Abstract
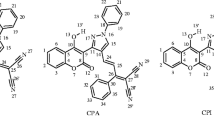
Two coumarin derivatives, 7-diethylamino-3-(4-nitrophenyl)coumarin (DNC) and 7-hydroxy-3-(4-nitrophenyl)coumarin (HNC), were synthesized via Knoevenagel condensation of salicylaldehyde derivatives with 4-nitrophenylacetonitrile and then cyclization reaction. Both of them were characterized by single-crystal X-ray diffraction. The molecules of DNC are stacked via π-π interaction, while the hydrogen bond interactions instead of π-π interaction were observed in the crystal packing of HNC. Both of DNC and HNC showed solvatochromic properties and aggregation-induced emission (AIE) activities, but the AIE characteristics of them were entirely different. HNC exhibited an AIE phenomenon as the result of the restriction of twisted intramolecular charge transfer (TICT), while DNC emitted peculiar dual fluorescence which was assigned to the emission based on the inhibition of TICT state formation and the emission from the TICT state respectively.






Similar content being viewed by others
Availability of data and material/Data availability
The crystallographic information files that have been deposited in the Cambridge Crystallographic Data Centre (CCDC 2,130,543 for DNC and 2,130,544 for HNC) which are freely available for all. The link for CCDC: https://www.ccdc.cam.ac.uk/.
Code Availability
not applicable.
References
Cai Y, Qin A, Tang BZ (2017) Siloles in optoelectronic devices. J Mater Chem C 5:7375–7389. https://doi.org/10.1039/C7TC02511D
Liu D, Wei JY, Tian WW, Jiang W, Sun YM, Zhao Z, Tang BZ (2020) Endowing TADF luminophors with AIE properties through adjusting flexible dendrons for highly efficient solution-processed nondoped OLEDs. Chem Sci 11:7194–7203. https://doi.org/10.1039/D0SC02194F
Li CT, Kuo YL, Kumar CHP, Huang PT, Lin JT (2019) Tetraphenylethylene tethered phenothiazine-based double-anchored sensitizers for high performance dye-sensitized solar cells. J Mater Chem A 7:23225–23233. https://doi.org/10.1039/C9TA09025H
Qian J, Tang BZ (2020) AIE luminogens for bioimaging and theranostics: from organelles to animals. Chem 3:56–91. https://doi.org/10.1016/j.chempr.2017.05.010
Gao M, Tang BZ (2020) AIE-based cancer theranostics. Coordin Chem Rev 402:213076. https://doi.org/10.1016/j.ccr.2019.213076
Mei J, Leung NLC, Kwok RTK, Lam JWY, Tang BZ (2015) Aggregation-induced emission: together we shine, united we soar. Chem Rev 115:11718–11940. https://doi.org/10.1021/acs.chemrev.5b00263
Gao BR, Wang HY, Hao YW, Fu LM, Fang HH, Jiang Y, Wang L, Chen QD, Xia H, Pan LY, Ma YG, Sun HB (2010) Time-resolved fluorescence study of aggregation-induced emission enhancement by restriction of intramolecular charge transfer state. J Phys Chem B 114:128–134. https://doi.org/10.1021/jp909063d. )
Abedi SAA, Chi W, Tan D, Shen T, Wang C, Ang ECX, Tan CH, Anariba F, Liu X (2021) Restriction of twisted intramolecular charge transfer enables the aggregation-induced emission of 1-( N,N -dialkylamino)-naphthalene derivatives. J Phys Chem A 125:8397–8403. https://doi.org/10.1021/acs.jpca.1c06263. )
Qian Y, Cai MM, Xie LH, Yang GQ, Wu SK, Huang W (2011) Restriction of photoinduced twisted intramolecular charge transfer. ChemPhysChem 12:397–404. https://doi.org/10.1002/cphc.201000457. )
Li J, Qian Y, Xie L, Yi Y, Li W, Huang W (2015) () From dark TICT state to emissive quasi-TICT state: the AIE mechanism of N-(3-(benzo[d]oxazol-2-yl)phenyl)-4-tert-butylbenzamide. J Phys Chem C 119: 2133–2141. https://doi.org/10.1021/jp5089433
Chiang Y, Lai Z, Chen C, Chang C, Liu B (2018) Construction of emission-tunable nanoparticles based on a TICT-AIEgen: Impact of aggregation-induced emission versus twisted intramolecular charge transfer. J Mater Chem B 6:2869–2876. https://doi.org/10.1039/C8TB00539G
Li Q, Niu ZG, Liu YL, Wang EJ (2020) Crystal structure and aggregation-induced emission of an azine derivative. Chin J Struct Chem 39:693–697. https://. doi10.14102/j.cnki.0254-5861.2011-2464
Zhang ZY, Niu ZG, Wang EJ (2019) () Chemical conversion of a coumarin derivative containing arylhydrazono: crystal structures and fluorescence properties. Chin J Struct Chem 38: 384–389. https://doi.10.14102/j.cnki.0254-5861.2011-2098
Sheldrick GM (2014) SHELXL-2014/7. Program for the Solution of Crystal Structures. University of Göttingen, Göttingen, )
Sasaki S, Drummen GPC, Konishi G (2016) () Recent advances in twisted intramolecular charge transfer (TICT) fluorescence and related phenomena in materials chemistry. J Mater Chem C 4: 2731 – 2743. https://doi.org/10.1039/C5TC03933A
Sun H, Tang XX, Miao BX, Yang Y, Ni Z (2018) () A new AIE and TICT-active tetraphenylethene-based thiazole compound: synthesis, structure, photophysical properties and application for water detection in organic solvents. Sensor Actuat B-Chem 267: 448 – 456. https://doi.org/10.1016/j.snb.2018.04.022
Wang DH, Chen LJ, Zhao X, Yan XP (2021) Enhancing near-infrared AIE of photosensitizer with twisted intramolecular charge transfer characteristics via rotor effect for AIE imaging-guided photodynamic ablation of cancer cells. Talanta 225:122046. https://doi.org/10.1016/j.talanta.2020.122046. )
Hu R, Lager E, Aguilar-Aguilar A, Liu J, Lam JWY, Sung HHY, Williams ID, Zhong Y, Wong KS, Peña-Cabrera E, Tang BZ (2009) Twisted Intramolecular Charge Transfer and Aggregation-Induced Emission of BODIPY Derivatives. J Phys Chem C 113:15845–15853. https://doi.org/10.1021/jp902962h. )
Zhang J, Xu B, Chen J, Wang L, Tian W (2013) Oligo(phenothiazine)s: twisted intramolecular charge transfer and aggregation-induced emission. J Phys Chem C 117:23117–23125. https://doi.org/10.1021/jp405664m. )
Sun H, Tang XX, Miao BX, Yang Y, Ni Z (2018) A new AIE and TICT-active tetraphenylethene-based thiazole compound: synthesis, structure, photophysical properties and application for water detection in organic solvents. Sens Actuat B-Chem 267:448–456. https://doi.org/10.1016/j.snb.2018.04.022
Sun H, Tang XX, Zhang R, Sun WH, Miao BX, Zhao Y, Ni ZH (2020) Tetraphenylethene-substituted benzothiadiazoles: AIE and TICT properties, tunable intramolecular conjugation and application in detecting trace water in organic solvents. Dyes Pigm 174:108051. https://doi.org/10.1016/j.dyepig.2019.108051
Hu Q, Gong T, Mao Y, Yin Q, Wang Y, Wang H (2021) Two-phase activated colorimetric and ratiometric fluorescent sensor for visual detection of phosgene via AIE coupled TICT processes. Spectrochim Acta A 253:119589. https://doi.org/10.1016/j.saa.2021.119589
Jiang M, Gu X, Lam JWY, Zhang Y, Kwok RTK, Wong KS, Tang BZ (2017) Two-photon AIE bio-probe with large stokes shift for specific imaging of lipid droplets. Chem Sci 8:5440–5446. https://doi.org/10.1039/C7SC01400G
Zhao Y, He S, Yang J, Sun H, Shen X, Han X, Ni Z (2018) Study on TICT emission of TPE-BODIPY derivatives mediated by methyl group on BODIPY. Opt Mater 81:102–108. https://doi.org/10.1016/j.optmat.2018.05.023
Naito H, Nishi K, Morisaki Y, Tanaka K, Chujo Y (2016) Solid-state emission of the anthracene-o-carborane dyad from the twisted-intramolecular charge transfer in the crystalline state. Angew Chem Int Ed 55:1–7. https://doi.org/10.1002/anie.201609656
Funding
This work is financially supported by the National Natural Science Foundation of China (22061016) and Program for Innovative Research Team in University (IRT-16R19).
Author information
Authors and Affiliations
Contributions
EW contributed to the study conception and design, and was the major contributor in writing the manuscript. The synthesis, spectrum test, data analysis and so on were performed by QL with the assistance of YZ. The X-ray diffraction data collection and structure determination was performed by ZN.
Corresponding author
Ethics declarations
Conflicts of Interest/Competing interests
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Q., Zhao, Y., Niu, Z. et al. Substituent Effect on AIE mechanism of two coumarin derivatives: uncommon TICT fluorescence in aggregation state. J Fluoresc 32, 1443–1448 (2022). https://doi.org/10.1007/s10895-022-02933-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-022-02933-2