Abstract
Microcystin-LR (MC-LR) is widely distributed in natural lakes and could strongly inhibit protein phosphatase activity; it is also a potent liver tumor promoter. Over the last two decades, tremendous efforts have been devoted to enhance the detection of MC-LR in water samples. However, the traditional method is complex and costly, and achieving the fast, sensitive, and accurate determination of MC-LR in the cells and natural lakes by using fluorescence signal changes is fairly difficult. Our work explores novel fluorescent probes that are capable of concurrently analyzing and detecting MC-LR in the cells and water. In this study, we introduce, for the first time, 5-AF and 6-AF as small-molecule fluorescent probes suitable for MC-LR detection in the cells and water samples based on fluorescence signal changes. We titrated 5-AF and 6-AF with MC-LR in pure water, scanned the fluorescence of the sample, and then obtained the equation the fluorescence intensity versus MC-LR concentration curve. MC-LR in lake water samples was crudely purified, and then 5-AF was added to measure its fluorescence peak. The fluorescence intensity of 5-AF is significantly enhanced with increasing MC-LR concentration. This enhancement trend is stable and could be mathematically modeled. We also comprehensively analyzed the mechanism and recognition principle of the probe response to MC-LR in natural lake water. Moreover, we believe that 5-AF may be capable of detecting exogenous MC-LR in cells. The results of this study reveal that these unique fluorescent probes may be applied to construct near-infrared fluorescent probes that could detect MC-LR levels in vivo.
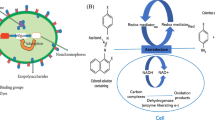
Graphical Abstract















Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this manuscript and its supplementary information files.
References
Bouaicha N, Miles CO, Beach DG, Labidi Z, Djabri A, Benayache NY, Nguyen-Quang T (2019) Structural Diversity, Characterization and Toxicology of Microcystins. Toxins 11(12). https://doi.org/10.3390/toxins11120714
Li LI, Xie P (2009) Hepatic Histopathological Characteristics and Antioxidant Response of Phytoplanktivorous Silver Carp Intraperitoneally Injected with Extracted Microcystins. Biomed Environ Sci 22(4):297–302. https://doi.org/10.1016/S0895-3988(09)60059-3
Ng A, Chinnappan R, Eissa S, Liu H, Tlili C, Zourob M (2012) Selection, Characterization, and Biosensing Application of High Affinity Congener-Specific Microcystin-Targeting Aptamers. Environ Sci Technol 46(19):10697–10703. https://doi.org/10.1021/es301686k
Pearson L, Mihali T, Moffitt M, Kellmann R, Neilan B (2010) On the Chemistry, Toxicology and Genetics of the Cyanobacterial Toxins, Microcystin, Nodularin Saxitoxin and Cylindrospermopsin Mar. Drugs 8(5):1650–1680. https://doi.org/10.3390/md8051650
Puddick J, Prinsep MR, Wood SA, Cary SC, Hamilton DP, Holland PT (2015) Further Characterization of Glycine-Containing Microcystins from the McMurdo Dry Valleys of Antarctica. Toxins 7(2):493–515. https://doi.org/10.3390/toxins7020493
Carmichael WW, Boyer GL (2016) Health impacts from cyanobacteria harmful algae blooms: Implications for the North American Great Lakes. Harmful Algae 54:194–212. https://doi.org/10.1016/j.hal.2016.02.002
Kaasalainen U, Fewer DP, Jokela J, Wahlsten M, Sivonen K, Rikkinen J (2012) Cyanobacteria produce a high variety of hepatotoxic peptides in lichen symbiosis. PNAS 109(15):5886–5891. https://doi.org/10.1073/pnas.1200279109
Metcalf JS, Richer R, Cox PA, Codd GA (2012) Cyanotoxins in desert environments may present a risk to human health. Sci Total Environ 421–422:118–123. https://doi.org/10.1016/j.scitotenv.2012.01.053
Armah AdlC, Maria GA, Anastasia H, Miguel P, Weihua S, Kevin EOS, Xuexiang H, Dionysios DD (2011) Can We Effectively Degrade Microcystins? - Implications on Human Health. Anticancer Agents Med Chem 11(1):19–37.https://doi.org/10.2174/187152011794941217
Harada K-I, Tsuji K, Watanabe MF, Kondo F (1996) Stability of microcystins from cyanobacteria—III. Effect of pH and temperature. Phycologia 35(sup6):83–88. https://doi.org/10.2216/i0031-8884-35-6S-83.1
Metcalf JS, Codd GA (2000) Microwave oven and boiling waterbath extraction of hepatotoxins from cyanobacterial cells. FEMS Microbiol Lett 184(2):241–246. https://doi.org/10.1111/j.1574-6968.2000.tb09021.x
Chen L, Wang Y, Giesy JP, Chen F, Shi T, Chen J, Xie P (2018) Microcystin-LR affects the hypothalamic-pituitary-inter-renal (HPI) axis in early life stages (embryos and larvae) of zebrafish. Environ Pollut 241:540–548. https://doi.org/10.1016/j.envpol.2018.05.024
Chen L, Xie P (2016) Mechanisms of Microcystin-induced Cytotoxicity and Apoptosis. Mini Rev Med Chem 16:1–1. https://doi.org/10.2174/1389557516666160219130407
Baohong C, Kang W, Xu D, Hui L (2021) Long-term changes in red tide outbreaks in Xiamen Bay in China from 1986 to 2017. Estuar Coast Shelf Sci 249:107095. https://doi.org/10.1016/j.ecss.2020.107095
Li X-Y, Yu R-C, Geng H-X, Li Y-F (2021) Increasing dominance of dinoflagellate red tides in the coastal waters of Yellow Sea, China. Mar Pollut Bull 168:112439. https://doi.org/10.1016/j.marpolbul.2021.112439
Zhang Y, Xu N, Li Y (2021) Effect of the Extracts of Sargassum fusiforme on Red Tide Microalgae in East China Sea. Front Mar Sci 8(871).https://doi.org/10.3389/fmars.2021.628095
Zhao K, Wang L, You Q, Pan Y, Liu T, Zhou Y, Zhang J, Pang W, Wang Q (2021) Influence of cyanobacterial blooms and environmental variation on zooplankton and eukaryotic phytoplankton in a large, shallow, eutrophic lake in China. Sci Total Environ 773:145421. https://doi.org/10.1016/j.scitotenv.2021.145421
Zhu C, Zhang J, Wang X, Yang Y, Chen N, Lu Z, Ge Q, Jiang R, Zhang X, Yang Y, Chen T (2021) Responses of cyanobacterial aggregate microbial communities to algal blooms. Water Res 196:117014. https://doi.org/10.1016/j.watres.2021.117014
Wang H, Xu C, Liu Y, Jeppesen E, Svenning JC, Wu, J, Zhang W, Zhou T, Wang P, Nangombe S, Ma J, Duan H, Fang J, Xie P (2021) From unusual suspect to serial killer: Cyanotoxins boosted by climate change may jeopardize megafauna. The Innovation 2(2):100092. https://doi.org/10.1016/j.xinn.2021.100092
Allum LL, Mountfort DO, Gooneratne R, Pasco N, Goussain G, Hall EAH (2008) Assessment of protein phosphatase in a re-usable rapid assay format in detecting microcystins and okadaic acid as a precursor to biosensor development. Toxicon 52(7):745–753. https://doi.org/10.1016/j.toxicon.2008.08.010
Díez-Quijada Jiménez L, Guzmán-Guillén R, Cătunescu GM, Campos A, Vasconcelos V, Jos Á, Cameán AM (2020) A new method for the simultaneous determination of cyanotoxins (Microcystins and Cylindrospermopsin) in mussels using SPE-UPLC-MS/MS. Environ Res 185:109284. https://doi.org/10.1016/j.envres.2020.109284
Pelander A, Ojanpera I, Lahti K, Niinivaara K, Vuori E (2000) Visual detection of cyanobacterial hepatotoxins by thin-layer chromatography and application to water analysis. Water Res 34(10):2643–2652. https://doi.org/10.1016/S0043-1354(00)00006-3
Thuret-Benoist H, Pallier V, Feuillade-Cathalifaud G (2019) Quantification of microcystins in natural waters by HPLC-UV after a pre-concentration step: validation of the analytical performances and study of the interferences. Environ Toxicol Pharmacol 72:103223. https://doi.org/10.1016/j.etap.2019.103223
Wu S, Duan N, Zhang H, Wang Z (2015) Simultaneous detection of microcysin-LR and okadaic acid using a dual fluorescence resonance energy transfer aptasensor. Anal Bioanal Chem 407(5):1303–1312. https://doi.org/10.1007/s00216-014-8378-3
Zheng B, Fu H, Berry JP, McCord B (2016) A rapid method for separation and identification of microcystins using capillary electrophoresis and time-of-flight mass spectrometry. J Chromatogr A 1431:205–214. https://doi.org/10.1016/j.chroma.2015.11.034
Liu Y, Meng F, He L, Liu K, Lin W (2016) A dual-site two-photon fluorescent probe for visualizing lysosomes and tracking lysosomal hydrogen sulfide with two different sets of fluorescence signals in the living cells and mouse liver tissues. Chem Comm 52(43):7016–7019. https://doi.org/10.1039/C6CC02368A
Liu Y, Niu J, Wang W, Ma Y, Lin W (2018) Simultaneous Imaging of Ribonucleic Acid and Hydrogen Sulfide in Living Systems with Distinct Fluorescence Signals Using a Single Fluorescent Probe Adv Sci 5(7):1700966. https://doi.org/10.1002/advs.201700966
Chen L, Chen J, Zhang X, Xie P (2016) A review of reproductive toxicity of microcystins. J Hazard Mater 301:381–399. https://doi.org/10.1016/j.jhazmat.2015.08.041
Su H, Feng Y, Chen J, Chen J, Ma S, Fang J, Xie P (2021) Determinants of trophic cascade strength in freshwater ecosystems: a global analysis. Ecology 102(7):e03370. https://doi.org/10.1002/ecy.3370
Su H, Wang R, Feng Y, Li Y, Li Y, Chen J, Xu C, Wang S, Fang J, Xie P (2020) Long-term empirical evidence, early warning signals and multiple drivers of regime shifts in a lake ecosystem. J Ecol 109(9):3182–3194. https://doi.org/10.1111/1365-2745.13544
Xie P (2003) Three-Gorges Dam: Risk to Ancient Fish. Science 302(5648):1149–1151. https://doi.org/10.1126/science.302.5648.1149b
Yang H, Xie P, Ni L, Flower RJ (2012) Pollution in the Yangtze. Science 337(6093):410. https://doi.org/10.1126/science.337.6093.410-a
Zhang X, Xie P, Wang W, Li D, Li L, Tang R, Lei H, Shi Z (2008) Dose-dependent effects of extracted microcystins on embryonic development, larval growth and histopathological changes of southern catfish (Silurus meridionalis). Toxicon 51(3):449–456. https://doi.org/10.1016/j.toxicon.2007.11.005
Kremer P, Fardanesh M, Ding R, Pritsch M, Zoubaa S, Frei E (2009) Intraoperative Fluorescence Staining of Malignant Brain Tumors Using 5-Aminofluorescein-Labeled Albumin. Oper Neurosurg 64:ONS53-ONS61. https://doi.org/10.1227/01.NEU.0000335787.17029.67
Ding R, Frei E, Fardanesh M, Schrenk H-H, Kremer P, Haefeli WE (2011) Pharmacokinetics of 5-Aminofluorescein-Albumin, a Novel Fluorescence Marker of Brain Tumors During Surgery. J Clin Pharmacol 51:672–678. https://doi.org/10.1177/0091270010372626
Hartlieb M, Pretzel D, Wagner M, Hoeppener S, Bellstedt P, Görlach M, Englert C, Kempe K, Schubert US (2015) Core cross-linked nanogels based on the self-assembly of double hydrophilic poly(2-oxazoline) block copolymers. J Mater Chem B 3:1748–1759. https://doi.org/10.1039/C4TB02069C
Xing Y, Wang S, Mao X, Zhao X, Wei D (2011) An Easy and Efficient Fluorescent Method for Detecting Aldehydes and Its Application in Biotransformation. J Fluoresc 21:587–594. https://doi.org/10.1007/s10895-010-0746-6
McHedlov-Petrossyan NO, Cheipesh TA, Roshal AD, Shekhovtsov SV, Moskaeva EG, Omelchenko IV (2019) Aminofluoresceins Versus Fluorescein: Peculiarity of Fluorescence. J Phys Chem A 123:8860–8870. https://doi.org/10.1021/acs.jpca.9b05812
Kong H, Lu Y, Dong X, Zeng S (2021) Quantification of the Coordination Degree between Dianchi Lake Protection and Watershed Social-Economic Development: A Scenario-Based Analysis. Sustainability 13(1):116. https://doi.org/10.3390/su13010116
Li Y, Li Y, Zhou Y, Shi Y, Zhu X (2012) Investigation of a coupling model of coordination between urbanization and the environment. J Environ Manage 98:127–133. https://doi.org/10.1016/j.jenvman.2011.12.025
Chen Q, Wang S, Ni Z, Guo Y, Liu X, Wang G, Li H (2021) No-linear dynamics of lake ecosystem in responding to changes of nutrient regimes and climate factors: Case study on Dianchi and Erhai lakes, China. Sci Total Environ 781: 146761. https://doi.org/10.1016/j.scitotenv.2021.146761
Zheng L, An Z, Chen X, Liu H (2021) Changes in Water Environment in Erhai Lake and Its Influencing Factors. Water 13(10):1362. https://doi.org/10.3390/w13101362
Yang S, Chen X, Lu J, Hou X, Li W, Xu Q (2021) Impacts of agricultural topdressing practices on cyanobacterial bloom phenology in an early eutrophic plateau Lake, China. J Hydrol 594:125952. https://doi.org/10.1016/j.jhydrol.2020.125952
Harada K, Ogawa K, Matsuura K, Murata H, Suzuki M, Watanabe MF, Itezono Y, Nakayama N (1990) Structural determination of geometrical isomers of microcystins LR and RR from cyanobacteria by two-dimensional NMR spectroscopic techniques. Chem Res Toxicol 3(5):473–481. https://doi.org/10.1021/tx00017a014
Namikoshi M, Rinehart KL, Sakai R, Sivonen K, Carmichael WW (1990) Structures of three new cyclic heptapeptide hepatotoxins produced by the cyanobacterium (blue-green alga) Nostoc sp. strain 152. J Org Chem 55(25):6135–6139. https://doi.org/10.1021/jo00312a019
Joniak J, Stankovičová H, Filo J, Gaplovská-Kyselá K, Garaj V, Cigáň M (2020) Small-molecule coumarin fluorescent pH probes for extremely acidic conditions. Sens Actuators B Chem 307:127646. https://doi.org/10.1016/j.snb.2019.127646
Acknowledgements
This work was financially supported by NSFC (51973082, 51503077), Yunnan Provincial Department of Science and Technology (202001BB050078) and Special project for social development of Yunnan Province (202103AC100001). This research was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB31000000), high-level talents program of Yunnan University (20198006, 20200105) and the Postgraduate Research and Innovation Foundation of Yunnan University (2021Y046).
Funding
National Natural Science Foundation of China, 51973082, Yong Liu, Special project for social development of Yunnan Province, 202103AC100001, Yong Liu.
Author information
Authors and Affiliations
Contributions
Yipeng Liu conceived the ideas supported by previous ideas of Yong Liu and Ping Xie. All authors conceptualized the paper; Yipeng Liu and Bingyan Li completed data measurement and collection; Yipeng Liu and Yong Liu wrote this manuscript; All authors contributed critically to the drafts and gave final approval for publication.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent to Participate
Not applicable.
Conflicts of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Y., Li, B., Zhang, H. et al. Detection of Microcystin-LR in the Cells and Natural Lake Water Samples by A Unique Fluorescence-Based Method. J Fluoresc 32, 505–519 (2022). https://doi.org/10.1007/s10895-021-02882-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-021-02882-2