Abstract
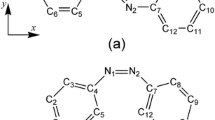
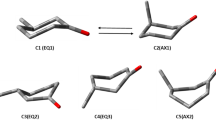
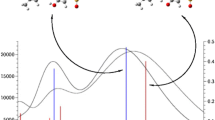
The results from the electrooptical absorption measurements (EOAM) on the equilibrium ground and excited Franck-Condon state dipole moments of Prodan and Laurdan in 1,4-dioxane are presented. As follows from experiments Prodan and Laurdan in the equilibrium ground and excited Franck-Condon state have two conformers with considerably different dipole moments. The electrical dipole moments and the transition dipole moment, obtained from the short-wavelength region of the absorption spectrum are parallel. The electrical dipole moments measured at the long-wavelength spectral region are parallel to each other but not parallel to the transition dipole moment m a. The angle θ between the transition dipole moment m a and the dipole moment in the equilibrium ground state μ g of the long-wavelength conformer is about 300 for both probes. Obtained results evidence that donor-acceptor pairs of the short-wavelength and long-wavelength conformers are not located on the same axis. Two low-energy conformers of Prodan have been found by density functional theory (DFT) calculations, differing in the orientation of the carbonyl group towards the naphthalene system.








Similar content being viewed by others
Abbreviations
- Laurdan:
-
6-decanoyl-2-dimethylamino-naphthalene
- Prodan:
-
6-propionyl-2-dimethylamino-naphthalene
- EOAM:
-
Electrooptical absorption measurements
- DFT:
-
Density functional theory
References
Moss GP (1996) Basic terminology of stereochemistry. Pure Appl Chem 68:2193–2222
Freidzon AY, Bagaturґyants AA, Gromov SP, Alfimov MV (2003) Recoordination of a metal ion in the cavity of a crown compound: a theoretical study 1. Conformers of arylazacrown ethers and their complexes with Ca2+ cation. Rus Cheml Bull, International Edition 52: 2646—2655
Baryshnikov GV, Minaev BF, Minaeva VA, Еgren H (2010) Theoretical study of the conformational structure and thermodynamic properties of 5-(oxo-1,3,thiazolidine 2- ylidene)-rhodanine-3-acetic acid as acceptor groups of indoline dyes. J Struct Chem 51:817–823
Al-Basheer W (2011) Solvent effects on IR modes of (R)-3-methylcyclopentanone conformers: a computational investigation. J Solut Chem 41:1495–1506. doi:10.1007/s10953-012-9890-8
Cruz-Cabeza AJ, Bernstein J (2014) Conformational polymorphism. Chem Rev 114:2170–2191
Upadhyayula S, Nunez V, Espinoza EM, Larsen JM, Bao D, Shi D, Mac JT, Anvaria B, Vullev VI (2015) Photoinduced dynamics of a cyanine dye: parallel pathways of non-radiative deactivation involving multiple excited-state twisted transients. Chem Sci 6:2237–2251
Lakowicz JR (1999) Principles of fluorescence spectroscopy. Second edition. Kluwer Academic/Plenum Publishers
Parasassi T, Ravagnan G, Rusch RM, Gratton E (1993) Modulation and dynamics of phase properties in phospholipid mixtores detected by Laurdan fluorescence. Photochem Photobiol 57:403–410
Zeng J, Chong PL (1995) Effect of ethanol-induced lipid interdigitation on the membrane solubility of Prodan, Acdan, and Laurdan. Biophys J 68:567–573. doi:10.1016/S0006-3495(95)80218-3
Pande AH, Qin S, Tatulian SA (2000) Membrane fluidity is a key modulator of membrane binding, insertion, and activity of 5-lipoxygenase. Biochem Biophys 88:4084–4094
Viard M, Gallay J, Vincent M, Paternostre M (2001) Origin of laurdan sensitivity to the vesicle-to-micelle transition of phospholipid-octylglucoside system: a time-resolved fluorescence study. Biophys J 80:347–359
Lissi EA, Abuin EB, Rubio MA, Ceron A (2000) Fluorescence of Prodan and laurdan in AOT/heptane/water microemulsions: partitioning of the probes and characterization of microenvironments. Langmuir 16:178–181
Kozyra KA, Heldt JR, Gondek G, Kwiek P, Heldt J (2004) Influence of DPPC liposome concentration on the fluorescence properties of Prodan and Laurdan. Z Naturforsch 59:809–818
De Vequi-Suplicy CC, Benatti CR, Lamy MT (2006) Laurdan in fluid bilayers: position and structural sensitivity. J Fluoresc 16(3):431–439
Marsh D (2009) Reaction fields in the environment of fluorescent probes: polarity profiles in membranes. Biophys J 96(7):2549–2558
Lucio AD, Vequi-Suplicy CC, Fernandez RM, Lamy MTD (2010) Laurdan spectrum decomposition as a tool for the analysis of surface bilayer structure and polarity: a study with DMPG, peptides and cholesterol. J Fluoresc 20:473–482
Ottavia J (2013) Lifetime and spectral phasors: exploiting Laurdan's fluorescence to characterize cell membranes. Dissertation, University of California
Rollinson AM, Drickamer HG (1980) High pressure study of luminescence from intramolecular CT compounds. J Chem Phys 73:5981–5996
Heisel F, Miehb JA, Szemik AW (1987) Experimental evidence of an intramolecular reaction in excited Prodan solution. Chem Phys Lett 138:321–326
Viard M, Gallay J, VincentmM MO, Robert B, Paternostre M (1997) Laurdan solvatochromism: solvent dielectric relaxation and intramolecular excited-state reaction. Biophys J 73:2221–2234
Parusel A (1998) Semiempirical studies of solvent effects on the intramolecular charge transfer of the fluorescence probe PRODAN. J Chem Soc Faraday Trans 94:2923–2927
Kozyra KA, Heldt JR, Heldt J, Engelke M, Diehl HA (2003) Concentration and temperature dependence of Laurdan fluorescence in glycerol. Z Naturforsch A 58:581–588
Tomin VI, Brozis M, Heldt J (2003) The red-edge effects in Laurdan solutions. Z Naturforsch 58A:109–117
Tomin VI, Hubisz K (2006) Inhomogeneous spectral broadening and the decay kinetics of the luminescence spectra of Prodan. Opt Spectrosc 101:98–104
Novaira M, Moyano F, BiasuttiMA SJJ, Correa NM (2008) An example of how to use AOT reverse micelle interfaces to control a photoinduced intramolecular charge-transfer process. Langmuir 24:4637–4646
Adhikary R, Barnes CA, Petrich JW (2009) Solvation dynamics of the fluorescent probe PRODAN in heterogeneous environments: contributions from the locally excited and charge-transferred states. J Phys Chem B 113:11999–12004
Everett RK, Nguyen AA, Abelt CJ (2010) Does PRODAN possess an O-TICTexcited state? Synthesis and properties of two constrained derivatives. J Phys Chem A 114(14):4946–4950
Pederzoli M, Sobek L, Brabec J, Kowalski K, Cwiklik L, Pittner J (2014) Fluorescence of PRODAN in water: a computational QM/MM MD study. Chem Phys Lett 597:57–62
Catalan J, Perez P, Laynez J, Blanco FG (1991) Analysis of the solvent effect on the photophysics properties of 6-propionyl-2-(dimethylamino)naphthalene (PRODAN). J Fluoresc 1:215–223
Samanta A, Fassenden RW (2000) Excited state dipole moment of PRODAN as determined from transient dielectric loss measurements. J Phys Chem A 104:8972–8975
Lobo BC, Abelt CJ (2003) Does PRODAN possess a planar or twisted charge-transfer excited state? Photophysical properties of two PRODAN derivatives. J Phys Chem A 107:10938–10943
Mennucci B, Caricato M, Ingrosso F, Cappelli C, Cammi R, Tomasi J, Scalmani G, Frisch MJ (2008) How the environment controls absorption and fluorescence spectra of PRODAN: a quantum mechanical study in homogeneous and heterogeneous media. J Phys Chem B 112(2):414–423
Vequi-Suplicy CC, Coutinho K, Lamy MT (2015) New insights on the fluorescent emission spectra of Prodan and Laurdan. J Fluoresc 25:621–629
Weber G, Farris FJ (1979) Synthesis and spectral properties of a hydrophobic fluorescent-probe-6-propionyl-2-(dimethylamino)naphthalene. Biochemistry 18(14):3075–3078
Balter A, Novak W, Pawelkiewicz W, Kowalczuk W (1988) Some remarks on the interpretation of the spectral properties of Prodan. Chem Phys Lett 143:565–570
Bunker CE, Bowen TL, Sun YP (1993) A photophysical study of solvatochromic probe 6-propionyl-2-(N,N-Dimethylamino)Naphthalene (Prodan) in solution. Photochem Photobiol 58(4):499–505
Kawski A (1999) Ground- and excited-state dipole moments of 6-propionyl-2-(N,N-Dimethylamino)naphthalene determined from solvatochromic shifts. Z Naturforsch 54a:379–381
Kawski A, Kuklinski B, Bojarski P (2000) Thermochromic shifts of absorption and fluorescence spectra and excited state dipole moment of PRODAN. Z Naturforsch 55a:550–554
Kawski A, Kuklinski B, Bojarski P, Diehl (2000) Ground and excited state dipole moments of LAURDAN determined from solvatochromic and thermochromic shifts of absorption and fluorescence spectra. Z Naturforsch 55a:817–822
Bacalum M, Zorila B, Radu M, Popescu A (2013) Laurdan solvatochromism: influence of solvent polarity and hydrogen bonds. Optoelectronics and Advanced Materials – Rapid Communications 7:456–460
Vequi-Suplicy CC, Coutinho K, Lamy MT (2014) Electric dipole moments of the fluorescent probes Prodan and Laurdan: experimental and theoretical evaluations. Biophys Rev 6:63–74
Nemkovich NA, Baumann W (2007) Molecular stark-effect spectroscopy of Prodan and Laurdan in different solvents and electric dipole moments in their equilibrated ground and Franck–Condon excited state. J Photochem Photobiol A Chem 185:26–31
Peteanu L, Locknar S (1997) Electrooptical absorption measurements and ab initio calculations of the dipolar properties of 2-(2′-hydroxyphenyl)-benzothiazole and -benzoxazole: two photostabilizers that undergo excited-state proton. Chem Phys Lett 296:521–529
Premvardhan LL, Peteanu LA (1999) Dipolar properties of and temperature effects on the electronic states of 3-hydroxyflavone (3HF) determined using stark-effect spectroscopy and compared to electronic structure calculations. J Phys Chem A 103:7506–7514
Nemkovich NA, Baumann W, Reis H, Detzer N (1995) Dipole moments of aminophtalimides determined by modified electro-optical absorption and emission measurements. J Photochem Photobiol A Chem 89:127–133
Nemkovich NA, Baumann W, Reis H (1997) Electro-optical and laser spectrofluorimetry study of coumarins 7 and 30: evidence for the existence of the close-lying electronic states and conformers. J Photochem Photobiol A Chem 109:287–292
Nemkovich NA, Baumann W, Kruchenok JV, Reis H, Rubinov AN (1997) Dipole moments of phenylnaphthylamine fluorescence probes and study of dielectric interactions in human erythrocyte ghosts. J Fluoresc 7:363–370
Nemkovich NA, Kruchenok JV, Rubinov AN, Pivovarenko VG, Baumann W (2001) Site selectivity in excited-state intramolecular proton transfer in flavonols. J Photochem Photobiol A Chem 139:53–62
Nemkovich NA, Baumann W, Pivovarenko VG (2002) Dipole moments of 4_-aminoflavonols using electro-optical absorption measurements or molecular stark-effect spectroscopy. J Photochem Photobiol A Chem 153:19–24
Nemkovich NA, Pivovarenko VG, Sobchuk AN, Baumann W, Rubinov AN (2006) Ultrafast time-resolved fluorescence spectroscopy of novel ketocyanine dyes. Opt Spectrosc 100:608–614
Nemkovich NA, Kruchenok JV, Sobchuk AN, Detert WN, Chernyavskii EA (2009) Subnanosecond spectrofluorimetry of new indolocarbazole derivatives in solutions and protein complexes and their dipole moments. Opt Spectrosc 107:292–299
Nemkovich NA, Detert H, Schmitt V (2010) Localized excitation effect on dipole moments of oligophenylenevinylenes in their excited Franck–Condon state. Chem Phys 378:37–41
Nemkovich NA, Detert H, Sobchuk AN (2012) Localized excitation effect in [(E,E)-2,5-bis-(4-N,N-dipropylaminophenyl)ethylenyl]-3,6-dimethylpyrazine and generation of its different forms in solutions. J Appl Spectrosc 78:787–793
Nemkovich NA, Detert H, Schmitt V, Kruchenok JV (2012) Influence of local excitation of octupolar oligophenylenvinylenes on their dipole moments. J Appl Spectrosc 79:353–359
Nemkovich NA, Baumann W, Kruchenok JV, Kurilo GI, Pivovarenko VG, Rubinov AN (2011) Molecular stark effect spectroscopy of diflavonols and inhomogeneous broadening of its electronic spectra in erythrocyte membranes. Opt Spectrosc 110:541–549
Liptay W (1974) Dipole moments and polarizabilities of molecules in excited electronic states. In: Lim ES (ed) Excited states, vol V.1. Academic Press, New York, pp. 129–205
Baumann W (1989) Determination of dipole moments in ground and excited state. In: Rossiter BW, Hamilton JF (eds) Physical methods of chemistry, vol V.3b. Wiley, New York, pp. 45–131
Rettig W, Baumann W (1992) In: Ralek JF (ed) Progress in photochemistry and photophysics, vol 6. CRC Press, Boca Raton, pp. 79–134
Neese F (2012) The ORCA program system. WIREs Comput Mol Sci 2:73–78
Neese F, Wennmohs F, Hansen A, Becker U (2009) Efficient, approximate and parallel Hartree–Fock and hybrid DFT calculations. A ‘chain-of-spheres’ algorithm for the Hartree–Fock exchange. Chem Phys 356:98–109
Izsák R, Neese F (2011) An overlap fitted chain of spheres exchange method. J Chem Phys 135:144105
Becke AD (1993) Density-functional thermochemistry. III The role of exact exchange. J Chem Phys 98:5648–5652
Schäfer A, Horn H, Ahlrichs R (1992) Fully optimized contracted Gaussian basis sets for atoms Li to Kr. J Chem Phys 97:2571–2577
Schäfer A, Huber C, Ahlrichs R (1994) Fully optimized contracted Gaussian basis sets of triple zeta valence quality for atoms Li to Kr. J Chem Phys 100:5829–5835
Weigend F, Ahlrichs R (2005) Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys Chem Chem Phys 7:3297–3305
Brozis M, Tomin VI, Heldt J (2002) Electron energies, dipole moments, and distribution of the Laurdan molecules over the conformational states of methyl groups. J Appl Spectrosc 69:678–682
Sobolewski AL, Domcke W (1996) Charge transfer in aminobenzonitriles: do they twist? Chem Phys Lett 250:428–436
Sobolewski AL, Domcke W (1996) Promotion of intramolecular charge transfer in dimethylamino derivatives: twisting versus acceptor-group rehybridization. Chem Phys Lett 259:119–127
Zachariasse KA (2000) Comment on “Pseudo-Jahn–Teller and TICT-models: a photophysical comparison of meta-and para-DMABN derivatives” [Chem. Phys. Lett. 305 1999 8] The PICT model for dual fluorescence of aminobenzonitriles. Chem Phys Lett 320:8–13
Grabowski ZR, Rotkiewicz K, Rettig W (2003) Structural changes accompanying intramolecular electron transfer: focus on twisted intramolecular charge-transfer states and structures. Chem Rev 103:3899–4031
Georgieva I, Aquino AJA, Plasser F, Trendafilova N, Kohn A, Lischka H (2015) Intramolecular charge-transfer excited-state processes in 4-(N,N-dimethylamino)benzonitrile: The role of twisting and the πσ* state. J Phys Chem A 119:6232–6243
Lewis FD, Holman B (1980) Singlet states of benzonitrile and p-dimethylaminobenzonitrile. J Phys Chem 84:2326–2328
Acknowledgments
N.N.A. thanks German Academic Exchange Service (DAAD) for the scholarship.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nemkovich, N.A., Detert, H. & Roeder, N. Electrooptical Absorption Measurements (EOAM) Testify Existence of two Conformers of Prodan and Laurdan with Different Dipole Moments in Equilibrium Ground and Franck-Condon Excited State. J Fluoresc 26, 1563–1572 (2016). https://doi.org/10.1007/s10895-016-1809-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1809-0