Abstract
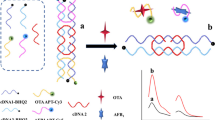
Tuning fork, consisting of two fork arms and a fork handle, has a stable and rigid structure. Inspired by this structure, a tuning fork-shaped DNA (TF-DNA) fluorescence aptasensor was constructed to detect ochratoxin A (OTA) and aflatoxin B1 (AFB1). A TF-DNA double-stranded structure capable of attaching both OTA aptamer labeled with the FAM fluorescent group (FAM-Apt) and AFB1 aptamer labeled with the ROX fluorescent group (ROX-Apt) was designed and linked to magnetic beads. This TF-DNA double-stranded structure can provide a stable platform for dual-target detection. In the presence of OTA and AFB1, FAM-Apt and ROX-Apt preferentially bound to them and detached from the TF-DNA double-stranded structure. Dual-signal fluorescent probes were collected from the supernatant by magnetic separation, and achieved fluorescence enhancement at 520 nm and 607 nm, respectively. The linear ranges are 0.05 ng/mL to 100 ng/mL for OTA and 0.1 ng/mL to 100 ng/mL for AFB1, and the detection limits are 0.015 ng/mL and 0.045 ng/mL, respectively. The developed sensor has the advantages of simple and fast preparation, good specificity and reproducibility, which is promising for the simultaneous determination of multiple hazardous substances in food.








Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ma Q, Lin Y, Yang Y, Yang W, Yun W, Zhang M, Yang L (2023) Target controlled alternative hybridization chain reaction for fluorescent detection of dual mycotoxins. Anal Chim Acta 1237:340595. https://doi.org/10.1016/j.aca.2022.340595
Suo Z, Liang X, Jin H, He B, Wei M (2021) A signal-enhancement fluorescent aptasensor based on the stable dual cross DNA nanostructure for simultaneous detection of OTA and AFB1. Anal Bioanal Chem 413(30):7587–7595. https://doi.org/10.1007/s00216-021-03723-8
Sanzani S, Reverberi M, Fanelli C, Ippolito A (2015) Detection of ochratoxin a using molecular beacons and real-time PCR thermal cycler. Toxins (Basel) 7(3):812–820. https://doi.org/10.3390/toxins7030812
Taghdisi S, Danesh N, Ramezani M, Abnous K (2018) A new amplified fluorescent aptasensor based on hairpin structure of G-quadruplex oligonucleotide-Aptamer chimera and silica nanoparticles for sensitive detection of aflatoxin B1 in the grape juice. Food Chem 268:342–346. https://doi.org/10.1016/j.foodchem.2018.06.101
Tittlemier S, Cramer B, Dall’Asta C, Iha M, Lattanzio V, Malone R, Maragos C, Solfrizzo M, Stranska-Zachariasova M, Stroka J (2019) Developments in mycotoxin analysis: an update for 2017–2018. World Mycotoxin J 12(1):3–29. https://doi.org/10.3920/WMJ2018.2398
Zhang X, Zhang L, Zhou T, Zhou Y (2022) Fungal flora and mycotoxin contamination in tea: Current status, detection methods and dietary risk assessment-A comprehensive review. Trends Food Sci Technol 127:207–220. https://doi.org/10.1016/j.tifs.2022.05.019
Sedova I, Kiseleva M, Tutelyan V (2018) Mycotoxins in tea: occurrence, methods of determination and risk evaluation. Toxins (Basel) 10(11):444. https://doi.org/10.3390/toxins10110444
Zhai S, Zhu Y, Feng P, Li M, Wang W, Yang L, Yang Y (2021) Ochratoxin A: its impact on poultry gut health and microbiota, an overview. Poult Sci 100(5):101037. https://doi.org/10.1016/j.psj.2021.101037
Martinez L, He L (2021) Detection of mycotoxins in food using surface-enhanced Raman spectroscopy: a review. Acs Appl Bio Mater 4(1):295–310. https://doi.org/10.1021/acsabm.0c01349
Li J, Cai T, Li W, Li W, Song L, Li Q, Lv G, Sun J, Jiao S, Wang S, Jin Y, Zheng T (2021) Highly sensitive simultaneous detection of multiple mycotoxins using a protein microarray on a tio2-modified porous silicon surface. J Agric Food Chem 69(1):528–536. https://doi.org/10.1021/acs.jafc.0c06859
Qing H, Huang S, Zhan K, Zhao L, Zhang J, Ji C, Ma Q (2022) Combined toxicity evaluation of ochratoxin A and aflatoxin B1 on kidney and liver injury, immune inflammation, and gut microbiota alteration through pair-feeding pullet model. Front Immunol 13:920147. https://doi.org/10.3389/fimmu.2022.920147
Zhao L, Liang X, Liu Y, Wei M, Jin H (2023) A novel fluorescent aptasensor based on dual-labeled DNA nanostructure for simultaneous detection of Ochratoxin A and Aflatoxin B1. J Fluoresc 33(4):1641–1650. https://doi.org/10.1007/s10895-022-03071-5
Qian J, Ren C, Wang C, Chen W, Lu X, Li H, Liu Q, Hao N, Li H, Wang K (2018) Magnetically controlled fluorescence aptasensor for simultaneous determination of ochratoxin A and aflatoxin B1. Anal Chim Acta 1019:119–127. https://doi.org/10.1016/j.aca.2018.02.063
Sun J, Li W, Zhu X, Jiao S, Chang Y, Wang S, Dai S, Xu R, Dou M, Li Q, Li J (2021) A novel multiplex mycotoxin surface-enhanced Raman spectroscopy immunoassay using functional gold nanotags on a silica photonic crystal microsphere biochip. J Agric Food Chem 69(38):11494–11501. https://doi.org/10.1021/acs.jafc.1c03469
Joo M, Baek S, Cheon S, Chun H, Choi S, Park T (2017) Development of aflatoxin B1 aptasensor based on wide-range fluorescence detection using graphene oxide quencher. Colloids Surf B Biointerfaces 154:27–32. https://doi.org/10.1016/j.colsurfb.2017.03.010
Qiao M, Liu M, Wan Z, Suo Z, Liu Y, Wei M (2023) A novel fluorescent aptasensor based on 3D porous nitrogen-sulfur co-doped carbon mesh and hybridization chain reaction for sensitive detection of ochratoxin A. Mikrochim Acta 190(8):313. https://doi.org/10.1007/s00604-023-05897-7
Liu X, Xu N, Gai P, Li F (2018) Triplex dna formation-mediated strand displacement reaction for highly sensitive fluorescent detection of melamine. Talanta 185:352–358. https://doi.org/10.1016/j.talanta.2018.03.094
Wang C, Zhang W, Qian J, Wang L, Ren Y, Wang Y, Xu M, Huang X (2021) A FRET aptasensor for sensitive detection of aflatoxin B1 based on a novel donor–acceptor pair between ZnS quantum dots and Ag nanocubes. Anal Methods 13(4):462–468. https://doi.org/10.1039/d0ay02017f
Li F, Zhang H, Wang Z, Newbigging A, Reid M, Li X, Le X (2015) Aptamers facilitating amplified detection of biomolecules. Anal Chem 87(1):274–292. https://doi.org/10.1021/ac5037236
Jia Y, Zhou G, Wang X, Zhang Y, Li Z, Liu P, Yu B, Zhang J (2020) A metal-organic framework/aptamer system as a fluorescent biosensor for determination of aflatoxin B1 in food samples. Talanta 219:121342. https://doi.org/10.1016/j.talanta.2020.121342
Seeman NC (2003) DNA in a material world. Nature 421(6921):427–431. https://doi.org/10.1038/nature01406
Madsen M, Gothelf K (2019) Chemistries for DNA nanotechnology. Chem Rev 119(10):6384–6458. https://doi.org/10.1021/acs.chemrev.8b00570
Shang Z, Ma P, Khan I, Zhang Y, Wang Z (2023) A DNA tweezers fluorescence aptasensor based on split aptamer-assisted magnetic nanoparticles for the detection of enrofloxacin in food. Food Control 145:109437. https://doi.org/10.1016/j.foodcont.2022.109437
Wang D, Wang J, Cui Y, Wang Y, Tang A, Kong D (2019) Nanolantern-based DNA probe and signal amplifier for tumor-related biomarker detection in living cells. Anal Chem 91(20):13165–13173. https://doi.org/10.1021/acs.analchem.9b03453
Hughes R, Ellington A (2017) Synthetic DNA synthesis and assembly: putting the synthetic in synthetic biology. Cold Spring Harb Perspect Biol 9(1):a023812. https://doi.org/10.1101/cshperspect.a023812
Xavier P, Chandrasekaran A (2018) DNA-based construction at the nanoscale: emerging trends and applications. Nanotechnology 29(6):062001. https://doi.org/10.1088/1361-6528/aaa120
Shiu S, Whitehouse W, Tanner J (2022) Designing aptamer-enabled DNA polyhedra using paper origami. Trends Analyt Chem 157:116723. https://doi.org/10.1016/j.trac.2022.116723
Zhang Y, Zhao J, Yang G, He Y, Chen S, Yuan R (2021) Ultrasensitive detection of amyloid β oligomers based on the “DD–A” FRET binary probes and quadrivalent cruciform DNA nanostructure-mediated cascaded amplifier. Acs Appl Mater Interfaces 13(27):32013–32021. https://doi.org/10.1021/acsami.1c07598
Chen R, Mao Z, Lu R, Wang Z, Hou Y, Zhu W, Li S, Ren S, Han D, Liang J, Gao Z (2022) Simple and programmed three-dimensional DNA tweezer for simultaneous one-step detection of ochratoxin A and zearalenone. Spectrochim Acta a Mol Biomol Spectrosc 272:120991. https://doi.org/10.1016/j.saa.2022.120991
Hong C, Zhang X, Ye S, Yang H, Huang Z, Yang D, Cai R, Tan W (2021) Aptamer-pendant DNA tetrahedron nanostructure probe for ultrasensitive detection of tetracycline by coupling target-triggered rolling circle amplification. Acs Appl Mater Interfaces 13(17):19695–19700. https://doi.org/10.1021/acsami.1c02612
Zeng R, Wang W, Cai G, Huang Z, Tao J, Tang D, Zhu C (2020) Single-atom platinum nanocatalyst-improved catalytic efficiency with enzyme-DNA supermolecular architectures. Nano Energy 74:104931. https://doi.org/10.1016/j.nanoen.2020.104931
Li L, Lv W, Wang Y, Li Y, Li C, Huang C (2021) DNA logic nanodevices for real-time monitoring of ATP in lysosomes. Anal Chem 93(46):15331–15339. https://doi.org/10.1021/acs.analchem.1c02829
Lian F, Wang D, Yao S, Ge L, Wang Y, Zhao Y, Zhao J, Song X, Zhao C, Li J, Liu Y, Jin M, Xu K (2021) A detection method of Escherichia coli O157: H7 based on immunomagnetic separation and aptamers-gold nanoparticle probe quenching Rhodamine B’s fluorescence: Escherichia coli O157: H7 detection method based on IMS and Apt-AuNPs probe quenching Rho B’s fluorescence. Food Sci Biotechnol 30(8):1129–1138. https://doi.org/10.1007/s10068-021-00947-3
Ding Y, Shang H, Wang X, Chen L (2020) A SERS-based competitive immunoassay for highly sensitive and specific detection of ochratoxin A. Analyst 145(18):6079–6084. https://doi.org/10.1039/d0an01220c
Zhu C, Liu D, Li Y, Ma S, Wang M, You T (2021) Hairpin DNA assisted dual-ratiometric electrochemical aptasensor with high reliability and anti-interference ability for simultaneous detection of aflatoxin B1 and ochratoxin A. Biosens Bioelectron 174:112654. https://doi.org/10.1016/j.bios.2020.112654
Xiao M, Bai X, Liu Y, Yang L, Liao X (2018) Simultaneous determination of trace Aflatoxin B1 and Ochratoxin A by aptamer-based microchip capillary electrophoresis in food samples. J Chromatogr A 1569:222–228. https://doi.org/10.1016/j.chroma.2018.07.051
Hao N, Lu J, Zhou Z, Hua R, Wang K (2018) A pH-resolved colorimetric biosensor for simultaneous multiple target detection. Acs Sens 3(10):2159–2165. https://doi.org/10.1021/acssensors.8b00717
Frenich A, Vidal J, Romero-González R, Aguilera-Luiz M (2009) Simple and high-throughput method for the multimycotoxin analysis in cereals and related foods by ultra-high performance liquid chromatography/tandem mass spectrometry. Food Chem 117(4):705–712. https://doi.org/10.1016/j.foodchem.2009.04.045
García-Moraleja A, Font G, Mañes J, Ferrer E (2015) Development of a new method for the simultaneous determination of 21 mycotoxins in coffee beverages by liquid chromatography tandem mass spectrometry. Food Res Int 72:247–255. https://doi.org/10.1016/j.foodres.2015.02.030
Giray B, Atasayar S, Sahin G (2009) Determinatıon of ochratoxin A and total aflatoxin levels in corn samples from Turkey by enzyme-linked immunosorbent assay. Mycotoxin Res 25(2):113–116. https://doi.org/10.1007/s12550-009-0016-0
Liu R, Li W, Cai T, Deng Y, Ding Z, Liu Y, Zhu X, Wang X, Liu J, Liang B, Zheng T, Li J (2018) TiO2 nanolayer-enhanced fluorescence for simultaneous multiplex mycotoxin detection by aptamer microarrays on a porous silicon surface. Acs Appl Mater Interfaces 10(17):14447–14453. https://doi.org/10.1021/acsami.8b01431
Wang X, Gao X, He J, Hu X, Li Y, Li X, Fan L, Yu H (2019) Systematic truncating of aptamers to create high-performance graphene oxide (GO)-based aptasensors for the multiplex detection of mycotoxins. Analyst 144(12):3826–3835. https://doi.org/10.1039/c9an00624a
Qian J, Cui H, Lu X, Wang C, An K, Hao N, Wang K (2020) Bi-color FRET from two nano-donors to a single nano-acceptor: a universal aptasensing platform for simultaneous determination of dual targets. Chem Eng J 401:126017. https://doi.org/10.1016/j.cej.2020.126017
Ma Q, Nie D, Sun X, Xu Y, He J, Yang L, Yang L (2022) A versatile Y shaped DNA nanostructure for simple, rapid and one-step detection of mycotoxins. Spectrochim Acta a Mol Biomol Spectrosc 281:121634. https://doi.org/10.1016/j.saa.2022.121634
Funding
This study was funded by Special Project for Collaborative Innovation of Zhengzhou (21ZZXTCX15), the Innovative Funds Plan of Henan University of Technology (2021ZKCJ13), National Key Research and Development Program of China (2023YFF1104600), and Joint Research Fund for science and technology R&D Projects of Henan Province (225200810066).
Author information
Authors and Affiliations
Contributions
Mengyao Wang: Experimentation, Writing-original draft, Formal analysis, Investigation. Zeping Lv: Conceptualization, Investigation, Writing-review & editing. Yong Liu: Supervision, Data analysis. Min Wei: Conceptualization, Supervision, Funding acquisition, Resources, Writing-review & editing.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publish
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Lv, Z., Liu, Y. et al. Simultaneous Detection of Ochratoxin A and Aflatoxin B1 Based on Stable Tuning Fork-shaped DNA Fluorescent Aptasensor. J Fluoresc (2024). https://doi.org/10.1007/s10895-024-03668-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10895-024-03668-y