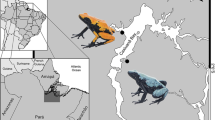
Abstract
Many chemically-defended/aposematic species rely on diet for sequestering the toxins with which they defend themselves. This dietary acquisition can lead to variable chemical defenses across space, as the community composition of chemical sources is likely to vary across the range of (an aposematic) species. We characterized the alkaloid content of two populations of the Dyeing Poison Frog (Dendrobates tinctorius) in northeastern French Guiana. Additionally, we conducted unpalatability experiments with naive predators, Blue Tits (Cyanistes caeruleus), using whole-skin secretion cocktails to assess how a model predator would respond to the defense of individuals from each population. While there was some overlap between the two D. tinctorius populations in terms of alkaloid content, our analysis revealed that these two populations are markedly distinct in terms of overall alkaloid profiles. Predator responses to skin secretions differed between the populations. We identified 15 candidate alkaloids (including three previously undescribed) in seven classes that are correlated with predator response in one frog population. We describe alkaloid profile differences between populations for D. tinctorius and provide a novel method for assessing unpalatability of skin secretions and identifying which toxins may contribute to the predator response. In one population, our results suggest 15 alkaloids that are implicated in predator aversive response. This method is the first step in identifying the causal link between alkaloids and behavioral responses of predators, and thus makes sense of how varying alkaloid combinations are capable of eliciting consistent behavioral responses, and eventually driving evolutionary change in aposematic characters (or characteristics).

Similar content being viewed by others
Data Availability
Data are available at Lawrence et al. 2019b.
References
Alvarado JB, Alvarez A, Saporito RA (2013) Oophaga pumilio (strawberry poison frog). Predation Herpetol Rev 44:298
Amézquita A, Ramos Ó, González MC, Rodríguez C, Medina I, Simões PI, Lima AP (2017) Conspicuousness color resemblance and toxicity in geographically diverging mimicry: The pan-Amazonian frog Allobates femoralis. Evolution 71(4):1039–1050. https://doi.org/10.1111/evo.2017.71.issue-410.1111/evo.13170
Blount JD, Rowland HM, Drijfhout FP et al (2012) How the ladybird got its spots: Effects of resource limitation on the honesty of aposematic signals. Funct Ecol 26:334–342. https://doi.org/10.1111/j.1365-2435.2012.01961.x
Bolton SK, Dickerson K, Saporito RA (2017) Variable alkaloid defenses in the Dendrobatid poison Frog Oophaga pumilio are perceived as differences in palatability to Arthropods. J Chem Ecol 43:273–289. https://doi.org/10.1007/s10886-017-0827-y
Brower LP, Van Brower J, Corvino JM (1967) Plant poisons in a terrestrial food chain. Proceedings of the National Academy of Sciences 57:893–898
Burdfield-Steel E, Brain M, Rojas B, Mappes J (2018) The price of safety: food deprivation in early life influences the efficacy of chemical defence in an aposematic moth. Oikos 128(2):245–253. https://doi.org/10.1111/oik.2019.v128.i210.1111/oik.05420
Burdfield-Steel E, Brain M, Rojas B, Mappes J (2019) The price of safety: food deprivation in early life influences the efficacy of chemical defence in an aposematic moth. Oikos 128:245–253. https://doi.org/10.1111/oik.05420
Chouteau M, Angers B (2011) The role of predators in maintaining the geographic organization of aposematic signals. Am Nat 178:810–817. https://doi.org/10.1086/662667
Comeault AA, Noonan BP (2011) Spatial variation in the fitness of divergent aposematic phenotypes of the poison frog, Dendrobates tinctorius. J Evol Biol 24:1374–1379. https://doi.org/10.1111/j.1420-9101.2011.02258.x
Daly JW, Garraffo HM, Spande TF et al (2008) Individual and geographic variation of skin alkaloids in three swamp-forest species of madagascan poison frogs (Mantella). J Chem Ecol 34:252–279. https://doi.org/10.1007/s10886-015-0616-4
Daly JW, Myers CW (1967) Toxicity of Panamanian poison frogs (Dendrobates): some biological and chemical aspects. Science 156:970–973
Daly JW, Myers CW, Whittaker N (1987) Further classification of skin alkaloids from neotropical poison frogs (Dendrobatidae), with a general survey of toxic/noxious substances in the Amphibia. Toxicon 25:1023–1095
Daly JW, Spande TF, Garraffo HM (2005) Alkaloids from amphibian skin: a tabulation of over eight-hundred compounds. J Nat Prod 68:1556–1575
Darst CR, Cummings ME (2006) Predator learning favours mimicry of a less-toxic model in poison frogs. Nature 440:208–211
Darst CR, Menendez-Guerrero PA, Coloma LA, Cannatella DC (2005) Evolution of dietary specialization and chemical defense in poison frogs (Dendrobatidae): a comparative analysis. Am Nat 165:56–69
Darst CR, Cummings ME, Cannatella DC (2006) A mechanism for diversity in warning signals: Conspicuousness versus toxicity in poison frogs. Proceedings of the National Academy of Sciences 103(15):5852–5857. https://doi.org/10.1073/pnas.0600625103
Holen OH (2013) Disentangling taste and toxicity in aposematic prey. Proc Biol Sci 280:20122588. https://doi.org/10.1098/rspb.2012.2588
Hovey KJ, Seiter EM, Johnson EE, Saporito RA (2018) Sequestered alkaloid defenses in the dendrobatid poison frog Oophaga pumilio provide variable protection from microbial pathogens. J Chem Ecol 44:312–325
Lawrence JP, Rojas B, Fouquet A et al (2019a) Weak warning signals can persist in the absence of gene flow. Proc Natl Acad Sci 116:19037–19045. https://doi.org/10.1073/pnas.1901872116
Lawrence JP, Rojas B, Fouquet A et al (2019b) Original data for article: Weak warning signals can persist in the absence of gene flow - JYX. University of Jyväskylä. https://doi.org/10.17011/jyx/dataset/65263
Lindström L, Alvtalo RV, Mappes J et al (1999) Can aposematic signals evolve by gradual change? Nature 397:249–251
Maan ME, Cummings ME (2012) Poison frog colors are honest signals of toxicity, particularly for bird predators. Am Nat 179:E1–E14. https://doi.org/10.1086/663197
Master TL (1999) Predation by Rufous Motmot on black-and-green poison dart frog. Wilson Bull 111:439–440
McGugan JR, Byrd GD, Roland AB et al (2016) Ant and mite diversity drives toxin variation in the little devil poison frog. J Chem Ecol 42:537–551. https://doi.org/10.1007/s10886-016-0715-x
Murray EM, Bolton SK, Berg T, Saporito RA (2016) Arthropod predation in a dendrobatid poison frog: does frog life stage matter? Zoology 119:169–174. https://doi.org/10.1016/j.zool.2016.01.002
Nishida R (2002) Sequestration of defensive substances from plants by Lepidoptera. Annu Rev Entomol 47:57–92. https://doi.org/10.1146/annurev.ento.47.091201.145121
Nokelainen O, Hegna RH, Reudler JH, Lindstedt C, Mappes J (2012) Trade-off between warning signal efficacy and mating success in the wood tiger moth. Proc R Soc B 279:257–265
Noonan BP, Gaucher P (2006) Refugial isolation and secondary contact in the dyeing poison frog Dendrobates tinctorius. Mol Ecol 15:4425–4435. https://doi.org/10.1111/j.1365-294X.2006.03074.x
Ojala K, Julkunen-Tiitto R, Lindström L, Mappes J (2005) Diet affects the immune defence and life-history traits of an arctiid moth Parasemia plantaginis Evol Ecol Res 7:1153–1170
Ottocento C, Winters AE, Rojas B, Mappes J, Burdfield-Steel E (2022) Not just the sum of its parts: Geographic variation and nonadditive effects of pyrazines in the chemical defence of an aposematic moth. J Evol Biol. https://doi.org/10.1111/jeb.14142
Paluh DJ, Hantak MM, Saporito RA (2014) A test of aposematism in the dendrobatid poison frog Oophaga pumilio: the importance of movement in clay model experiments. J Herpetol 48:249–254. https://doi.org/10.1670/13-027
Poulton, EB (1890) The colours of animals: their meaning and use, especially considered in the case of insects, vol 67. D. Appleton, New York
Prates I, Paz A, Brown JL, Carnaval AC (2019) Links between prey assemblages and poison frog toxins: a landscape ecology approach to assess how biotic interactions affect species phenotypes. 14317–14329. https://doi.org/10.1002/ece3.5867
Proksch P (1994) Defensive roles for secondary metabolites from marine sponges and sponge-feeding nudibranchs. Toxicon 32:639–655. https://doi.org/10.1016/0041-0101(94)90334-4
Protti-Sánchez F, Quirós-Guerrero L, Vásquez V et al (2019) Toxicity and alkaloid profiling of the skin of the Golfo Dulcean poison frog Phyllobates vittatus (Dendrobatidae). J Chem Ecol 45:914–925
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Rojas B (2017) Behavioural ecological and evolutionary aspects of diversity in frog colour patterns. Biol Rev 92(2):1059–1080. https://doi.org/10.1111/brv.12269
Rojas B, Burdfield-Steel E, Pakkanen H et al (2017) How to fight multiple enemies: target-specific chemical defences in an aposematic moth. Proc R Soc B-Biol Sci 284:20171424
Rojas B, Endler JA (2013) Sexual dimorphism and intra-populational colour pattern variation in the aposematic frog Dendrobates tinctorius Evol Ecol 27:739–753. https://doi.org/10.1007/s10682-013-9640-4
Rojas B, Mappes J, Burdfield-Steel E (2019) Multiple modalities in insect warning displays have additive effects against wild avian predators. Behav Ecol Sociobiol 73:1–12
Rojas B, Rautiala P, Mappes J (2014) Differential detectability of polymorphic warning signals under varying light environments. Behav Process 109:164–172. https://doi.org/10.1016/j.beproc.2014.08.014
Rowland HM, Speed MP, Ruxton GD et al (2007) Countershading enhances cryptic protection: an experiment with wild birds and artificial prey. Anim Behav 74:1249–1258. https://doi.org/10.1016/j.anbehav.2007.01.030
Sandre SL, Tammaru T, Esperk T et al (2007) Carotenoid-based colour polyphenism in a moth species: search for fitness correlates. Entomol Exp Appl 124:269–277. https://doi.org/10.1111/j.1570-7458.2007.00579.x
Santos JC, Coloma LA, Cannatella DC (2003) Multiple, recurring origins of aposematism and diet specialization in poison frogs. Proc Natl Acad Sci U S A 100:12792–12797. https://doi.org/10.1073/pnas.2133521100
Saporito RA, Donnelly MA, Garraffo MA et al (2006) Geographic and seasonal variation in alkaloid-based chemical defenses of Dendrobates pumilio from Bocas del Toro, Panama. J Chem Ecol 32:795–814
Saporito RA, Donnelly MA, Hoffman RL et al (2003) A siphonotid millipede (Rhinotus) as the source of spiropyrrolizidine oximes of dendrobatid frogs. J Chem Ecol 29:2781–2786
Saporito RA, Donnelly MA, Jain P et al (2007a) Spatial and temporal patterns of alkaloid variation in the poison frog Oophaga pumilio in Costa Rica and Panama over 30 years. Toxicon 50:757–778
Saporito RA, Donnelly MA, Norton RA et al (2007b) Oribatid mites as a major dietary source for alkaloids in poison frogs. Proc Natl Acad Sci U S A 104:8885–8890
Saporito RA, Donnelly MA, Madden A et al (2010) Sex-related differences in alkaloid chemical defenses of the dendrobatid frog Oophaga pumilio from Cayo Nancy, Bocas del Toro, Panama. J Nat Prod 73:317–321. https://doi.org/10.1021/np900702d
Saporito RA, Donnelly MA, Spande TF, Garraffo HM (2011) A review of chemical ecology in poison frogs. Chemoecology 22:159–168. https://doi.org/10.1007/s00049-011-0088-0
Saporito RA, Grant T (2018) Comment on Amézquita (2017) Conspicuousness, color resemblance, and toxicity in geographically diverging mimicry: The pan-Amazonian frog Allobates femoralis. Evolution 1009–1014. https://doi.org/10.1111/evo.13468
Siddiqi A, Cronin TW, Loew ER et al (2004) Interspecific and intraspecific views of color signals in the strawberry poison frog Dendrobates pumilio. J Exp Biol 207:2471–2485
Skelhorn J, Rowe C (2009) Distastefulness as an antipredator defence strategy. Anim Behav 78:761–766
Speed MP, Ruxton GD, Blount JD, Stephens PA (2010) Diversification of honest signals in a predator-prey system. Ecol Lett 13:744–753. https://doi.org/10.1111/j.1461-0248.2010.01469.x
Speed MP, Ruxton GD, Broom M (2006) Automimicry and the evolution of discrete prey defences. Biol J Linn Soc 87:393–402. https://doi.org/10.1111/j.1095-8312.2006.00577.x
Speed MP, Ruxton GD, Mappes J, Sherratt TN (2012) Why are defensive toxins so variable? An evolutionary perspective. Biol Rev 87:874–884. https://doi.org/10.1111/j.1469-185X.2012.00228.x
Summers K, Clough ME (2001) The evolution of coloration and toxicity in the poison frog family (Dendrobatidae). Proc Natl Acad Sci U S A 98:6227–6232. https://doi.org/10.1073/pnas.101134898
Summers K, Speed MP, Blount JD, Stuckert AMM (2015) Are aposematic signals honest? A review. J Evol Biol 28:1583–1599. https://doi.org/10.1111/jeb.12676
Vacher J-P, Chave J, Ficetola FG et al (2020) Large-scale DNA-based survey of frogs in Amazonia suggests a vast underestimation of species richness and endemism. J Biogeogr 47:1781–1791. https://doi.org/10.1111/jbi.13847
Vidal-Cordero JM, Moreno-Rueda G, López-Orta A et al (2012) Brighter-colored paper wasps (Polistes dominula) have larger poison glands. Front Zool 9:1–5
Wang IJ (2011) Inversely related aposematic traits: reduced conspicuousness evolves with increased toxicity in a polymorphic poison-dart frog. Evolution 65:1637–1649. https://doi.org/10.1111/j.1558-5646.2011.01257.x
Wang K, Zhao H (2015) Birds generally carry a small repertoire of bitter taste receptor genes. Genome Biol Evol 7:2705–2715. https://doi.org/10.1093/gbe/evv180
Weldon PJ (2017) Poison frogs, defensive alkaloids, and sleepless mice: critique of a toxicity bioassay. Chemoecology 27:123–126. https://doi.org/10.1007/s00049-017-0238-0
Wollenberg KC, Lötters S, Mora-Ferrer C, Veith M (2008) Disentangling composite colour patterns in a poison frog species. Biol J Linn Soc 93:433–444
Zvereva EL, Kozlov MV (2015) The costs and effectiveness of chemical defenses in herbivorous insects: a meta-analysis. Ecol Monogr 86:107–124
Acknowledgements
We highly value equity, diversity, and inclusion in science (EDI) and thus took into account EDI best practice. Our team includes researchers from (4) different countries (United States, Colombia, Finland, and France), and diverse backgrounds, and career stages, all of which significantly contributed to the fulfillment and quality of our study. The data used in this study can be found in Lawrence, et al. 2019b. Blue tit experiments were conducted under the Central Finland Centre for Economic Development, Transport, and Environment and license from the National Animal Experiment Board (ESAVI/9114/04.10.07/2014), and the Central Finland Regional Environment Centre (VARELY/294/2015), respectively. Animals were exported under CITES permit FR1397300015-E. This study was supported in part by an Investissement d’Avenir Grant (CEBA ANR-10-LABX-25-01) of the Agence Nationale de la Recherche; the American Society of Ichthyologists and Herpetologists’ Gaige Award (to JPL); and a Society for the Study of Amphibians and Reptiles’ grant in Herpetology (to JPL). BR and JM were funded by the Finnish Centre of Excellence in Biological Interactions (Project 28466, to JM.). BR also acknowledges funding from the Academy of Finland (Academy Research Fellowship, No. 318404).
Funding
This study benefited from an “Investissement d’Avenir” grant managed by the Agence Nationale de la Recherche (CEBA, ref. ANR-10- LABX-25-01. B.R. and J.M were funded by the Finnish Centre of Excellence in Biological Interactions (Project 28466, to J.M.). B.R. is supported by funding from the Academy of Finland (Academy Research Fellowship No. 318404).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material collection, data preparation, and analysis were performed by J.P.L., B.R., J.M., A.B., and R.S. Samples were collected by A.F. and B.R. J.P.L. and B.N. wrote the first draft of the manuscript, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Conflict of Interest
The authors declare no conflicts of interest in relation to this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lawrence, J.P., Rojas, B., Blanchette, A. et al. Linking Predator Responses to Alkaloid Variability in Poison Frogs. J Chem Ecol 49, 195–204 (2023). https://doi.org/10.1007/s10886-023-01412-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-023-01412-7