Abstract
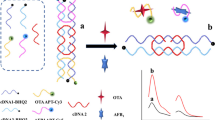
Based on DNA strand replacement reaction and aptamer-specific recognition, a simple dual-labeled DNA nanostructure is designed for the simultaneous detection of Ochratoxin A (OTA) and aflatoxin B1 (AFB1). C1 is labeled with Cy3 and Cy5, while C2 and C3 are labeled with BHQ2. The fluorescence intensity of DNA nanostructure composed of C1, C2 and C3 is weak because of fluorescence resonance energy transfer. When OTA Aptamer (OTA-Apt) and AFB1 Aptamer (AFB1-Apt) are added to the homogeneous system at the same time, C1 can be replaced with the help of toehold strand displacement, resulting in fluorescence enhancement. In the presence of both OTA and AFB1, the toehold strand displacement reaction is inhibited due to preferential binding between the target and their corresponding aptamers. The limit of detection of OTA was 0.007 ng/mL and that of AFB1 was 0.03 ng/mL. The recoveries of OTA and AFB1 were 96%-101% and 97%-101% in the corn sample, and 99%-101% and 92%-106% in the wine sample. Compared with other sensors, the preparation of this aptasensor needs simpler experimental steps and a shorter total-preparing time, confirming the convenient, rapid, and time-saving operation process.







Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Sun S, Zhao R, Xie Y, Liu Y (2019) Photocatalytic degradation of aflatoxin B1 by activated carbon supported TiO2 catalyst. Food Control 100:183–188. https://doi.org/10.1016/j.foodcont.2019.01.014
Tittlemier SA, Cramer B, Dall’Asta C, Iha MH, Lattanzio VMT, Malone RJ, Maragos C, Solfrizzo M, Stranska-Zachariasova M, Stroka J (2019) Developments in mycotoxin analysis: an update for 2017–2018. World Mycotoxin J 12(1):3–29. https://doi.org/10.3920/wmj2018.2398
Kolpin DW, Schenzel J, Meyer MT, Phillips PJ, Hubbard LE, Scott T-M, Bucheli TD (2014) Mycotoxins: Diffuse and point source contributions of natural contaminants of emerging concern to streams. Sci Total Environ 470:669–676. https://doi.org/10.1016/j.scitotenv.2013.09.062
Van Pamel E, Verbeken A, Vlaemynck G, De Boever J, Daeseleire E (2011) Ultrahigh-Performance Liquid Chromatographic-Tandem Mass Spectrometric Multimycotoxin Method for Quantitating 26 Mycotoxins in Maize Silage. J Agric Food Chem 59(18):9747–9755. https://doi.org/10.1021/jf202614h
Sun S, Zhao R, Xie Y, Liu Y (2021) Reduction of aflatoxin B1 by magnetic graphene oxide/TiO2 nanocomposite and its effect on quality of corn oil. Food Chem. https://doi.org/10.1016/j.foodchem.2020.128521
Liu C, Cao Y, Zhao T, Wang X, Fang G, Wang S (2021) A Novel Multi-purpose MIP for SPE-HPLC and QCM Detection of Carbaryl Residues in Foods. Food Anal Methods 14(2):331–343. https://doi.org/10.1007/s12161-020-01875-4
Xue X, Selvaraj JN, Zhao L, Dong H, Liu F, Liu Y, Li Y (2014) Simultaneous Determination of Aflatoxins and Ochratoxin A in Bee Pollen by Low-Temperature Fat Precipitation and Immunoaffinity Column Cleanup Coupled with LC-MS/MS. Food Anal Methods 7(3):690–696. https://doi.org/10.1007/s12161-013-9723-4
Khoshbin Z, Abnous K, Taghdisi SM, Verdian A, Sameiyan E, Ramezani M, Alibolandi M (2022) An ultra-sensitive dual-responsive aptasensor with combination of liquid crystal and intercalating dye molecules: A food toxin case study. Food Chem. https://doi.org/10.1016/j.foodchem.2022.132265
Khoshbin Z, Abnous K, Taghdisi SM, Verdian A (2021) Liquid crystal-based biosensors as lab-on-chip tools: Promising for future on-site detection test kits. TrAC, Trends Anal Chem 142. https://doi.org/10.1016/j.trac.2021.116325
Zhao F, Tian Y, Shen Q, Liu R, Shi R, Wang H, Yang Z (2019) A novel nanobody and mimotope based immunoassay for rapid analysis of aflatoxin B1. Talanta 195:55–61. https://doi.org/10.1016/j.talanta.2018.11.013
Wang Y-K, Yan Y-X, Li S-Q, Wang H-a, Ji W-H, Sun J-H (2013) Simultaneous Quantitative Determination of Multiple Mycotoxins in Cereal and Feedstuff Samples by a Suspension Array Immunoassay. J Agric Food Chem 61(46):10948–10953. https://doi.org/10.1021/jf4036029
Seok Y, Byun J-Y, Shim W-B, Kim M-G (2015) A structure-switchable aptasensor for aflatoxin B1 detection based on assembly of an aptamer/split DNAzyme. Anal Chim Acta 886:182–187. https://doi.org/10.1016/j.aca.2015.05.041
Zhu C, Liu D, Li Y, Shen X, Ma S, Liu Y, You T (2020) Ratiometric electrochemical aptasensor for ultrasensitive detection of Ochratoxin A based on a dual signal amplification strategy: Engineering the binding of methylene blue to DNA. Biosens Bioelectron. https://doi.org/10.1016/j.bios.2019.111814
Iliuk AB, Hu L, Tao WA (2011) Aptamer in Bioanalytical Applications. Anal Chem 83(12):4440–4452. https://doi.org/10.1021/ac201057w
Yang C, Wang Y, Marty J-L, Yang X (2011) Aptamer-based colorimetric biosensing of Ochratoxin A using unmodified gold nanoparticles indicator. Biosens Bioelectron 26(5):2724–2727. https://doi.org/10.1016/j.bios.2010.09.032
Khoshbin Z, Moeenfard M, Abnous K, Taghdisi SM (2023) Nano-gold mediated aptasensor for colorimetric monitoring of acrylamide: Smartphone readout strategy for on-site food control. Food Chem. https://doi.org/10.1016/j.foodchem.2022.133983
Wei M, Xin L, Feng S, Liu Y (2020) Simultaneous electrochemical determination of ochratoxin A and fumonisin B1 with an aptasensor based on the use of a Y-shaped DNA structure on gold nanorods. Microchim Acta 187(2). https://doi.org/10.1007/s00604-019-4089-y
Ren W, Pang J, Ma R, Liang X, Wei M, Suo Z, He B, Liu Y (2022) A signal on-off fluorescence sensor based on the self-assembly DNA tetrahedron for simultaneous detection of ochratoxin A and aflatoxin B1. Anal Chim Acta. https://doi.org/10.1016/j.aca.2022.339566
Suo Z, Liang X, Jin H, He B, Wei M (2021) A signal-enhancement fluorescent aptasensor based on the stable dual cross DNA nanostructure for simultaneous detection of OTA and AFB. Anal Bioanal Chem 413(30):7587–7595. https://doi.org/10.1007/s00216-021-03723-8
Huang R, He N, Li Z (2018) Recent progresses in DNA nanostructure-based biosensors for detection of tumor markers. Biosen Bioelectron 109:27–34. https://doi.org/10.1016/j.bios.2018.02.053
Gong X, Zhou W, Li D, Chai Y, Xiang Y, Yuan R (2015) RNA-regulated molecular tweezers for sensitive fluorescent detection of microRNA from cancer cells. Biosen Bioelectron 71:98–102. https://doi.org/10.1016/j.bios.2015.04.003
Xiong Z, Wang Q, Xie Y, Li N, Yun W, Yang L (2021) Simultaneous detection of aflatoxin B1 and ochratoxin A in food samples by dual DNA tweezers nanomachine. Food Chem. https://doi.org/10.1016/j.foodchem.2020.128122
Han B, Fang C, Sha L, Jalalah M, Al-Assiri MS, Harraz FA, Cao Y (2021) Cascade strand displacement reaction-assisted aptamer-based highly sensitive detection of ochratoxin A. Food Chem. https://doi.org/10.1016/j.foodchem.2020.127827
Park Y, Lee CY, Kang S, Kim H, Park KS, Park HG (2018) Universal, colorimetric microRNA detection strategy based on target-catalyzed toehold-mediated strand displacement reaction. Nanotechnology 29(8). https://doi.org/10.1088/1361-6528/aaa3a3
Song X, Ding Q, Pu Y, Zhang J, Sun R, Yin L, Wei W, Liu S (2021) Application of the Dimeric G-Quadruplex and toehold-mediated strand displacement reaction for fluorescence biosensing of ochratoxin A. Biosens Bioelectron. https://doi.org/10.1016/j.bios.2021.113537
Yin J, Liu Y, Wang S, Deng J, Lin X, Gao J (2018) Engineering a universal and label-free evaluation method for mycotoxins detection based on strand displacement amplification and G-quadruplex signal amplification. Sens Actuators B 256:573–579. https://doi.org/10.1016/j.snb.2017.10.083
Bader A, Cockroft SL (2020) Conformational enhancement of fidelity in toehold-sequestered DNA nanodevices. Chem Commun 56(38):5135–5138. https://doi.org/10.1039/d0cc00882f
Zhu X, Kou F, Xu H, Han Y, Yang G, Huang X, Chen W, Chi Y, Lin Z (2018) Label-free ochratoxin A electrochemical aptasensor based on target-induced noncovalent assembly of peroxidase-like graphitic carbon nitride nanosheet. Sens Actuators B 270:263–269. https://doi.org/10.1016/j.snb.2018.05.048
Bonel L, Vidal JC, Duato P, Castillo JR (2011) An electrochemical competitive biosensor for ochratoxin A based on a DNA biotinylated aptamer. Biosens Bioelectron 26(7):3254–3259. https://doi.org/10.1016/j.bios.2010.12.036
Wang Y, Hu X, Pei Y, Sun Y, Wang F, Song C, Yin M, Deng R, Li Z, Zhang G (2015) Selection of phage-displayed minotopes of ochratoxin A and its detection in cereal by ELISA. Anal Methods 7(5):1849–1854. https://doi.org/10.1039/c4ay02290d
Lai W, Guo J, Wu Q, Chen Y, Cai Q, Wu L, Wang S, Song J, Tang D (2020) A novel colorimetric immunoassay based on enzyme-regulated instant generation of Turnbull’s blue for the sensitive determination of ochratoxin A. Analyst 145(6):2420–2424. https://doi.org/10.1039/c9an02447f
Huang L, Chen K, Zhang W, Zhu W, Liu X, Wang J, Wang R, Hu N, Suo Y, Wang J (2018) ssDNA-tailorable oxidase-mimicking activity of spinel MnCo2O4 for sensitive biomolecular detection in food sample. Sens Actuators B 269:79–87. https://doi.org/10.1016/j.snb.2018.04.150
Liu R, Li W, Cai T, Deng Y, Ding Z, Liu Y, Zhu X, Wang X, Liu J, Liang B, Zheng T, Li J (2018) TiO2 Nanolayer-Enhanced Fluorescence for Simultaneous Multiplex Mycotoxin Detection by Aptamer Microarrays on a Porous Silicon Surface. ACS Appl Mater Interfaces 10(17):14447–14453. https://doi.org/10.1021/acsami.8b01431
Wu K, Ma C, Zhao H, Chen M, Deng Z (2019) Sensitive aptamer-based fluorescene assay for ochratoxin A based on RNase H signal amplification. Food Chem 277:273–278. https://doi.org/10.1016/j.foodchem.2018.10.130
Goud KY, Catanante G, Hayat A, Satyanarayana M, Gobi KV, Marty JL (2016) Disposable and portable electrochemical aptasensor for label free detection of aflatoxin B1 in alcoholic beverages. Sens Actuators B 235:466–473. https://doi.org/10.1016/j.snb.2016.05.112
Goud KY, Hayat A, Catanante G, Satyanarayana M, Gobi KV, Marty JL (2017) An electrochemical aptasensor based on functionalized graphene oxide assisted electrocatalytic signal amplification of methylene blue for aflatoxin B1 detection. Electrochim Acta 244:96–103. https://doi.org/10.1016/j.electacta.2017.05.089
Migliorini FL, dos Santos DM, Soares AC, Mattoso LHC, Oliveira Jr ON, Correa DS (2020) Design of a low-cost and disposable paper-based immunosensor for the rapid and sensitive detection of aflatoxin B1. Chemosensors 8(3). https://doi.org/10.3390/chemosensors8030087
Lerdsri J, Chananchana W, Upan J, Sridara T, Jakmunee J (2020) Label-free colorimetric aptasensor for rapid detection of aflatoxin B1 by utilizing cationic perylene probe and localized surface plasmon resonance of gold nanoparticles. Sens Actuators B. https://doi.org/10.1016/j.snb.2020.128356
Guo M, Hou Q, Waterhouse GIN, Hou J, Ai S, Li X (2019) A simple aptamer-based fluorescent aflatoxin B1 sensor using humic acid as quencher. Talanta. https://doi.org/10.1016/j.talanta.2019.120131
Wei M, Zhao F, Xie Y (2020) A novel gold nanostars-based fluorescent aptasensor for aflatoxin B1 detection. Talanta. https://doi.org/10.1016/j.talanta.2019.120599
Funding
This study was funded by the Key Scientific and Technological Project of Henan Province (212102310001), the Special Project for Collaborative Innovation of Zhengzhou (21ZZXTCX15), and the Innovative Funds Plan of Henan University of Technology (2021ZKCJ13).
Author information
Authors and Affiliations
Contributions
Luyang Zhao: Investigation, Methodology, Validation and Writing-Original Draft. Xiujun Liang: Validation and Writing-Original Draft. Yong Liu: Supervision, Resources. Min Wei: Conceptualization, Methodology, Resources, Supervision and Funding acquisition. Huali Jin: Methodology, Resources, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, L., Liang, X., Liu, Y. et al. A Novel Fluorescent Aptasensor Based on Dual-labeled DNA Nanostructure for Simultaneous Detection of Ochratoxin A and Aflatoxin B1. J Fluoresc 33, 1641–1650 (2023). https://doi.org/10.1007/s10895-022-03071-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-022-03071-5