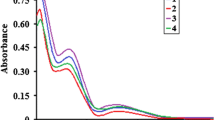
Abstract
Copper(I) complexes of the formula [Cu(L)(PPh3)2]X (1–4) (X = Cl(1), ClO4(2), BF4(3) and PF6(4)) [where L = N-(2-{[(2E)-2-(4-nitrobenzylidenyl)hydrazinyl]carbonyl}phenyl)benzamide; PPh3 = triphenylphosphine] have been prepared by the condensation of N-[2-(hydrazinocarbonyl)phenyl]benzamide with 4-nitrobenzaldehyde followed by the reaction with CuCl, [Cu(MeCN)4]ClO4, [Cu(MeCN)4]BF4 and [Cu(MeCN)4]PF6 in presence of triphenylphosphine as a coligand. Complexes 1–4 were then characterized by elemental analyses, FTIR, UV-visible and 1H NMR spectroscopy. Mononuclear copper(I) complexes 1–4 were formed with L in its keto form by involvement of azomethine nitrogen and the carbonyl oxygen along with two PPh3 groups. A single crystal X-ray diffraction study of the representative complex [(Cu(L)(PPh3)2]CIO4 (2) reveals a distorted tetrahedral geometry around Cu(I). Crystal data of (2): space group = C2/c, a = 42.8596 (9) Å, b = 14.6207 (3) Å, c = 36.4643 (7) Å, V = 20,653.7 (7) Å3, Z = 16. Complexes 1–4 exhibit quasireversible redox behaviour corresponding to a Cu(I)/Cu(II) couple. All complexes show blue-green emission as a result of fluorescence from an intra-ligand charge transition (ILCT), ligand to ligand charge transfer transition (LLCT) or mixture of both. Significant increase in size of the counter anion shows marked effect on quantum efficiency and lifetime of the complexes in solution.




Similar content being viewed by others
References
Hattori S, Wada Y, Yanagida S, Fukuzumi S (2005) Blue copper model complexes with distorted tetragonal geometry acting as effective electron-transfer mediators in dye-sensitized solar cells. J Am Chem Soc 127:9648–9654
Zeng L, Miller EW, Pralle A, Isacoff EY, Chang CJ (2006) A selective turn-on fluorescent sensor for imaging copper in living cells. J Am Chem Soc 128:10–11
Miller MT, Karpishin TB (1999) Oxygen sensing by photoluminescence quenching of a heteroleptic copper (I) bis(phenanthroline) complex immobilized in polystyrene. Sensors Actuators B Chem 61:222–224
Perruchas S, Goff XFL, Maron S, Maurin I, Guillen F, Garcia A, Gacoin T, Boilot JP (2010) Mechanochromic and thermochromic luminescence of a copper iodide cluster. J Am Chem Soc 132:10967–10969
Li X, Ding J, Jin W, Cheng Y (2009) Synthesis, crystal structures and spectroscopic properties of copper(I) complexes containing b-dialdiminato supporting ligands. Inorg Chim Acta 362:233–237
Mamais M, Cox PJ, Aslanidis P (2008) Three- and four-coordinate copper(I) halide complexes of 2-(diphenylphosphano)benzaldehyde: dimerization induced by thione-S ligation. Polyhedron 27:175–180
Gennari M, Tegoni M, Lanfranchi M, Pellinghelli MA, Giannetto M, Marchio L (2008) CuI Complexes with N,N′,S,S′ Scorpionate Ligands: Evidence for Dimer-Monomer Equilibria. Inorg Chem 47:2223–2232
Qi-Ming Q, Liu M, Li Z-F, Qiong-Hua J, Huang X, Zhang Z-W, Zhang C-L, Meng Q-X (2014) Synthesis, structure, terahertz spectroscopy and luminescent properties of copper(I) complexes with mercaptan ligands and triphenylphosphine. J Mol Struct 1062:125–132
Małecki JG, Łakomska I, Maroń A, Szala M, Fandzloch M, Nycz JE (2015) Phosphorescent emissions of phosphine copper(I) complexes bearing 8-hydroxyquinoline carboxylic acid analogue ligands. J Lumin 161:382–388
Darensbourg DT, Larkins DL, Reibenspies JH (1998) Bis(triphenylphosphine) copper (I) complexes of Orotate and L-dihydroorotate. Inorg Chem 37:6125–6128
Li D, Li R-Z, Zheng N, Qi Z-Y, Feng X-L, Cai J-W (2003) Synthesis and crystal structure of photoluminescent copper(I)–phosphine complexes with oxygen and nitrogen donor ligands. Inorg Chem Commun 6:469–473
Ji YF, Wang R, Ding S, Du CF, Liu ZI (2012) Synthesis, crystal structures and fluorescence studies of three new Zn(II) complexes with multidentate Schiff base ligands. Inorg Chem Commun 16:47–50
Moraes RS, Aderne RE, Cremona M, Rey NA (2016) Luminescent properties of a di-hydrazone derived from the antituberculosis agent isoniazid: potentiality as an emitting layer constituent for OLED fabrication. Optical. Mater 52:186–191
Wu JZ, De Angelis F, Carrell TG, Yap GPA, Sheats J, Car R, Dismukes GC (2006) Tuning the Photoinduced O2-evolving reactivity of Mn4O4 7+, Mn4O4 6+, and Mn4O3(OH)6+ manganese-Oxo Cubane complexes. Inorg Chem 45:189–195
Wen-Xiu Ni, Mian Li, Xiao-Ping Zhou, Zhen Li, Xiao-Chun Huang and Dan Li (2007) pH-Induced formation of metalloligand: increasing structure dimensionality by tuning number of ligand functional sites. Chem Commun 3479–3481.
Plass W, Yozgaltli HP, Anorg Z (2003) Synthesis, Reactivity, and Structural Characterization of Dioxovanadium(V) Complexes with Tridentate Schiff Base Ligand: Vanadium Complexes in Supramolecular Networks. Allg Chem 629:65–70
E. Colacia, M. Ghazi, R. Kivekas, M. Klinga, F. lioret, J. M. Moreno (2000) A Rational Design for Imidazolate-Bridged Linear Trinuclear Compounds from Mononuclear Copper(II) Complexes with 2-[((Imidazol-2-ylmethylidene)amino)ethyl]pyridine (HL): Syntheses, Structures, and Magnetic Properties of [Cu(L)(hfac)M(hfac)2Cu(hfac)(L)] (M) Zn(II), Cu(II), Mn(II). Inorg Chem 39: 2770–2776.
Pouralimardan O, Chamayou A-C, Janiak C, Hosseini-Monfared H (2007) Hydrazone Schiff base-manganese(II) complexes: synthesis, crystal structure and catalytic reactivity. Inorg Chim Acta 360:1599–1608
Basu C, Chowdhury S, Banerjee R, Evans HS, Mukherjee S (2007) A novel blue luminescent high-spin iron(III) complex with interlayer O–H---Cl bridging: synthesis, structure and spectroscopic studies. Polyhedron 26:3617–3624
Perez-Rebolledo A, Piro OF, Castellano EE, Teixeira LR, Batista AA, Beraldo H (2006) Metal complexes of 2-benzoylpyridine semicarbazone: spectral, electrochemical and structural studies. J Mol Struct 794:18–23
Buss JL, Neuzil J, Ponka P (2002) The role of oxidative stress in the toxicity of pyridoxal isonicotinoyl hydrazone (PIH) analogues. Biochem Soc Trans 30:755–757
Seelam N, Prasanthi S, Shrivastava SP (2016) Synthesis and anti-mycobacterial studies of some novel 1,3,4-Thiadiazoles. Der Pharma Lett 8(1):1–7
A. I. Vogel (1955) Text book of practical organic chemistry, third Ed., Woolwich Polytechnic, London 190.
Duriska MB, Neville SM, Lu J, Iremonger SS, Boas JF, Kepert CJ, Batten SR (2009) Systematic metal variation and solvent and hydrogen-gas storage in supramolecular nanoballs. Angew Chem Int Ed 48:8919–8922
Kubas GJ, Monzyk B, Crubliss AL (1979) Tetrakis(acetonitrile) copper(I) hexafluorophosphate. Inorg Synth 19:90–92
Fielden J, Long D-L, Cronin L, Kogerler P (2009) Synthesis of Cu(I) octamolybdates using tetrakis-acetonitrilecopper (I) hexafluorophosphate. Polyhedron 28:2803–2807
Shashidhar, Chopra D, Shivashankar SA, Row TNG (2006) N-(2-{[(2E)-2-(2-Hydroxybenzylidene)hydrazino]carbonyl}phenyl)benzamide. Acta Cryst E62:04473–04475
Betteridge PW, Carruthers JR, Cooper RI, Prout K, Watkin DJ (2003) CRYSTALS version 12:software for guided crystal structure analysis. J Appl Crystallogr 36:1487–1487
Chavan SS, Sawant VA, Jadhav AN (2014) Copper(II) complexes of N-(2-{[(2E)-2-(2-Hydroxy-(5-substituted)-benzylidene)-hydrazino]carbonyl}phenyl)benzamide ligands and heterocyclic coligands. Spectrochim Acta Part A 117:360–365
Chavan SS, Gaikwad GA, Sawant VA, Lahiri GK (2011) Synthesis, characterization and luminescence properties of copper(I) complexes containing 2-phenyl-3-(benzylamino)-1,2-dihydroquinazolin-4(3H)-one and triphenylphosphine as ligands. Polyhedron 30:1871–1875
Rapheal PF, Manoj E, Kurup MRP (2007) Copper(II) complexes of N(4)-substituted thiosemicarbazones derived from pyridine-2-carbaldehyde: crystal structure of a binuclear complex. Polyhedron 26:818–828
Vinogradova KA, Krivopalov VP, Nikolaenkova EB, Pervukhina NV, Naumov DY, Sheludyakova LA, Bushuev MB (2012) Copper(II) and copper(I) tetrafluoroborate complexes with 4-(3,5-diphenyl-1H-pyrazol-1-yl)-6-(piperidin-1-yl) pyrimidine (L): synthesis, structures and luminescence. Inorg Chim Acta 386:116–121
Govindswamy P, Mozharivskyj YA, Kollipara MR (2004) Synthesis and characterization of cyclopentadienylruthenium(II) complexes containing N,N′-donor Schiff base ligands: crystal and molecular structure of [(ƞ5-C5H5)Ru(C5H4N-2-CH = N–C6H4–p-OCH3)(PPh3)]PF6. Polyhedron 23:1567–1572
Ray A, Banerjee S, Butcher RJ, Desplanches C, Mitra S (2008) Two new end-on azido bridged dinuclear copper(II) and cobalt(III) complexes derived from the (E)-N′-((pyridin-2-yl)methylene) acetohydrazide Schiff base ligand: characterisation, crystal structures and magnetic study. Polyhedron 27:2409–2415
P. Krishnamoorthy, P. Sathyadevi, K. Senthilkumar, P. Thomas Muthiah, R. Ramesh, N. Dharmaraj (2011) Copper(I) hydrazone complexes: Synthesis, structure, DNA binding, radical scavenging and computational studies. Inorg Chem Commun14: 1318–1322.
Chavan SS, Sawant SK, Pawal SB, More MS (2016) Copper(I) complexes of 2-methoxy-(5-trifluoromethyl-phenyl)-pyridine-2yl-methylene-amine: Impact of phosphine ancillary ligands on luminescence and catalytic properties of the copper(I) complexes. Polyhedron 105:192–199
Chen M-S, Hua Q, Bai Z-S, Okamura T-a, Zhi S, Wei-Yin S, Ueyama N (2010) Syntheses and characterization of inorganic–organic hybrids with 4-(isonicotinamido)phthalate and some divalent metal centers. Polyhedron 29:2454–2461
Zu Y, Ma Y, Zhu J (2013) The counter anion effect of ion-type phosphorescent dye tris(4,7-diphenyl-1,10-phenanthroline)ruthenium(II) complexes as dopant for light-emitting diodes. J Lumin 137:198–203
Sun W, Zhang Q, Qin L, Cheng Y, Xie Z, Lu C, Wang L (2010) Phosphorescent cuprous complexes with N,O ligands–synthesis, photoluminescence, and electro- luminescence. Eur J Inorg Chem:4009–4017
Lavie.-Cambot A, Cantuel M, Leydet Y, Jonusauskas G, Bassani DM, McClenaghana ND (2008) Improving the photophysical properties of copper(I) bis(phenanthroline) complexes. Coord Chem Rev 252:2572–2584
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Materials
Rights and permissions
About this article
Cite this article
Chavan, S.S., Pawal, S.B., More, M.S. et al. Copper(I) Complexes of N-(2-{[(2E)-2-(4-Nitrobenzylidenyl)Hydrazinyl]Carbonyl}Phenyl)Benzamide and Triphenylphosphine: Synthesis, Characterization and Luminescence Properties. J Fluoresc 26, 2033–2040 (2016). https://doi.org/10.1007/s10895-016-1897-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1897-x