Abstract
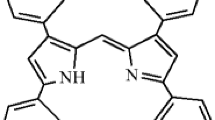
Monoiodo- and dibromsubstituted dipyrromethenes HL1 – HL3 were described as a highly sensitive and selective «Off-On» fluorescent chemosensor for Zn2+ based on the chelation-enhanced fluorescence (CHEF) effect. Сoordination reactions of HL1 – HL3 with Zn2+ cations are accompanied by a significant (124 to 215-fold) increase in fluorescence intensity against the background of other metal ions in the binary propanol-1/cyclohexane mixture (1:30). The fluorometric detection limit of Zn2+ ions using HL1 – HL3 sensors is from 3.0∙10−8 to 3.3·10−9 mol/L. The presence of Na+, K+, Ca2+, Mg2+, Mn2+, Ni2+, Co2+, Pb2+ cations does not interfere with the detection of Zn2+. Complexation reactions are accompanied by a visual change in the color of the solution from yellow-orange to pink-raspberry so that the HL1 – HL3 ligands can also be used as a «naked-eye» indicators of the presence of Zn2+ ions.









Similar content being viewed by others
Data Availability
All the data are available.
References
Wu D, Sedgwick AC, Gunnlaugsson T, Akkaya EU, Yoon J, James TD (2017) Fluorescent chemosensors: the past, present and future. Chem Soc Rev 46:7105–7123. https://doi.org/10.1039/C7CS00240H
Hao Z, Zhu R, Chen PR (2018) Genetically encoded fluorescent sensors for measuring transition and heavy metals in biological systems. Curr Opin Chem Biol 43:87–96. https://doi.org/10.1016/j.cbpa.2017.12.002
Rasheed T, Bilal M, Nabeel F, Iqbal HN, Li C, Zhou Y (2018) Fluorescent sensor based models for the detection of environmentally-related toxic heavy metals. Sci Total Environ 615:476–485. https://doi.org/10.1016/j.scitotenv.2017.09.126
Berg JM, Shi Y (1996) The galvanization of biology: a growing appreciation for the roles of zinc. Science 271:1081–1085. https://doi.org/10.1126/science.271.5252.1081
Frederickson CJ, Koh JY, Bush AI (2005) The neurobiology of zinc in health and disease. Nat Rev Neurosci 6:449–462. https://doi.org/10.1038/nrn1671
Que EL, Domaille DW, Chang CJ (2008) Metals in neurobiology: probing their chemistry and biology with molecular imaging. Chem Rev 108:1517–1549. https://doi.org/10.1021/cr078203u
Suhy DA, Simon KD, Linzer DH, O'Halloran TV (1999) Metallothionein is part of a zinc scavenging mechanism for cell survival under conditions of extreme zinc deprivation. J Biol Chem 274:9183–9192. https://doi.org/10.1074/jbc.274.14.9183
Chausmer AB (1998) Zinc, insulin and diabetes. J Am Coll Nutr 17:109–115. https://doi.org/10.1080/07315724.1998.10718735
Bush AI (2003) The metallobiology of Alzheimer's disease. Trends Neurosci 26:207–214. https://doi.org/10.1016/j.nurt.2008.05.001
Strausak D (2001) Copper in disorders with neurological symptoms: Alzheimer's, Menkes, and Wilson diseases. Brain Res Bull 55:175–185. https://doi.org/10.1016/s0361-9230(01)00454-3
Xie X, Smart TG (1991) A physiological role for endogenous zinc in rat hippocampal synaptic neurotransmission. Nature 349:521–524. https://doi.org/10.1038/349521a0
Smith JL, Xiong S, Markesbery WR, Lovell MA (2006) Altered expression of zinc transporters-4 and -6 in mild cognitive impairment, early and late Alzheimer's disease brain. Neuroscience 140:879–888. https://doi.org/10.1016/j.neuroscience.2006.02.049
Zhang JF, Bhuniya S, Lee YH, Bea CW, Lee JH, Kim JS (2010) Novel 2,2′-bipyridine-modified calix[4]arenes: ratiometric fluorescent chemosensors for Zn2+ ion. Tetrahedron Lett 51:3719–3723. https://doi.org/10.1016/j.tetlet.2010.04.135
Liu Y, Feng Q, Li N, Li K, Hou H, Zhang B (2018) ‘Turn-on’ fluorescent chemosensors based on naphthaldehyde-2-pyridinehydrazone compounds for the detection of zinc ion in water at neutral pH. Luminescence 33:29–33. https://doi.org/10.1002/bio.3368
Song H, Zhang Z (2019) A quinoline-based ratiometric fluorescent probe for discriminative detection of Zn2+ and Cd2+ with different bindingmodes, and its Zn2+ complex for relay sensing of pyrophosphate and adenosine triphosphate. Dyes Pigm 165:172–181. https://doi.org/10.1016/j.dyepig.2019.02.011
Bai X, Yan J, Qin J, Yang Z (2018) A multi-ion fluorescent probe for Mg2+/Zn2+ based on a novel chromone-dendron Schiff base. Inorg Chim Acta 474:44–50. https://doi.org/10.1016/j.ica.2018.01.008
Wang L, Li W, Zhi W, Huang Y, Han J, Wang Y, Ren Y, Ni L (2018) A new coumarin schiff based fluorescent-colorimetric chemosensor for dual monitoring of Zn2+ and Fe3+ in different solutions: An application to bio-imaging. Sensors Actuators B Chem 260:243–254. https://doi.org/10.1016/j.snb.2017.12.200
Li J, Chen Y, Chen T, Qiang J, Zhang Z, Wei T, Zhang W, Wang F, Chen X (2018) A benzothiazole-based fluorescent probe for efficient detection and discrimination of Zn2+ and Cd2+, using cysteine as an auxiliary reagent. Sensors Actuators B Chem 268:446–455. https://doi.org/10.1016/j.snb.2018.04.130
Mehta PK, Oh ET, Park HJ, Lee KH (2017) Ratiometric fluorescent probe based on symmetric peptidyl receptor with picomolar affinity for Zn2+ in aqueous solution. Sensors Actuators B Chem 245:996–1003. https://doi.org/10.1016/j.snb.2017.01.154
Lee H, Lee HS, Reibenspies JH, Hancock RD (2012) Mechanism of “Turn-on” Fluorescent Sensors for Mercury(II) in Solution and Its Implications for Ligand Design. Inorg Chem 51:10904–10915. https://doi.org/10.1021/ic301380w
Xie Y, Li X, Hill JP, Zhang W, Zhu W (2011) Selective and sensitive “turn-on” fluorescent Zn2+ sensors based on di- and tripyrrins with readily modulated emission wavelengths. Chem Commun 47:5431–5433. https://doi.org/10.1039/c1cc11493j
Dudina NA, Antina EV, Guseva GB, V’yugin AI (2014) The high sensitive and selective “off-on” fluorescent Zn2+ sensor based on the bis(2,4,7,8,9-pentamethyldipyrrolylmethene-3-yl)methane. J Fluoresc 24:13–17. https://doi.org/10.1007/s10895-013-1278-7
Dudina NA, Antina EV, Sozonov DI, V’yugin AI (2015) Effect of alkyl substitution in 3,30-Bis(dipyrrin) on chemosensor activity of fluorescent detection of Zn2+ cations. Russ J Org Chem 51:1155–1161. https://doi.org/10.1134/S107042801508014X
Bumagina NA, Antina EV, Berezin MB, Ksenofontov AA, V’yugin AI, Nikonova AY (2016) A new sensitive and selective «off-on» fluorescent Zn2+ chemosensor based on 3,3′,5,5′-tetraphenylsubstituted dipyrromethene. J Fluoresc 26:1967–1974. https://doi.org/10.1007/s10895-016-1890-4
Antina EV, Bumagina NA, V'yugin AI, Solomonov AV (2017) Fluorescent indicators of metal ions based on dipyrrоmethene platform. Dyes Pigm 136:368–381. https://doi.org/10.1016/j.dyepig.2016.08.070
Dudina NA, Antina EV, Guseva GB, V’yugin AI, Semeikin AS (2013) New fluorescent chemosensor for Zn2+ ions on the basis of 3,3'-Bis(dipyrrolylmethene). Russ J Org Chem 49:1734–1739. https://doi.org/10.1134/S107042801312004X
Nikonova AY, Dudina NA, Nuraneeva EN, Antina EV, Kuznetsova RT, Aksenova YV, Tel’minov EN, Mayer GV (2016) Optical properties of zinc(II) and boron(III) dipyrrinates with different structures. Opt Spectrosc 120:395–402. https://doi.org/10.1134/S0030400X16030176
Dudina NA, Antina EV, Berezin MB, Guseva GB, V'Yugin AI, Nikonova AY (2013) Preparation and spectral properties of Zn(II) complexes with aryl-substituted dipyrrolylmethene and azadipyrrolylmethene. Russ J Gen Chem 83:1941–1943. https://doi.org/10.1134/S107036321310023X
Kuznetsova RT, Aksenova YV, Bashkirtsev DE, Prokopenko AA, Tel'minov EN, Mayer GV, Dudina NA, Antina EV, Nikonova AY, Berezin MB, Semeikin AS (2015) Photonics of zinc(II) and boron(III) chelates with methyl- And phenyl-substituted dipyrromethenes and azadipyrromethenes. High Energy Chem 49:16–23. https://doi.org/10.1134/S0018143915010075
Antina EV, Berezin MB, Dudina NA, Burkova SL, Nikonova AY (2014) Synthesis, spectral-luminescent properties of B(III) and Zn(II) complexes with alkyl- and aryl-substituted dipyrrins and azadipyrrins. Russ J Inorg Chem 59:1187–1194. https://doi.org/10.1134/S0036023614100027
Nuraneeva EN, Guseva GB, Antina EV (2017) Effect of halogenation features on the stability of zinc(II) complexes with monoiodo- and dibromodipyrromethenes. Russ J Gen Chem 87:1550–1556. https://doi.org/10.1134/S1070363217070180
Nuraneeva EN, Guseva GB, Antina EV, Berezin MB, V'Yugin AI (2016) Synthesis and luminescent properties of zinc(II) complexes with iodo- and bromosubstituted 2,2′-dipyrrines. J Lumin 170:248–254. https://doi.org/10.1016/jjlumin.2015.10.061
Aksenova IV, Kuznetsova RT, al PIP e (2017) Spectral-kinetic properties and efficiency of singlet oxygen generation by some dipyrromethenes. J Photochem Photobiol A 344:206–211. https://doi.org/10.1016/jjphotochem.2017.05.022
González-Delgado JA, Kennedy PJ, Ferreira M, Tomé JC, Sarmento B (2016) Use of Photosensitizers in Semisolid Formulations for Microbial Photodynamic Inactivation. J Med Chem 59:4428–4442. https://doi.org/10.1021/acs.jmedchem.5b01129
Turksoy A, Yildiz D, Akkaya EU (2019) Photosensitization and controlled photosensitization with BODIPY dyes. Coord Chem Rev 379:47–64. https://doi.org/10.1016/j.ccr.2017.09.029
Durantini AM, Heredia DA, Durantini JE, Durantini EN (2018) BODIPYs to the rescue: Potential applications in photodynamic inactivation. Eur J Med Chem 144:651–661. https://doi.org/10.1016/j.ejmech.2017.12.068
Anbu S, Shanmugaraju S, Ravishankaran R, Karande AA, Mukherjee PS (2012) A phenanthrene based highly selective fluorogenic and visual sensor for Cu2+ ion with nanomolar detection limit and its application in live cell imaging. Inorg Chem Commun 25:26–29. https://doi.org/10.1016/j.inoche.2012.08.012
Shrivastava A, Gupta V (2011) Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chron Young Sci 2:21–25. https://doi.org/10.4103/2229-5186.79345
Dong Y-J, Meng Z, Ho C-L, Wong W-Y (2017) A novel “turn-on” dipyrrin-type fluorescent detector of Zn2+ based on tetraphenylethene. Tetrahedron 73:3305–3309. https://doi.org/10.1016/j.tet.2017.04.039
Mei Q, Shi Y, Hua Q, Tong B (2013) Phosphorescent chemosensor for Hg2+ based on an iridium(III) complex coordinated with 4-phenylquinazoline and carbazole dithiocarbamate. Chem Commun 49:7231–74931 https://doi.org/10.1039/C5RA09609J
Wang B, Anslyn EV (eds) (2011) Chemosensors: principles, strategies, and applications. John Wiley & Sons
Guseva GB, Dudina NA, Antina EV, V'Yugin AI, Semeikin AS (2009) Complexation between decamethyl-3,3′-bis(dipyrrolylmethene) and zinc(II), copper(II), and cobalt(II) acetates. Russ J Coord Chem 35:65–72. https://doi.org/10.1134/S1070328409010138
Rumyantsev EV, Desoki A, Antina EV (2009) Trends in formation and stability of homo- and heteroligand d-metal complexes with 2,2′-dipyrrolylmethenes in dimethylformamide. Russ J Inorg Chem 54:1735–1741. https://doi.org/10.1134/S0036023609110084
Dudina NA, Antina EV, Guseva GB (2011) Regularities of the formation of binuclear homo- and heteroleptic complexes of d-metals with 3,3′-Bis(dipyrrolylmethenes) in DMF. Russ J Coord Chem 37:333–342. https://doi.org/10.1134/S1070328411040026
Guseva GB, Antina EV, V'yugin AI, Loginova AE (2008) Complex formation of Cu(II), Ni(II), Zn(II), Co(II), and Cd(II) acetates with 3,3′,4,4′,5,5′-hexamethyl dipyrrolylmethene. Russ J Coord Chem 34:599–605. https://doi.org/10.1134/S1070328408080071
Ramamurthy V, Schanze KS (eds) (2001) Optical sensors and switches. CRC Press
Valeur B, Leray I (2000) Design principles of fluorescent molecular sensors for cation recognition. Coord Chem Rev 205:3–40. https://doi.org/10.1016/S0010-8545(00)00246-0
Benesi HA, Hildebrand JH (1949) A spectrophotometr icinvestigation of the interaction of iodine with aromatic hydrocarbons. J Am Chem Soc 71:2703–2707. https://doi.org/10.1021/ja01176a030
Das S, Sahana A, Lohar S, Sarkar B, Kanti Mukhopadhya S, Banerjee A, Das D (2014) A visible light excitable pyrenena phthaleneconjugate for ON fluorescencesensing of histidine in living cells. RSC Adv 4:7495–7499. https://doi.org/10.1039/c3ra46604c
Liu H, Tan Y, Dai Q, Liang H, Song J, Qu J, Wong WY (2018) A simple amide fluorescent sensor based on quinoline for selective and sensitive recognition of zinc(II) ions and bioimaging in living cells. Dyes Pigm 158:312–318. https://doi.org/10.1016/j.dyepig.2018.05.026
Rawat M, Rawat DS (2018) Copper oxide nanoparticle catalysed synthesis of imidazo[1,2-a]pyrimidine derivatives, their optical properties and selective fluorescent sensor towards zinc ions. Tetrahedron Lett 59:2341–2346. https://doi.org/10.1016/j.tetlet.2018.05.005
Casanueva-Marenco MJ, Diaz-de-Alba M, Herrera-Armario A, Galindo-Riano MD, Granado-Castro MD (2020) Design and optimization of a single-use optical sensor based on a polymer inclusion membrane for zinc determination in drinks, food supplement and foot health care products. Mater Sci Eng C 110:110680. https://doi.org/10.1016/j.msec.2020.110680
Guseva GB, Antina EV, Berezin MB, V'yugin AI (2004) Thermodynamics of copper(II), zinc(II), cobalt(II), mercury(II), and nickel(II) complexation with α,α-dipyrrolylmethene in DMF. Rus J Coord Chem 30:30–33. https://doi.org/10.1023/B:RUCO.0000011639.86600.87
Krämer R (1998) Fluorescent chemosensors for Cu2+ ions: fast, selective, and highly sensitive. Angew Chem Int Ed 37:772–773. https://doi.org/10.1002/(SICI)1521-3773(19980403)37:6<772::AID-ANIE772>3.0.CO;2-Z
Funding
This work was supported by the Russian Foundation for Basic Research as part of research on project No. 19-33-60052 «Perspective».
Author information
Authors and Affiliations
Contributions
All authors were involved with the design and interpretation of experiments and with the writing of the manuscript. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
Not applicable.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Code Availability
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nuraneeva, E.N., Guseva, G.B. & Antina, E.V. A New «off-on» Fluorescence Zinc Ion Sensors Based on Iodo- and Bromosubstituted Dipyrromethenes. J Fluoresc 31, 415–425 (2021). https://doi.org/10.1007/s10895-020-02670-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-020-02670-4