04.019.
Balleine, B. W., & O’Doherty, J. P. (2010). Human and rodent homologies in action control: Corticostriatal determinants of goal-directed and habitual action. Neuropsychopharmacology, 35(1), 48–69. https://doi.org/10.1038/npp.2009.131.
Baron, A., Mikorski, J., & Schlund, M. (1992). Reinforcement magnitude and pausing on progressive-ratio schedules. Journal of the Experimental Analysis of Behavior, 58(2), 377–388.
Bechara, A., Damasio, H., Damasio, A. R., & Lee, G. P. (1999). Different contributions of the human amygdala and ventromedial prefrontal cortex to decision-making. The Journal of Neuroscience, 19(13), 5473–5481.
Belin, D., Balado, E., Piazza, P. V., & Deroche-Gamonet, V. (2009). Pattern of intake and drug craving predict the development of cocaine addiction-like behavior in rats. Biological Psychiatry, 65(10), 863–868. https://doi.org/10.1016/j.biopsych.2008.05.031.
Belin, D., Mar, A. C., Dalley, J. W., Robbins, T. W., & Everitt, B. J. (2008). High impulsivity predicts the switch to compulsive cocaine-taking. Science, 320(5881), 1352–1355. https://doi.org/10.1126/science.1158136.
Belin-Rauscent, A., & Belin, D. (2012). Animal models of drug addiction. In D. Belin (Ed.), Addictions—from pathophysiology to treatment. Poitiers: Université de Poitiers.
Blaszczynski, A., & Nower, L. (2002). A pathways model of problem and pathological gambling. Addiction, 97, 487–499.
Boren, M. C. P. (1973). Fixed-ratio and variable-ratio schedules of brief stimuli in second-order schedules of matching to sample. Journal of the Experimental Analysis of Behavior, 20(2), 219–233.
Bostwick, J. M., Hecksel, K. A., Stevens, S. R., Bower, J. H., & Ahlskog, J. E. (2009). Frequency of new-onset pathologic compulsive gambling or hypersexuality after drug treatment of idiopathic Parkinson disease. Mayo Clinic Proceedings, 84(4), 310–316.
Bradshaw, C. M., & Killeen, P. R. (2012). A theory of behaviour on progressive ratio schedules, with applications in behavioural pharmacology. Psychopharmacology (Berlin), 222(4), 549–564. https://doi.org/10.1007/s00213-012-2771-4.
Caine, S. B., & Koob, G. F. (1995). Pretreatment with the dopamine agonist 7-OH-DPAT shifts the cocaine self-administration dose-effect function to the left under different schedules in the rat. Behavioural Pharmacology, 6(4), 333–347.
Chambers, R. A., Taylor, J. R., & Potenza, M. N. (2003). Developmental neurocircuitry of motivation in adolescence: A critical period of addiction vulnerability. The American Journal of Psychiatry, 160, 1041–1052.
Clark, L. (2010). Decision-making during gambling: An integration of cognitive and psychobiological approaches. Philosophical Transactions of the Royal Society of London. Series B, Biological sciences, 365(1538), 319–330. https://doi.org/10.1098/rstb.2009.0147.
Cocker, P. J., Dinelle, K., Kornelson, R., Sossi, V., & Winstanley, C. A. (2012). Irrational choice under uncertainty correlates with lower striatal D(2/3) receptor binding in rats. The Journal of Neuroscience, 32(44), 15450–15457. https://doi.org/10.1523/JNEUROSCI.0626-12.2012.
Crossman, E. K., Bonem, E. J., & Phelps, B. J. (1987). A comparison of response patterns on fixed-, variable-, and random-ratio schedules. Journal of the Experimental Analysis of Behavior, 48(3), 395–406.
Delfabbro, P. H., & Winefield, A. H. (1999). Poker-machine gambling: An analysis of within session characteristics. British Journal of Psychology, 90, 425–439.
Depoortere, R., Perrault, G., & Sanger, D. J. (1996). Behavioural effects in the rat of the putative dopamine D3 receptor agonist 7-OH-DPAT: Comparison with quinpirole and apomorphine. Psychopharmacology (Berlin), 124(3), 231–240.
Deroche-Gamonet, V., Belin, D., & Piazza, P. V. (2004). Evidence for addiction-like behavior in the rat. Science, 305(5686), 1014–1017. https://doi.org/10.1126/science.1099020.
Deroche-Gamonet, V., & Piazza, P. V. (2014). Psychobiology of cocaine addiction: Contribution of a multi-symptomatic animal model of loss of control. Neuropharmacology, 76 Pt B, 437–449. https://doi.org/10.1016/j.neuropharm.2013.07.014.
Diergaarde, L., Pattij, T., Nawijn, L., Schoffelmeer, A. N., & De Vries, T. J. (2009). Trait impulsivity predicts escalation of sucrose seeking and hypersensitivity to sucrose-associated stimuli. Behavioral Neuroscience, 123(4), 794–803. https://doi.org/10.1037/a0016504.
Dixon, M. J., MacLaren, V., Jarick, M., Fugelsang, J. A., & Harrigan, K. A. (2013). The frustrating effects of just missing the jackpot: Slot machine near-misses trigger large skin conductance responses, but no post-reinforcement pauses. Journal of Gambling Studies, 29(4), 661–674. https://doi.org/10.1007/s10899-012-9333-x.
Dixon, M. R., Marley, J., & Jacobs, E. A. (2003). Delay discounting by pathological gamblers. Journal of Applied Behavior Analysis, 36(4), 449–458. https://doi.org/10.1901/jaba.2003.36-449.
Dodd, M. L., Klos, K. J., Bower, J. H., Geda, Y. E., Josephs, K. A., & Ahlskog, J. E. (2005). Pathological gambling caused by drugs used to treat Parkinson disease. Archives of Neurology, 62(9), 1377–1381. https://doi.org/10.1001/archneur.62.9.noc50009.
Drapier, D., Drapier, S., Sauleau, P., Derkinderen, P., Damier, P., Allain, H., et al. (2006). Pathological gambling secondary to dopaminergic therapy in Parkinson’s disease. Psychiatry Research, 144(2–3), 241–244. https://doi.org/10.1016/j.psychres.2006.04.017.
Edwards, S., & Koob, G. F. (2013). Escalation of drug self-administration as a hallmark of persistent addiction liability. Behavioural Pharmacology, 24(5–6), 356–362. https://doi.org/10.1097/FBP.0b013e3283644d15.
Ersche, K. D., Turton, A. J., Pradhan, S., Bullmore, E. T., & Robbins, T. W. (2010). Drug addiction endophenotypes: Impulsive versus sensation-seeking personality traits. Biological Psychiatry, 68(8), 770–773. https://doi.org/10.1016/j.biopsych.2010.06.015.
Everitt, B. J. (2014). Neural and psychological mechanisms underlying compulsive drug seeking habits and drug memories—indications for novel treatments of addiction. European Journal of Neuroscience, 40(1), 2163–2182.
Everitt, B. J., Belin, D., Economidou, D., Pelloux, Y., Dalley, J. W., & Robbins, T. W. (2008). Review. Neural mechanisms underlying the vulnerability to develop compulsive drug-seeking habits and addiction. Philosophical Transactions of the Royal Society of London. Series B, Biological sciences, 363(1507), 3125–3135. https://doi.org/10.1098/rstb.2008.0089.
Everitt, B. J., & Robbins, T. W. (2016). Drug addiction: Updating actions to habits to compulsions ten years on. Annual Review of Psychology, 67(1), 23–50. https://doi.org/10.1146/annurev-psych-122414-033457.
Fantino, E. (1967). Preference for mixed- versus fixed-ratio schedules. Journal of the Experimental Analysis of Behavior, 10(1), 35–43.
Farmer, J., & Schoenfeld, W. N. (1967). Response rates under varying probability of reinforcement. Psychonomic Science, 7(5), 173–174.
Felton, M., & Lyon, D. O. (1966). The post-reinforcement pause. Journal of the Experimental Analysis of Behavior, 9(2), 131–134.
Fernández, F. M., & González, T. M. (2009). Pathological gambling and hypersexuality due to dopaminergic treatment in Parkinson’s disease. Actas Españolas de Psiquiatría, 37(2), 118–122.
Fiorillo, C. D., Tobler, P. N., & Schultz, W. (2003). Discrete coding of reward probability and uncertainty by dopamine neurons. Science, 299(5614), 1898–1902.
Giladi, N., Weitzman, N., Schreiber, S., Shabtai, H., & Peretz, C. (2007). New onset heightened interest or drive for gambling, shopping, eating or sexual activity in patients with Parkinson’s disease: The role of dopamine agonist treatment and age at motor symptoms onset. Journal of Psychopharmacology, 21(5), 501–506. https://doi.org/10.1177/0269881106073109.
Goudriaan, A. E., Oosterlaan, J., de Beurs, E., & van den Brink, W. (2005). Decision making in pathological gambling: A comparison between pathological gamblers, alcohol dependents, persons with Tourette syndrome, and normal controls. Brain Research. Cognitive Brain Research, 23(1), 137–151. https://doi.org/10.1016/j.cogbrainres.2005.01.017.
Hoebel, B. G., Avena, N. M., Bocarsly, M. E., & Rada, P. (2009). Natural addiction: A behavioral and circuit model based on sugar addiction in rats. Journal of Addiction Medicine, 3(1), 33–41. https://doi.org/10.1097/ADM.0b013e31819aa621.
Holden, C. (2001). ‘Behavioral’ addictions: Do they exist? Science, 294(5544), 980–982. https://doi.org/10.1126/science.294.5544.980.
Imamura, A., Uitti, R. J., & Wszolek, Z. K. (2006). Dopamine agonist therapy for Parkinson disease and pathological gambling. Parkinsonism & Related Disorders, 12(8), 506–508. https://doi.org/10.1016/j.parkreldis.2006.02.004.
Izzo, E., Orsini, C., Koob, G. F., & Pulvirenti, L. (2001). A dopamine partial agonist and antagonist block amphetamine self-administration in a progressive ratio schedule. Pharmacology, Biochemistry and Behavior, 68, 701–708.
Johnson, P. S., Madden, G. J., Brewer, A. T., Pinkston, J. W., & Fowler, S. C. (2011). Effects of acute pramipexole on preference for gambling-like schedules of reinforcement in rats. Psychopharmacology (Berlin), 213(1), 11–18. https://doi.org/10.1007/s00213-010-2006-5.
Johnson, P. S., Madden, G. J., & Stein, J. S. (2012). Effects of acute pramipexole on male rats’ preference for gambling-like rewards II. Experimental and Clinical Psychopharmacology, 20(3), 167–172. https://doi.org/10.1037/a0027117.
Killcross, S., & Coutureau, E. (2003). Coordination of actions and habits in the medial prefrontal cortex of rats. Cerebral Cortex, 13(4), 400–408.
Killeen, P. R., & Fetterman, J. G. (1993). The behavioral theory of timing: Transition analyses. Journal of the Experimental Analysis of Behavior, 59(2), 411–422.
Killeen, P. R., Posadas-Sanchez, D., Johansen, E. B., & Thrailkill, E. A. (2009). Progressive ratio schedules of reinforcement. Journal of Experimental Psychology: Animal Behavior Processes, 35(1), 35–50. https://doi.org/10.1037/a0012497.
Lagorio, C. H., & Winger, G. (2014). Random-ratio schedules produce greater demand for i.v. drug administration than fixed-ratio schedules in rhesus monkeys. Psychopharmacology (Berlin), 231(15), 2981–2988. https://doi.org/10.1007/s00213-014-3477-6.
Lobo, D. S., Aleksandrova, L., Knight, J., Casey, D. M., el-Guebaly, N., Nobrega, J. N., et al. (2015). Addiction-related genes in gambling disorders: New insights from parallel human and pre-clinical models. Molecular Psychiatry, 20(8), 1002–1010. https://doi.org/10.1038/mp.2014.113.
Lowe, C. F., Harzem, P., & Bagshaw, M. (1978). Species differences in temporal control of behaviour II: Human performance. Journal of the Experimental Analysis of Behavior, 29(3), 351–361.
Madden, G. J., Ewan, E. E., & Lagorio, C. H. (2007). Toward an animal model of gambling: Delay discounting and the allure of unpredictable outcomes. Journal of Gambling Studies, 23(1), 63–83. https://doi.org/10.1007/s10899-006-9041-5.
Madden, G. J., Johnson, P. S., Brewer, A. T., Pinkston, J. W., & Fowler, S. C. (2010). Effects of pramipexole on impulsive choice in male wistar rats. Experimental and Clinical Psychopharmacology, 18(3), 267–276. https://doi.org/10.1037/a0019244.
Mazur, J. E. (1983). Steady-state performance on fixed-, mixed-, and random-ratio schedules. Journal of the Experimental Analysis of Behavior, 39(2), 293–307.
Pelloux, Y., Everitt, B. J., & Dickinson, A. (2007). Compulsive drug seeking by rats under punishment: Effects of drug taking history. Psychopharmacology (Berlin), 194, 127–137.
Piazza, P. V., Deminiere, J. M., Le Moal, M., & Simon, H. (1989). Factors that predict individual vulnerability to amphetamine self-administration. Science, 245(4925), 1511–1513.
Powell, R. W. (1968). The effect of small sequential changes in fixed-ratio size upon the post-reinforcement pause. Journal of the Experimental Analysis of Behavior, 11(5), 589–593.
Priddle-Higson, P. J., Lowe, C. F., & Harzem, P. (1976). Aftereffects of reinforcement on variable-ratio schedules. Journal of the Experimental Analysis of Behavior, 25(3), 347–354.
Ross, D., Sharp, C., Vuchinich, R. E., & Spurrett, D. (2008). Midbrain mutiny: The picoeconomics and neuroeconomics of disordered gambling: Economic theory and cognitive science. Cambridge, MA: MIT Press.
Schlinger, H. D., Derenne, A., & Baron, A. (2008). What 50 years of research tell us about pausing under ratio schedules of reinforcement. The Behavior Analyst, 31(1), 39–60.
Schultz, W. (2016). Dopamine reward prediction-error signalling: A two-component response. Nature Reviews Neuroscience, 17(3), 183–195. https://doi.org/10.1038/nrn.2015.26.
Seeman, P. (2015). Parkinson’s disease treatment may cause impulse-control disorder via dopamine D3 receptors. Synapse (New York, N. Y.), 69(4), 183–189. https://doi.org/10.1002/syn.21805.
Shaham, Y., Erb, S., & Stewart, J. (2000). Stress-induced relapse to heroin and cocaine seeking in rats: A review. Brain Research Reviews, 33(1), 13–33. https://doi.org/10.1016/s0165-0173(00)00024-2.
Shaham, Y., Shalev, U., Lu, L., De Wit, H., & Stewart, J. (2003). The reinstatement model of drug relapse: History, methodology and major findings. Psychopharmacology (Berlin), 168(1–2), 3–20. https://doi.org/10.1007/s00213-002-1224-x.
Sherman, J. A., & Thomas, J. R. (1968). Some factors controlling preference between fixed-ratio and variable-ratio schedules of reinforcement. Journal of the Experimental Analysis of Behavior, 11(6), 689–702.
Singer, B. F., Scott-Railton, J., & Vezina, P. (2012). Unpredictable saccharin reinforcement enhances locomotor responding to amphetamine. Behavioural Brain Research, 226(1), 340–344. https://doi.org/10.1016/j.bbr.2011.09.003.
Sinha, R. (2001). How does stress increase risk of drug abuse and relapse? Psychopharmacology (Berlin), 158(4), 343–359. https://doi.org/10.1007/s002130100917.
Skinner, B. F., & Ferster, C. B. (1957). Schedules of reinforcement. Engelwood Cliffs, NJ: Prentice-Hall.
Stafford, D., LeSage, M. G., & Glowa, J. R. (1998). Progressive-ratio schedules of drug delivery in the analysis of drug self-administration: A review. Psychopharmacology (Berlin), 139(3), 169–184.
Tremblay, M., Silveira, M. M., Kaur, S., Hosking, J. G., Adams, W. K., Baunez, C., et al. (2017). Chronic D2/3 agonist ropinirole treatment increases preference for uncertainty in rats regardless of baseline choice patterns. European Journal of Neuroscience, 45(1), 159–166. https://doi.org/10.1111/ejn.13332.
Pharmacia & Upjohn, I. (1997). Mirapex (pramipexole dihydrochloride) tablets drug approval package.
Vanderschuren, L. J., & Everitt, B. J. (2004). Drug seeking becomes compulsive after prolonged cocaine self-administration. Science, 305, 1017–1019.
Velázquez-Sánchez, C., Ferragud, A., Moore, C. F., Everitt, B. J., Sabino, V., & Cottone, P. (2014). High trait impulsivity predicts food addiction-like behavior in the rat. Neuropsychopharmacology, 39(10), 2463–2472. https://doi.org/10.1038/npp.2014.98.
Verdejo-Garcia, A., Lawrence, A. J., & Clark, L. (2008). Impulsivity as a vulnerability marker for substance-use disorders: Review of findings from high-risk research, problem gamblers and genetic association studies. Neuroscience and Biobehavioral Reviews, 32(4), 777–810. https://doi.org/10.1016/j.neubiorev.2007.11.003.
Voon, V., Potenza, M. N., & Thomsen, T. (2007). Medication-related impulse control and repetitive behaviors in Parkinson’s disease. Current Opinion in Neurology, 20(4), 484–492. https://doi.org/10.1097/WCO.0b013e32826fbc8f.
Waters, R. P., Moorman, D. E., Young, A. B., Feltenstein, M. W., & See, R. E. (2014). Assessment of a proposed “three-criteria” cocaine addiction model for use in reinstatement studies with rats. Psychopharmacology (Berlin), 231(16), 3197–3205. https://doi.org/10.1007/s00213-014-3497-2.
Webbe, F. M., & Malagodi, E. F. (1978). Second-order schedules of token reinforcement: Comparisons of performance under fixed-ratio and variable-ratio exchange schedules. Journal of the Experimental Analysis of Behavior, 30(2), 219–224.
Weintraub, D., & Claassen, D. O. (2017). Impulse control and related disorders in Parkinson’s disease. International Review of Neurobiology, 133, 679–717. https://doi.org/10.1016/bs.irn.2017.04.006.
Weintraub, D., Siderowf, A. D., Potenza, M. N., Goveas, J., Morales, K. H., Duda, J. E., et al. (2006). Association of dopamine agonist use with impulse control disorders in Parkinson disease. Archives of Neurology, 63(7), 969–973. https://doi.org/10.1001/archneur.63.7.969.
Williams, D. C., Saunders, K. J., & Perone, M. (2011). Extended pausing by humans on multiple fixed-ratio schedules with varied reinforcer magnitude and response requirements. Journal of the Experimental Analysis of Behavior, 95(2), 203–220. https://doi.org/10.1901/jeab.2011.95-203.
Winstanley, C. A., & Clark, L. (2016). Translational models of gambling-related decision making. Current Topics in Behavioral Neursciences, 28, 93–120. https://doi.org/10.1007/7854_2015_5014.
Winstanley, C. A., Cocker, P. J., & Rogers, R. D. (2011). Dopamine modulates reward expectancy during performance of a slot machine task in rats: Evidence for a ‘near-miss’ effect. Neuropsychopharmacology, 36(5), 913–925. https://doi.org/10.1038/npp.2010.230.
Zack, M., Featherstone, R. E., Mathewson, S., & Fletcher, P. J. (2014). Chronic exposure to a gambling-like schedule of reward predictive stimuli can promote sensitization to amphetamine in rats. Frontiers in Behavioral Neuroscience, 8, 36. https://doi.org/10.3389/fnbeh.2014.00036.
Zald, D. H., Boileau, I., El-Dearedy, W., Gunn, R., McGlone, F., Dichter, G. S., et al. (2004). Dopamine transmission in the human striatum during monetary reward tasks. The Journal of Neuroscience, 24(17), 4105–4112. https://doi.org/10.1523/JNEUROSCI.4643-03.2004.
Zand, R. (2008). Is dopamine agonist therapy associated with developing pathological gambling in Parkinson’s disease patients? European Neurology, 59, 183–186.
Zoratto, F., Romano, E., Pascale, E., Pucci, M., Falconi, A., Dell’Osso, B., et al. (2017). Down-regulation of serotonin and dopamine transporter genes in individual rats expressing a gambling-prone profile: A possible role for epigenetic mechanisms. Neuroscience, 340, 101–116. https://doi.org/10.1016/j.neuroscience.2016.10.041.
Acknowledgements
The authors wish to thank Kyler Fisher, Kate Godfrey, Sara-Lynn Pelegrin and Dr. Aaron Gruber for their contributions to this work including data collection and procedural advice.
Funding
This study was funded by the Alberta Gambling Research Institute (Grant No. #70) via grants awarded to both David R. Euston and Darren R. Christensen. The Alberta Gambling Research Institute had no involvement in preparation of this article, study design, data collection, analysis and interpretation of data, or the decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Catherine S. Laskowski declares that she has no conflict of interest. Danika L. Dorchak is an undergraduate student who during the course of these experiments, worked part-time as a croupier at a local casino. Kathleen M. Ward declares that she has no conflict of interest. Darren R. Christensen declares that he has no conflict of interest. David R. Euston declares that he has no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Laskowski, C.S., Dorchak, D.L., Ward, K.M. et al. Can Slot-Machine Reward Schedules Induce Gambling Addiction in Rats?. J Gambl Stud 35, 887–914 (2019). https://doi.org/10.1007/s10899-019-09852-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10899-019-09852-1