Abstract
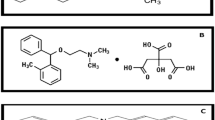
Sensitive and simple spectrophotometric (Method I) and spectrofluorimetric (Method II) methods were developed and validated for the determination of oxybutynin HCl (OXB) in its dosage forms. The method was based on the reaction of OXB with malonic acid anhydride in acetic acid anhydride to form a highly yellow colored product that was measured at 375 nm spectrophotometrically. The same reaction product exihibits strong fluorescence that was measured at 440 nm after excitation at 390 nm. The factors affecting formation and stability of the reaction product were carefully studied and optimized, and the reaction mechanism was postulated. The absorbance-concentration plot is rectilinear over the range 4–40 μg/mL with LOD of 1.12 μg/mL and LOQ of 3.39 μg/mL. The fluorescence-concentration plot is rectilinear over the range 0.5–6 μg/mL with LOD of 0.11 μg/mL and LOQ of 0.33 μg/mL. The method was applied to the analysis of commercial tablets Detronin® and Uripan®. Statistical comparison of the results with those of the reference method revealed good agreement and proved that there were no significant difference in the accuracy and precision between the two methods respectively. The study was extended to content uniformity testing.







Similar content being viewed by others
References
The British Pharmacopoeia (2009) London, The Stationery Office. Electronic version
Sweetman S (2009) Martindale (The Complete Drug Reference). London, The Pharmaceutical Press. Electronic version
Wagieh NE, Hegazy MA, Abdelkawy M, Abdelaleem EA (2010) Quantitative determination of oxybutynin hydrochloride by spectrophotometry, chemometry and HPTLC in presence of its degradation product and additives in different pharmaceutical dosage forms. Talanta 80:2007–2015
Ramadan NK, Mohamed HM, El Laithy MM (2007) Different methods for the determination of oxybutynin hydrochloride. Bull Fac Pharm (Cairo Univ) 31:40–45
Michelitsch A, Likussar W, Schubert-Zsilavecz M (1994) Determination of oxybutynin hydrochloride by differential pulse polarography. Monatsh Chem 125:1183–1187
da Fonseca P, de Freitas LAP, Pinto LFR, Pestana CR, Bonato PS (2008) Enantioselective analysis of oxybutynin and N-desethyloxybutynin with application to an in vitro biotransformation study. J Chromatogr B 875:161–167
Guo N, Gao X, Xu G, Guo X (2008) High performance liquid chromatographic separation of oxybutynin enantiomers using chiral mobile phase additive. Sepu 26:259–261
Ding X, Gao S, Cao Q, Miao C, Zhong Y, Yu Y, Gao J (2003) Reversed-phase high performance liquid chromatographic determination of oxybutynin hydrochloride in its tablets. Dier Junyi Daxue Xuebao 24:327–329
Thomas AD (1976) Spectrophotometric determination of some drugs containing a tertiary amine group. J Pharm Pharmacol 28:838–839
Thomas AD (1975) Spectrofluorimetric determination of some alkaloids containing a tertiary amino group. Talanta 22:865–869
Mokhtar MM (1998) Spectrofluorimetric determination of acyclovir and praziquantel in spiked human plasma and dosage forms. Alex J Pharm Sci 12:1–5
Al-Majed AA, Al-Zehouri J, Belal F (2000) Use of mixed anhydrides for the determination of terfenadine in dosage forms and spiked human plasma. J Pharm Biomed Anal 23:281–289
Abdellatef HE, El-Henawee MM, El-Sayed HM, Ayad MM (2006) Spectrophotometric and spectrofluorimetric methods for analysis of tramadol, acebutolol and dothiepin in pharmaceutical preparations. Spectrochim Acta Part A 65:1087–1092
Aly MT, El-Shabouri SR, Rageh AI (1980) Improved spectrophotometric determination of tertiary amine using malonic acid / acetic anhydride reagent. J Pharm Sci 21:363–372
Ian RCW, Paul JW (1987) Spectrofluorometric flow injection determination of tertiary amines in non aqueous media. Anal Chim Acta 192:77–83
(2007) The United States Pharmacopoeia 30, the National Formulary 25, US Pharmacopeial Convention: Rockville, MD. Electronic version
ICH Harmonized Tripartite Guideline, Validation of Analytical Procedures: Text and Methodology, Q2(R1), Current Step 4 Version, Parent Guidelines on Methodology Dated November 6; 1996, Incorporated in November 2005. Through: (http://www.ich.org/LOB/media/MEDIA417.pdf). (accessed June 9, 2010)
Miller JN, Miller JC (2005) Statistics and chemometrics for analytical chemistry, 5th edn. Prentice Hall, England, p 256
Srikanth K, Emmanuel KA, Raju KR (2010) Spectrophotometric determination of oxybutynin through ion-association complex formation. RASĀYAN J. Chem. 3: 179–187. through www.rasayanjournal.com
Rose J (1964) Advanced physicochemical experiments. Pitman, London, p 67
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walash, M.I., Belal, F., El-Enany, N. et al. Determination of Oxybutynin in Pharmaceuticals via Reaction with Mixed Acids Anhydrides: Application to Content Uniformity Testing. J Fluoresc 21, 715–722 (2011). https://doi.org/10.1007/s10895-010-0761-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-010-0761-7