Abstract
The detection of dung odors is a crucial step in the food-searching behavior of dung beetles (Coleoptera: Scarabaeoidea). Yet, whether certain compounds characteristic of a given dung type contribute to a ‘choosy generalism’ behavior proposed for this taxonomic group is unknown. To address this, we analyzed the chemical composition of three types of dung (cow, horse, and rabbit) and conducted behavioral and electroantennogram (EAG) bioassays on 15 species of dung beetles using 19 volatile organic compounds representing the three dung samples. Chemical analyses revealed substantial qualitative and quantitative differences among dung types. When offered these food options in an olfactometer, 14 species exhibited a feeding preference. Surprisingly, all 19 compounds used in the EAG assays elicited antennal responses, with species displaying different olfactory profiles. The relationship between behavioral preferences and electrophysiological profiles highlighted that species with different food preferences had differences in antennal responses. Moreover, a specific set of EAG-active compounds (nonanal, sabinene, acetophenone, ρ-cresol, 2-heptanone, 1H-indole, and 6-methyl-5-hepten-2-one) were the strongest drivers in the distinct sensory profiles of the trophic preference groups. Our results point to the importance of the whole bouquet of dung-emanating compounds in driving food-searching behavior, but specific volatiles could aid in determining highly marked trophic preferences in certain species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A relevant distinction between animal consumers is whether they are specialized (monophagous and oligophagous) or generalized (polyphagous) in their diet. From a broad perspective, dung beetles could be classified as oligophagous if we consider that they are mainly coprophagous, a derived feeding habit associated with their filtering mouthparts adaptation, which arose from saprophagy in the Cretaceous, leading to a species radiation during mammal diversification in the Cenozoic (Ahrens et al. 2014; Gunter et al. 2016). However, some dung beetles a priori considered coprophagous, show a more generalist food selection, being able to exploit other types of resources, such as vertebrate and invertebrate carrion, fungi, and different fruits (Giménez Gómez et al. 2021; Halffter and Matthews 1966; Hanski and Cambefort 1991; Verdú et al. 2007; Weithmann et al. 2020). Within coprophagous species, due to the scattered and ephemeral presence of excrement, dung beetles show an opportunistic and generalized use of a wide range of dung types (Frank et al. 2018a; Hanski and Cambefort 1991), with species preferring only one dung type being very rare (Carpaneto et al. 2010; Galante and Cartagena 1999; Larsen et al. 2006; Lumaret and Iborra 1996). These observations are in congruence with the hypothesis of ‘choosy generalism’ proposed for coprophagous dung beetles (Dormont et al. 2004, 2007; Frank et al. 2018b).
Feces of different herbivorous mammals differ in terms of chemical and physical characteristics (Holter 2016). For example, the droppings of horses and cows, monogastric and ruminant herbivores respectively, differ in terms of their content of nutrients, plant fiber, water, and volatile compounds (Dormont et al. 2010; Frank et al. 2017, 2018b; Holter 2016; Stavert et al. 2014). At the extreme, certain excrement such as rabbit dung, which, due to its low nutritional and water content, is more similar to litter than to typical herbivore dung, e.g., horse or cow (Verdú and Galante 2004). This heterogeneity in chemical composition among different excrements of herbivorous mammals (Holter 2016; Nibaruta et al. 1980) could be a key factor in explaining the existence of feeding preferences in dung beetles.
Although there is evidence of preferences of dung beetles for certain dung types (Carpaneto et al. 2010; Dormont et al. 2004; Errouissi et al. 2004; Finn and Giller 2002; Frank et al. 2017; Giménez Gómez et al. 2021; Martín-Piera and Lobo 1996), there are few studies explaining the mechanisms that influence dung attraction through volatile organic compounds (VOCs). Dung beetles are attracted to dung by olfactory cues and their selection depends on the VOCs emitted by the different types of potential resource, the distance to the source, and the nutritional quality of each resource (Bogoni and Hernández 2014; Dormont et al. 2004, 2007, 2010; Hanski and Cambefort 1991; Holter and Scholtz 2007). Dung odors are complex mixtures, typically comprising between 50 and 400 VOCs (Aii et al. 1980; Amann et al. 2014; Dormont et al. 2010; Frank et al. 2018b). While some VOCs are common to all dung types, others appear to be characteristic of particular types (Dormont et al. 2010; Frank et al. 2018b). For example, common dung-emitted VOCs such as ρ-cresol, 1H-indole, and skatole (Dormont et al. 2007; Inouchi et al. 1988; Stavert et al. 2014) could serve as general cues for the presence of a food source. More specifically, ρ-cresol is an abundant VOC in cattle dung (Aii et al. 1980; Dormont et al. 2010; Frank et al. 2018b), several omnivore dung types (Blanes-Vidal et al. 2009; Stavert et al. 2014; Walton et al. 2013), as well as in a variety of domesticated and non-domesticated animal fecal samples (Apps et al. 2012; Martín et al. 2010; Terada et al. 1992). Skatole has been identified in various studies with pigs (Hobbs et al. 1996; Koziel et al. 2005), chickens (Cai et al. 2007), and various other vertebrates (Dehnhard et al. 1991). Dung beetles could use complex mixtures of VOCs as signals to detect and select preferred dung types by processing a few ‘key’ VOCs in odor (Stavert et al. 2014; Wurmitzer et al. 2017). Some studies suggest that dung beetles prefer complex mixtures rather than single compounds (Frank et al. 2018b; Wurmitzer et al. 2017). Dung beetles can be attracted in the field with a blend of VOCs, such as 2-butanone, butyric acid, 1H-indole, and skatole, or with butyric acid (Wurmitzer et al. 2017), or a blend of 1H-indole, skatole, phenol, butyric acid, 2-butanone, and ρ-cresol (Frank et al. 2018b), suggesting that mixtures are much more effective than single compounds alone. Despite information obtained in field and laboratory studies on attraction to certain mixtures of dung volatiles, electrophysiological studies on these compounds as possible semiochemicals are needed.
In insects, olfaction plays a key role in many aspects of life, including the search for food. The olfactory system of insects is remarkably sensitive, specific, and dynamic. Dung beetles have an acute olfactory sensitivity that allows them to locate ephemeral and often patchily distributed resources required for reproduction (Tribe and Burger 2011). Few studies have investigated the foraging behavior in dung beetles using electrophysiological techniques. Inouchi et al. (1988) studied the Japanese dung beetle, Geotrupes auratus, in which they demonstrated that single antennal olfactory cells were active to volatile compounds derived from dung, including 2-butanone, phenol, ρ-cresol, 1H-indole, and skatole. Likewise, in a study carried out with Anoplotrupes stercorosus using gas chromatography coupled with electroantennographic detection (GC–EAD), a large number of VOCs derived from carrion were active (Weithmann et al. 2020). Yet, the relationships between feeding preferences for a specific type of dung and the VOCs that elicit electrophysiological responses remain to be determined.
The objective of this study was to investigate, using a combination of behavioral and electrophysiological bioassays, whether specific VOCs characteristic of a type of dung may determine food preferences of dung beetle species. To do this, we first analyzed the characteristic VOCs of three types of excrement (cow, horse, and rabbit). Secondly, we analyzed the attraction and feeding preferences of a large number of dung beetle species, belonging to different taxonomic and functional groups, using olfactometer bioassays. Finally, we carried out electroantennogram (EAG) bioassays to determine VOCs that elicit responses in each species. We hypothesized that choosy generalism behavior proposed for coprophagous dung beetles may be related to the presence of a high number of VOCs that are physiologically active, and that the existence of food preferences might be explained by the presence of a few specific VOCs.
Methods and Materials
Dung Selection, Compositional Chemical Relations, and Candidate Semiochemicals. We selected three dung types for this study: cow, horse, and rabbit. These types differ in content of nutrients, fiber, water, and VOCs (Dormont et al. 2010; Frank et al. 2017, 2018b; Goodrich et al. 1981; Holter 2016; Stavert et al. 2014; Verdú and Galante 2004). Furthermore, cows are ruminant animals, horses are monogastric and rabbits are monogastric that use cecotrophy to maximize nutrient intake from their food. Odor samples of cow and horse dung from different individuals (n = 3) were collected in the field at Picos de Europa National Park (Principado de Asturias, Spain). Odor samples were collected immediately after defecation to avoid insect colonization and physical/chemical alteration of the dung. For rabbit dung, fresh samples were collected at the Sierra de la Carrasqueta and at the surroundings of the University of Alicante (Alicante, Comunidad Valenciana, Spain), from different rabbit latrines and were brought to the laboratory in individual plastic freezer bags (Ziploc, SC Johnson & Son, Racine, WI).
Volatile emissions from dung types were sampled using headspace sorptive extraction (HSSE) with Twisters® (stir bar, 0.5mm thick, 10mm long, polydimethylsiloxane coating, Gerstel GmbH & Co. KG, Mülheim an der Ruhr, Germany). The Twisters® were cleaned per manufacturer recommendations with acetonitrile (HPLC-grade) and conditioned at 250°C for 15h with a flow of 75 ml.min− 1 purified helium. For HSSE, the Twister® was fixed within the headspace volume by magnetic force using a neodymium disc magnet (Ø 5mm, height 3mm) placed inside a glass chamber. The dung sample was covered with the headspace glass chamber and HSSE extraction carried out by static sampling. Twisters® were exposed to headspace for 1h at 22°C (ambient temperature) for cow and horse dung samples, while rabbit dung was sampled in an incubator at 37°C for 24h. Extraction time was established in previous assays. A total of three replicates were performed for each dung type. After extraction, each stir bar was removed with tweezers and placed in a 2 ml vial to be transported to the laboratory, where it was thermally desorbed in a gas chromatograph/mass spectrometer (GC/MS).
Analysis was carried out using a 6890 Agilent GC system coupled to a Agilent 5973 inert quadrupole MS equipped with a thermo desorption system (TDS2) and a cryo-focusing CIS-4 PTV injector (Gerstel). Thermal desorption used a Gerstel TDS 2 (Gerstel GmbH & Co. KG, Mülheim an der Ruhr, Germany) at 300°C for 10min, with a helium flow of 55 ml.min− 1. The GC was fitted with a DB-5 capillary column (30m x 0.25mm I.D., 0.25μm film thickness), and used helium as carrier gas with a constant flow of 1.4 ml.min− 1. The initial oven temperature was set at 60°C for 5min, and increased by 5°C.min− 1 to 250°C, then held for 10min. The injector, in split mode, and the MS transfer line were set at 250 and 280°C respectively. The MS quadrupole and source were set at 150 and 250°C respectively. Mass spectra were taken in EI mode at 70eV with a scan range of 40–450 m/z and a scanning rate of 2.65 scans/s.
GC/MS data were processed using MSD ChemStation software (Agilent Technologies Inc., Santa Clara, CA, USA). Tentative compound identification was done by comparison of mass spectra in the WILEY and NIST mass spectral libraries. We calculated retention indices of VOCs using an alkane standard mixture (C7-C30 dissolved in hexane; Sigma-Aldrich Chemie GmBh, Steinheim, Germany) applying the method of Van den Dool and Kratz (1963), and compared these against literature values (Adams, 2007). Identifications were confirmed by comparison of spectra and retention times with those of authentic standards when available. Commercial standards were obtained from chemical suppliers (Sigma-Aldrich Chemie GmBh, Steinheim, Germany), with ≥ 98% purity, and were run under the same conditions as samples. Identified compounds were expressed as percentage of the total content of compounds (relative abundance).
Dung Beetle Species Selection. A total of 15 species of dung beetles was collected in different localities in Spain and France (see Table1). To broaden the response range, species belonging to three families (Aphodiidae, Geotrupidae, and Scarabaeidae) from 12 different genera were selected. We collected these species searching in various types of dung, including cow and horse pats and rabbit latrines (Table1). Specimens were placed in aerated plastic containers (38 × 32 × 15cm) with moist towel paper. The containers were then placed in a cooler at 20°C until they arrived at the laboratory. Separate terrariums were prepared for each species and placed in a climate chamber at 15 ± 1°C with 65% relative humidity (RH) and a photoperiod of 14:10h (light: dark). To standardize the condition of beetles, only mature specimens were selected according to external age-grading methods such as abrasion of the fore tibiae in conjunction with cuticle hardness of the pronotum and elytra, which makes it possible to sort out individuals of approximately the same age (Tyndale-Biscoe 1984). This work conforms to the Spanish legal requirements including those relating to conservation and welfare.
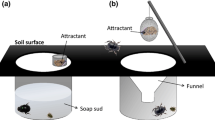
Food Preference using Olfactometer Tests. Behavioral bioassays were carried out in 2020 to test food preferences of adult beetles to different dung types (cow, horse, and rabbit). Tests used a four-arm olfactometer design based on that of Verdú et al. (2007). We used two designs, adapted to the different sizes of dung beetles. For species over 1cm in length, the olfactometer consisted of a central circular arena (60cm superior diameter and 40cm inferior diameter) with four 5cm-diam. holes to attach tubes (arms) of methacrylate (50cm length, 5cm o.d., and 4.75cm i.d.) placed horizontally. For species less than 1cm in length, the central circular arena was reduced to 30 × 20cm (superior and inferior diameter, respectively), and the length of the methacrylate tubes was 30cm each. There was a plastic container with test samples at the end of each arm to capture beetles that responded positively to the tested resources. The plastic containers were designed to permit the entrance and exit of beetles that responded to the tested resources. Air was passed through an activated carbon filter and drawn into the plastic containers of the olfactometer. In the center of the arena was a hole in the methacrylate roof to attach a tube that conducted air to a fume hood. Complete sealing of the system was ensured by Teflon® to join all connections. Outside light was blocked off by wrapping the transparent pieces of the olfactometer with aluminum foil.
In each olfactometer, the source of VOCs consisted of three fresh dung samples (15g each) placed in the different containers, and an empty container for a control. The arena was covered with sterile vermiculite. After placing beetles in an arena, we waited 10min before starting an experiment, to allow beetles to adapt to the conditions. Each bioassay consisted of 3–22 replicates that were run on different days using a group of 20 beetles per session, except for Ceratophyus hoffmannseggi and Ateuchetus cicatricosus (10 beetles per bioassay). The bioassays were conducted at 28 ± 3°C during the day (08:00–16:00h) or night (20:00–08:00h) for diurnal and crepuscular-nocturnal species, respectively. After each trial, the olfactometer was disassembled, and its arena, tubes, and containers washed with neutral dishwashing soap (5%) and disinfected with 70% ethanol (v/v). We measured the number of individuals attracted to each dung type for each replicate. Beetles that made no choice after this time were considered a null response. In all cases, each beetle was tested only once and treatments were randomly interchanged.
Electroantennography Bioassays. For EAGs, characteristic VOCs of each dung type, as well as compounds shared between two or more dung types, were selected (see Statistical Analyses, for details). Synthetic compounds were > 95% pure and purchased from commercial sources (Sigma-Aldrich Chemie GmBh, Steinheim, Germany). For EAG recordings, each compound was diluted to 1% in hexane (HPLC-grade) and stored at -20°C until needed. Immediately prior to an experiment, 1µl of each test solution or hexane (control) was placed onto a filter paper strip (1 cm2, Whatman No. 1) and into a Pasteur pipette (15cm long), which served as the odor cartridge.
Signals were recorded with an EAG system (Syntech, Kirchzarten, Germany) consisting of a universal single-ended probe (Type PRG–2), a data acquisition interface board (Type IDAC–02), and a stimulus air-controller (CS–55). Antennae were excised from the heads of beetles using micro-scissors, inserted into small droplets of electrode gel (Spectra 360, Parker Laboratories, Fairfield, NJ, USA), and mounted individually between two metal electrodes in an antenna holder, under a purified air flow (500 ml.min− 1). A Syntech PC–based signal processing system was used to amplify and process EAG signals. Stimulation tests were carried out by applying a puff of humidified pure air (200 ml.min− 1) for 2s using a stimulus controller through an odor cartridge directed over the antenna through the main branch of a glass tube (7cm long × 5mm diam.). Testing began once a relatively stable baseline had been established. A control stimulus (hexane) was applied every five test stimuli. The signals were further analyzed using the EAG 2010 software (Syntech, Kirchzarten, Germany). Odorants were tested on 8–12 antennae from different individuals for each species. EAG responses were evaluated by measuring the maximum amplitude of polarity (mV) elicited by a stimulus. The absolute value of the EAG amplitude (mV) to each test stimulus was adjusted to compensate for the solvent (hexane) by subtracting the mean EAG response of the most recent control.
Statistical Analyses. To analyze the chemical profile differences between dung types, a Permutational Analysis of Variance (PERMANOVA) with Bray-Curtis dissimilarity (BCD) matrix was applied to the GC/MS data. This was followed by a post hoc multilevel pairwise comparison from package ‘vegan’ (Oksanen et al. 2020) in R Studio®. To determine the characteristic VOCs of each dung type, an IndVal analysis (Dufrêne and Legendre 1997) was applied to the GC/MS data for each dung type.
Both the behavioral and EAG data were checked for normality with Shapiro-Wilk Tests (α = 0.05). Differences in median value responses for both datasets were analyzed using Kruskal-Wallis rank sum tests (α = 0.05) given that both datasets exhibited heteroscedasticity in their standard deviations. Following significant differences in behavioral responses, post hoc Dunn tests (α = 0.05) for multiple pairwise comparisons were performed.
To determine the relationships between food preferences and the EAG profiles, we performed a Permutational Analysis of Variance (PERMANOVA) with the Bray-Curtis dissimilarity (BCD) matrix obtained from the EAG data using ‘Feeding preference’ as a factor. Post-hoc pairwise comparisons among groups were obtained by calculating a pseudo-F statistic for each treatment and P values estimated by using a permutation procedure (9999 iterations in this study) followed by a Bonferroni correction to the P values. For a graphical illustration of the differences detected we ran a Canonical Variates Analysis (CVA) (Lavine and Rayens 2009) applied to the physiological data grouped by factor ‘Feeding preference’. To determine which compounds were primarily responsible for the differences among feeding preference groups, a Similarity Percentages analysis (SIMPER) was also tested using the Bray-Curtis dissimilarity measure (Clarke 1993) and radar plots were made to display multivariate data in the form of a two-dimensional chart showing the antennal response of each feeding preference group to each EAG-active compound. These analyses were performed using PAST software (Hammer et al. 2001).
Results
Chemical Relationships Among Cow, Horse, and Rabbit Dung. VOC profiles differed among dung types, both quantitatively and qualitatively (PERMANOVA on BCD, permutations = 9999, df = 2, pseudo-F = 15.00, P = 0.004). In total, we found 51 different compounds, 18 of which were found in rabbit, 27 in cow, and 40 in horse dung (Table2). We also identified 30 VOCs associated with a particular dung type (IndVal > 0.45, P < 0.05; see Table2). We found compounds with significant IndVal values exclusive to the different dung types, including 1H-indole for cow, skatole, acetophenone, and undecane for horse, and verbenone, 1,8-cineole, and camphene for rabbit dung. Other compounds were characteristic of both cow and horse dung, such as ρ-cresol and 6-methyl-5-hepten-2-one. Finally, some compounds with significant IndVal were shared among all three dung types, such as ρ-cymene, nonanal, β-caryophyllene, sabinene, and γ-terpinene.
Feeding Preference Behavior. The olfactometer tests showed five groups of species with different feeding preferences (see Table1). The first group, comprising Ammoecius elevatus, Onthophagus maki, and Ateuchetus cicatricosus, showed a preference to cow dung (P < 0.05, P < 0.001, and P < 0.001, respectively; for statistical details see Online Resource Fig. S1). Adults of Copris hispanus, O. emarginatus, and Typhaeus typhoeus preferred horse dung (P < 0.01, P < 0.01, and P < 0.001, respectively; for statistical details see Online Resource Fig. S1), two species, Anomius baeticus and Thorectes valencianus, preferred rabbit dung (P < 0.01, and P < 0.001, respectively; for statistical details see Online Resource Fig. S1), while, a group comprising Aphodius fimetarius, Bubas bison, O. fracticornis, O. melitaeus, Ceratophyus hoffmannseggi and Sericotrupes niger had equal preference to both cow and horse dung (P < 0.05, P < 0.01, P < 0.05, P < 0.001, P < 0.01, and P < 0.001, respectively; for statistical details see Online Resource Fig. S1). Finally, Jekelius hernandezi exhibited similar attraction to all three dung types (P = 0.35; see Online Resource Fig. S1).
Electroantennography Responses to VOCs. Having established trophic preferences of the dung beetles to the different dung types, we studied the olfactory basis of this attraction. A group of 19 compounds selected from the VOCs emitted by the three dung types was tested individually. EAG responses revealed that dung beetles responded to all compounds, albeit with diverse profiles among species (Fig.1 and Online Resource Table S1).
The relationships between behavioral preference and EAG profiles were plotted using a Canonical Variates Analysis of the EAG responses of 15 dung beetle species grouped by the ‘Feeding preference’ factor resulting from the application of stepwise discriminant function analysis to 19 EAG-active compounds (the three canonical variates represent 67.3% of the total variation; see Fig.2). PERMANOVA analysis revealed that dung beetles with different feeding preferences had differences in EAG responses to the compounds (PERMANOVA on BCD, permutations = 9999, df = 4, pseudo-F = 4.95, P < 0.001). EAG responses elicited by all five feeding-preference groups of dung beetles were different from each other in pairwise comparisons (Bonferroni-corrected P < 0.05, in all cases).
Plots of the Canonical Variates Analysis of eletroantennogram (EAG) responses of 15 dung beetle species grouped by ‘Feeding preference’ factor on the first three canonical variates, resulting from the application of stepwise discriminant function analysis to 19 EAG-active compounds. (A) Canonical variates 1 and 2 showing 42.9% of the total variation. (B) Canonical variates 1 and 3 showing 24.4% of the total variation
Considering the 19 EAG-active compounds, radar plots showed different sensitivity EAG profiles for each of the feeding groups (Fig.3). The SIMPER analysis, performed to explore if distinct sensory profiles of the groups of species can be attributed to a specific or set of EAG-active compounds, showed that a subset of compounds elicited strong dissimilarities among the feeding preference groups (Table3). Of these EAG responses, nonanal and sabinene were associated with species with a preference for rabbit dung, acetophenone, ρ-cresol, 2-heptanone, and 6-methyl-5-hepten-2-one to species with a preference to horse dung and 1H-indole to species with a preference for cow dung. EAG responses to 6-methyl-5-hepten-2-one were also relevant to species with a preference for both cow and horse dung. Finally, nonanal and ρ-cresol were associated with J. hernandezi, the only species considered a generalist.
Radar charts comparing dung beetle eletroantennogram profiles of the different feeding preference groups. For dissimilarity and contribution percentages that each compound contributes among the five feeding preference groups see Table3
Discussion
Volatile Chemical Profile of Dung Types and Feeding Preferences in Dung Beetles
Each dung type showed a diverse and characteristic assemblage of VOCs, including hydrocarbons, aldehydes, ketones, alcohols, phenols, monoterpenes, sesquiterpenes and a miscellaneous group of compounds that differed both quantitatively and qualitatively. Among these compounds, ρ-cresol, 1H-indole, and skatole are some of the most frequently cited dung volatiles in olfactory studies on dung beetles (Dormont et al. 2010; Frank et al. 2018b; Stavert et al. 2014). As found in previous studies, ρ-cresol was the most abundant compound in cow (28%) and horse (25%) dung (Table2). As such, the chemical profile of the VOCs identified in cow and horse dung are likely representative of freshly excreted dung samples, comprised primarily of anaerobically produced volatiles along with several mono- and sesquiterpenes that are normally obtained from a variety of shrub and pasture species (Elegbede and Gould 2002; Estell et al. 2008). Though not detected in this study, 1H-indole has previously been identified in horse dung (Hough et al. 2018), as well as various domesticated (DeMoss and Moser 1969; Martineau and Laflamme 2002) and non-domesticated mammals (Stahl et al. 2015; Vester et al. 2008). Though undecane was found only in horse dung, previous studies have identified this in various farm animals (Aii et al. 1980; Hobbs et al. 1996; Murphy et al. 2012), as well as in human, excrement (Raman et al. 2013). It is important to note that some of the VOCs identified in cow and horse dung were also found in rabbit dung, even though it had the lowest number of compounds (Table2, see also Goodrich et al. 1981). For example, nonanal was found in low abundance in all dung types examined. This compound has been found in several dung types, including dog, white rhino, red deer, wild boar, fox, and sheep (Arnould et al. 1998; Dormont et al. 2010; Frank et al. 2018b; Marneweck et al. 2018). The compound 6-methyl-5-hepten-2-one, absent in rabbit dung, has been identified in various other dungs (Marneweck et al. 2017; Murphy et al. 2012; Raman et al. 2013, see also Goodrich et al. 1981). Rabbit dung was the most chemically distinctive, with several exclusive VOCs, such as the monoterpenes camphene, 1,8-cineole, and verbenone (Table2) (see Couch et al. 2015; Marneweck et al. 2017; Pillai et al. 2010; Raman et al. 2013). Interestingly, rabbit dung contained a high content of 1,8-cineole, which is considered a toxin to some herbivorous marsupials, necessitating metabolism and excretion (McLean and Foley 1997). Furthermore, in some phytophagous beetles, such as the Christmas beetle, Anoplognatus montanus (Coleoptera: Rutelidae), the Mountain pine beetle, Dendroctonus ponderosae (Coleoptera: Curculionidae), and the Colorado potato beetle, Leptinotarsa decemlineata (Coleoptera: Chrysomelidae), the monoterpenes 1,8-cineole and verbenone have high antifeedant activity (Edwards et al. 1993; Ortiz de Elguea-Culebras et al. 2017).
The high heterogeneity observed among the chemical profiles of the different dung types suggests that food selection behavior by dung beetles may be determined by olfactory discrimination. Other studies on attraction of dung beetles to different dung types have provided evidence for feeding preferences (Dormont et al. 2004, 2007, 2010; Galante and Cartagena 1999; Lumaret and Iborra 1996; Martín-Piera and Lobo 1996). However, our olfactometer bioassays demonstrated that most species in our study do not show exclusive selection for one type of dung, which partially supports the hypothesis of ‘choosy generalism’ in coprophagous dung beetles (Dormont et al. 2004, 2007; Frank et al. 2018b). Despite the different chemical profiles among the three types of dung, a high number of dung beetle species showed similar preferences toward cow and horse dung. This last finding may be supported by the high degree of similarity between the chemical profiles of cow and horse dung (see Table2). Although most species presented a ‘choosy generalism’ pattern in attraction to the different feces, a few species showed a preference for a single food resource. Among them, the predilection of Anomius baeticus and Thorectes valencianus for rabbit dung supports the association of these species with rabbit latrines (Verdú and Galante 2004). This suggests that these species have a more specialized strategy, associated with their modified mouthparts, as rabbit dung is hard and dry (Verdú and Galante 2004).
Feeding Preferences in Dung Beetles May be Mediated by Electrophysiological Responses to VOCs
EAG response profiles to the different chemicals differed among the dung beetle species, especially among species with different feeding preferences to three dung sources. This suggests that trophic preferences in dung beetles are not mediated by single VOCs, but rather through recognition of volatile mixtures. Although individual VOCs can trigger attraction towards a food resource, insects more commonly respond to complex mixtures (Clifford and Riffell 2013; Riffell et al. 2009; Riffell 2012). A field study on attraction of dung beetles using different food odor mixtures and single components revealed little to no attraction to most of the single components, with the more complex mixtures giving higher attraction (Frank et al. 2018b). Our EAG results again support the hypothesis of ‘choosy generalism’ in dung beetles. Having the ability to detect a broad range of dung VOCs, many of them characteristic of different types of dung, could allow dung beetles to locate a great variety of food resources. Given that excrements are usually ephemeral and stochastic resources (Hanski and Cambefort 1991), it may be that responding to a blend of compounds is more reliable than a single VOC specific to a given type of dung.
It is likely that VOCs from intestinal anaerobe metabolism of amino acids (Mackie et al. 1998) are common to many types of dung. Some of these compounds, such as ρ-cresol, the product of tyrosine fermentation, and 1H-indole and skatole, end products of tryptophan metabolism (Saito et al. 2018), may be candidates for a possible ‘generalized dung bouquet’. A generalized response to such compounds across species supports the results of the olfactometer tests, in which many species showed a similar preference for cow and horse dung. Our EAG results showed strong dung beetle responses to ρ-cresol, 1H-indole, and skatole, but also suggested that other compounds, such as nonanal, acetophenone, and ρ-cymene, may also be involved in a ‘generalized dung bouquet’. Acetophenone, which was only identified in horse dung in our study, has previously been identified in dung samples of cows (Laor et al. 2008), pigs (Blanes-Vidal et al. 2009), and several wild vertebrates (Apps et al. 2012; Marneweck et al. 2017; Martín et al. 2010; Stahl et al. 2015), indicating that this may also be a general component of dung. Other electrophysiological and behavioral studies on dung beetles have shown the importance of compounds such as ρ-cresol, indole, and skatole in dung beetle responses (Frank et al. 2018b; Inouchi et al. 1988; Shibuya and Inouchi, 1982; Weithmann et al. 2020).
Although certain compounds could be part of a ‘generalized dung bouquet’, acting as a common attractant to a large number of dung beetle species, our results also suggested that some compounds could determine preferences for a particular dung. For example, acetophenone, 2-heptanone, and 6-methyl-5-hepten-2-one may influence beetles to select horse dung (Table3). Additionally, nonanal, sabinene, and verbenone may function as key components to attract specialized species such as Thorectes valencianus and Anomius baeticus to rabbit dung (Table3).
Conclusion
Chemical analyses showed that dung odor is composed of many components. Although the blend is often dominated by a few main components, this does not necessarily mean that these components provide the most important signal to dung beetles. Our combination of behavioral and physiological bioassays suggested the existence of key components for a possible ‘generalized dung bouquet’, as well as suggesting that some compounds could be involved in determining preferences for specific dung types. To further understand the role of key VOCs of dung it is crucial to conduct more detailed behavioral and electrophysiological studies to understand their biological, ecological, and evolutionary significance to dung beetles.
Data Availability
Data can be made available upon reasonable request.
References
Adams RP (2017) Identification of essential oil components by gas chromatography/mass spectroscopy, 4.1. Allured publishing
Ahrens D, Schwarzer J, Vogler AP (2014) The evolution of scarab beetles tracks the sequential rise of angiosperms and mammals. Proc R Soc B Biol Sci 281:20141470. https://doi.org/10.1098/rspb.2014.1470
Aii T, Yonaga M, Tanaka H (1980) Changes in headspace volatiles of feed in the digestive tracts of cattle. Japanese Soc Grassl Sci 26:223–230
Amann A, Costello BDL, Miekisch W, Schubert J, Buszewski B, Pleil J, Ratcliffe N, Risby T (2014) The human volatilome: Volatile organic compounds (VOCs) in exhaled breath, skin emanations, urine, feces and saliva. J Breath Res 8:17. https://doi.org/10.1088/1752-7155/8/3/034001
Apps P, Mmualefe L, McNutt JW (2012) Identification of volatiles from the secretions and excretions of african wild dogs (Lycaon pictus). J Chem Ecol 38:1450–1461. https://doi.org/10.1007/s10886-012-0206-7
Arnould C, Malusse C, Signoret JP, Descoins C (1998) Which chemical constituents from dog feces are involved in its food repellent effect in sheep? J Chem Ecol 24:559–576. https://doi.org/10.1023/A:1022321104758
Blanes-Vidal V, Hansen MN, Adamsen APS et al (2009) Characterization of odor released during handling of swine slurry: Part I. Relationship between odorants and perceived odor concentrations. Atmos Environ 43:2997–3005. https://doi.org/10.1016/j.atmosenv.2008.10.016
Bogoni JA, Hernández MIM (2014) Attractiveness of native mammal’s feces of different trophic guilds to dung beetles (Coleoptera: Scarabaeinae). J Insect Sci 14:1–7. https://doi.org/10.1093/jisesa/ieu161
Cai L, Koziel JA, Liang Y et al (2007) evaluation of zeolite for control of odorants emissions from simulated poultry manure storage. J Environ Qual 36:184–193. https://doi.org/10.2134/jeq2006.0052
Carpaneto GM, Mazziotta A, Ieradi M (2010) Use of habitat resources by scarab dung beetles in an african savanna. Environ Entomol 39:1756–1764. https://doi.org/10.1603/EN09249
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143. https://doi.org/10.1111/j.1442-9993.1993.tb00438.x
Clifford MR, Riffell JA (2013) Mixture and odorant processing in the olfactory systems of insects: A comparative perspective. J Comp Physiol A Neuroethol Sensory Neural Behav Physiol 199:911–928. https://doi.org/10.1007/s00359-013-0818-6
Couch RD, Dailey A, Zaidi F et al (2015) Alcohol induced alterations to the human fecal VOC metabolome. PLoS ONE 10:1–24. https://doi.org/10.1371/journal.pone.0119362
de Elguea-Culebras GO, Sánchez-Vioque R, Berruga MI, Herraiz-Peñalver D, Santana-Méridas O (2017) Antifeedant effects of common terpenes from Mediterranean aromatic plants on Leptinotarsa decemlineata. J Soil Sci Plant Nutr 17:475–485. https://doi.org/10.4067/S0718-95162017005000034
Dehnhard M, Bernal-Barragan H, Claus R (1991) Rapid and accurate high-performance liquid chromatographic method for the determination of 3-methylindole (skatole) in faeces of various species. J Chromatogr B Biomed Sci Appl 566:101–107. https://doi.org/10.1016/0378-4347(91)80114-R
DeMoss RD, Moser K (1969) Tryptophanase in diverse bacterial species. J Bacteriol 98:167–171. https://doi.org/10.1128/jb.98.1.167-171.1969
Dormont L, Epinat G, Lumaret JP, Rapior S, Lumaret JP (2004) Trophic preferences mediated by olfactory cues in dung beetles colonizing cattle and horse dung. Environ Entomol 33:370–377. https://doi.org/10.1603/0046-225x-33.2.370
Dormont L, Jay-Robert P, Bessiere JM et al (2010) Innate olfactory preferences in dung beetles. J Exp Biol 213:3177–3186. https://doi.org/10.1242/jeb.040964
Dormont L, Rapior S, McKey DB, Lumaret JP (2007) Influence of dung volatiles on the process of resource selection by coprophagous beetles. Chemoecology 17:23–30. https://doi.org/10.1007/s00049-006-0355-7
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for flexible asymmetrical approach. Ecol Monogr 67:345–366
Edwards PB, Wanjura WJ, Brown WV (1993) Selective herbivory by Christmas beetles in response to intraspecific variation in Eucalyptus terpenoids. Oecologia 95:551–557. https://doi.org/10.1007/BF00317440
Elegbede JA, Gould MN (2002) Monoterpenes reduced adducts formation in rats exposed to aflatoxin B1. Afr J Biotechnol 1:29–36. https://doi.org/10.4314/ajb.v1i2.14814
Errouissi F, Haloti S, Jay-Robert P, Janati-Idrissi A, Lumaret JP (2004) Effects of the attractiveness for dung beetles of dung pat origin and size along a climatic gradient. Environ Entomol 33:45–53. https://doi.org/10.1603/0046-225X-33.1.45
Estell RE, Fredrickson EL, Anderson DM, Remmenga MD (2008) Effects of cis-β-ocimene, cis-sabinene hydrate, and monoterpene and sesquiterpene mixtures on alfalfa pellet intake by lambs. J Anim Sci 86:1478–1484. https://doi.org/10.2527/jas.2007-0699
Finn JA, Giller PS (2002) Experimental investigations of colonisation by north temperate dung beetles of different types of domestic herbivore dung. Appl Soil Ecol 20:1–13. https://doi.org/10.1016/S0929-1393(02)00011-2
Frank K, Bruckner A, Bluthgen N, Schmitt T (2018a) In search of cues: dung beetle attraction and the significance of volatile composition of dung. Chemoecology 28:145–152. https://doi.org/10.1007/s00049-018-0266-4
Frank K, Hülsmann M, Assmann T, Schmitt T, Blüthgen N (2017) Land use affects dung beetle communities and their ecosystem service in forests and grasslands. Agric Ecosyst Environ 243:114–122. https://doi.org/10.1016/j.agee.2017.04.010
Frank K, Krell FT, Slade EM, Raine EH, Chiew LY, Schmitt T, Vairappan CS, Walter P, Blüthgen N (2018b) Global dung webs: high trophic generalism of dung beetles along the latitudinal diversity gradient. Ecol Lett 21:1229–1236. https://doi.org/10.1111/ele.13095
Galante E, Cartagena MC (1999) Comparison of mediterranean dung beetles (Coleoptera: Scarabaeoidea) in cattle and rabbit dung. Environ Entomol 28:420–424. https://doi.org/10.1093/ee/28.3.420
Giménez Gómez VC, Verdú JR, Velasco SJE, Zurita GA (2021) Dung beetle trophic ecology: are we misunderstanding resources attraction? Ecol Entomol 46:552–561. https://doi.org/10.1111/een.13001
Goodrich BS, Hesterman ER, Shaw KS, Mykytowycz R (1981) Identification of some volatile compounds in the odor of fecal pellets of the rabbit, Oryctolagus cuniculus. J Chem Ecol 7:817–827. https://doi.org/10.1007/BF00992380
Gunter NL, Weir TA, Slipinksi A, Bocak L, Cameron SL (2016) If dung beetles (Scarabaeidae: Scarabaeinae) arose in association with dinosaurs, did they also suffer a mass co-extinction at the K-Pg boundary? PLoS ONE 11(5):11. https://doi.org/10.1371/journal.pone.0153570
Halffter G, Matthews EG (1966) The natural history of dung beetles of the subfamily Scarabaeinae (Coleoptera, Scarabaeidae). Folia Entomologica Mexicana 12–14:1–312
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4:9
Hanski I (1991) The dung insect community. In: Hanski I, Cambefort Y (eds) Dung Beetle Ecology. Princeton University Press, pp 5–21
Holter P (2016) Herbivore dung as food for dung beetles: elementary coprology for entomologists. Ecol Entomol 41:367–377. https://doi.org/10.1111/een.12316
Holter P, Scholtz CH (2007) What do dung beetles eat? Ecol Entomol 32:690–697. https://doi.org/10.1111/j.1365-2311.2007.00915.x
Hough R, Archer D, Probert C (2018) A comparison of sample preparation methods for extracting volatile organic compounds (VOCs) from equine faeces using HS-SPME. Metabolomics 14(19):1–10. https://doi.org/10.1007/s11306-017-1315-7
Inouchi J, Shibuya T, Hatanaka T (1988) Food odor responses of single antennal olfactory cells in the Japanese dung beetle, Geotrupes auratus (Coleoptera: Geotrupidae). Appl Entomol Zool 23:167–174
Koziel JA, Spinhirne JP, Lloyd JD et al (2005) Evaluation of sample recovery of malodorous livestock gases from air sampling bags, solid-phase microextraction fibers, Tenax TA sorbent tubes, and sampling canisters. J Air Waste Manag Assoc 55:1147–1157. https://doi.org/10.1080/10473289.2005.10464711
Laor Y, Koziel JA, Cai L, Ravid U (2008) Chemical-sensory characterization of dairy manure odor using headspace solid-phase microextraction and multidimensional gas chromatography mass spectrometry-olfactometry. J Air Waste Manag Assoc 58:1187–1197. https://doi.org/10.3155/1047-3289.58.9.1187
Larsen TH, Lopera A, Forsyth A (2006) Extreme trophic and habitat specialization by Peruvian dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae). Coleopt Bull 60:315–324. https://doi.org/10.1649/0010-065X(2006)60[315:ETAHSB]2.0.CO;2
Lavine BK, Rayens WS, Elsevier BV, Amsterdam (2009) Netherlands https://doi.org/10.1016/B978-044452701-1.00024-7. ISBN: 978-0-444-52701-1
Lumaret JP, Iborra O (1996) Separation of trophic niches by dung beetles (Coleoptera, Scarabaeoidea) in overlapping habitats. Pedobiologia (Jena) 40:392–404
Mackie RI, Stroot PG, Varel VH (1998) Biochemical identification and biological origin of key odor components in livestock waste. J Anim Sci 76:1331–1342. https://doi.org/10.2527/1998.7651331x
Marneweck C, Jürgens A, Shrader AM (2017) Temporal variation of white rhino dung odours. J Chem Ecol 43:955–965. https://doi.org/10.1007/s10886-017-0890-4
Marneweck C, Jürgens A, Shrader AM (2018) Ritualised dung kicking by white rhino males amplifies olfactory signals but reduces odour duration. J Chem Ecol 44:875–885. https://doi.org/10.1007/s10886-018-0988-3
Martín J, Barja I, López P (2010) Chemical scent constituents in feces of wild Iberian wolves (Canis lupus signatus). Biochem Syst Ecol 38:1096–1102. https://doi.org/10.1016/j.bse.2010.10.014
Martín-Piera F, Lobo JM (1996) A comparative discussion of trophic preferences in dung beetle communities. Miscellà nia Zoològica 191:13–31
Martineau B, Laflamme DP (2002) Effect of diet on markers of intestinal health in dogs. Res Vet Sci 72:223–227. https://doi.org/10.1053/rvsc.2002.0548
McLean S, Foley WJ (1997) Metabolism of Eucalyptus terpenes by herbivorous marsupials. Drug Metab Rev 29:213–218. https://doi.org/10.3109/03602539709037582
Murphy KR, Wenig P, Parcsi G et al (2012) Characterizing odorous emissions using new software for identifying peaks in chemometric models of gas chromatography-mass spectrometry datasets. Chemom Intell Lab Syst 118:41–50. https://doi.org/10.1016/j.chemolab.2012.07.006
Nibaruta G, Desière M, Debaere R (1980) Etude comparée de la composition chimique des excréments de quelques grands mammifères herbivores africains. Acta Zool Pathol Antverp 75:59–70
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2020) vegan: Community Ecology Package. 298. R package version 2.5-7. https://CRAN.R-project.org/package=vegan
Pillai SM, Parcsi G, Wang X et al (2010) Assessment of direct headspace analysis of broiler chicken litter odorants. Chem Eng Trans 23:207–212. https://doi.org/10.3303/CET1023035
Raman M, Ahmed I, Gillevet PM et al (2013) Fecal microbiome and volatile organic compound metabolome in obese humans with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 11:868–875e3. https://doi.org/10.1016/j.cgh.2013.02.015
Riffell JA (2012) Olfactory ecology and the processing of complex mixtures. Curr Opin Neurobiol 22:236–242. https://doi.org/10.1016/j.conb.2012.02.013
Riffell JA, Lei H, Christensen TA, Hildebrand JG (2009) Characterization and Coding of Behaviorally Significant Odor Mixtures. Curr Biol 19:335–340. https://doi.org/10.1016/j.cub.2009.01.041
Saito Y, Sato T, Nomoto K, Tsuji H (2018) Identification of phenol- and p-cresol-producing intestinal bacteria by using media supplemented with tyrosine and its metabolites. FEMS Microbiol Ecol 94:1–11. https://doi.org/10.1093/femsec/fiy125
Shibuya T, Inouchi J (1982) Fine structure and odor responses of olfactory sensilla of the japanese dung beetle, Copris pecuarius (Coleoptera: Scarabaidae). Jap J appl Ent Zool 26:194–195
Stahl RS, Ellis CK, Nol P et al (2015) Fecal volatile organic ccompound profiles from white-tailed deer (Odocoileus virginianus) as indicators of mycobacterium bovis exposure or Mycobacterium bovis bacille calmette-guerin (BCG) vaccination. PLoS ONE 10:1–20. https://doi.org/10.1371/journal.pone.0129740
Stavert JR, Drayton BA, Beggs JR, Gaskett AC (2014) The volatile organic compounds of introduced and native dung and carrion and their role in dung beetle foraging behaviour. Ecol Entomol 39:556–565. https://doi.org/10.1111/een.12133
Terada A, Hara H, Oishi T et al (1992) Effect of dietary lactosucrose on faecal flora and faecal metabolites of dogs. Microb Ecol Health Dis 5:87–92. https://doi.org/10.3109/08910609209141294
Tribe GD, Burger BV (2011) Olfactory ecology. In: Simmons LW, Ridsdill-Smith TJ (eds) Ecology and evolution of dung beetles. John Wiley and Sons, Ltd, Chichester, UK, pp 87–106. https://doi.org/10.1002/9781444342000.ch5
Tyndale-Biscoe M (1984) Age-grading methods in adult insects: A review. Bull Entomol Res 74:341–377. https://doi.org/10.1017/S0007485300015637
Van den Dool H, Kratz PD (1963) A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J Chromatogr 11:463–471. https://doi.org/10.1016/S0021-9673(01)80947-X
Verdú JR, Galante E (2004) Behavioural and morphological adaptations for a low-quality resource in semi-arid environments: Dung beetles (Coleoptera, Scarabaeoidea) associated with the European rabbit (Oryctolagus cuniculus L.). J Nat Hist 38:705–715. https://doi.org/10.1080/0022293021000041707
Verdú JR, Lobo JM, Numa C, Pérez-Ramos IM, Galante E, Marañón T (2007) Acorn preference by the dung beetle, Thorectes lusitanicus, under laboratory and field conditions. Anim Behav 74:1697–1704. https://doi.org/10.1016/j.anbehav.2007.03.016
Vester BM, Burke SL, Dikeman CL et al (2008) Nutrient digestibility and fecal characteristics are different among captive exotic felids fed a beef-based raw diet. Zoo Biol 27:126–136. https://doi.org/10.1002/zoo.20172
Walton C, Fowler DP, Turner C et al (2013) Analysis of volatile organic compounds of bacterial origin in chronic gastrointestinal diseases. Inflamm Bowel Dis 19:2069–2078. https://doi.org/10.1097/MIB.0b013e31829a91f6
Weithmann S, Hoermann CV, Schmitt T, Steiger S, Ayasse M (2020) The attraction of the dung beetle Anoplotrupes stercorosus (Coleoptera: Geotrupidae) to volatiles from vertebrate cadavers. Insects 11:1–16. https://doi.org/10.3390/insects11080476
Wurmitzer C, Bluthgen N, Krell FT, Maldonado B, Ocampo F, Müller JK, Schmitt T (2017) Attraction of dung beetles to herbivore dung and synthetic compounds in a comparative field study. Chemoecology 27:75–84. https://doi.org/10.1007/s00049-017-0232-6
Wurmitzer C, Bluthgen N, Krell FT, Maldonado B, Ocampo F, Müller JK, Schmitt T (2017) Attraction of dung beetles to herbivore dung and synthetic compounds in a comparative field study. Chemoecology 27:75–84. https://doi.org/10.1007/s00049-017-0232-6
Acknowledgements
This research was supported by the projects PID2019-105418RB-I00 (Secretaría de Estado de Investigación, Desarrollo e Innovación – Ministerio de Ciencia e Innovación) and AICO-2020-031 (Dirección General de Ciencia e Investigación. Consellería de Innovación, Universidades, Ciencia y Sociedad Digital – Generalitat Valenciana). M.A. Urrutia acknowledges the support of the Dirección General de Ciencia e Investigación, Consellería de Innovación, Universidades, Ciencia y Sociedad Digital, Generalitat Valenciana, for supporting his Ph.D. scholarship (GRISOLIAP/2021/185). We are grateful to P. Blasco and P. Candela for their assistance with GC/MS-. We thank Dr. A. Ortiz for providing some standard compounds and for his valuable assistance with the mass spectrometry. We also thank Dr. F.-T. Krell for excellent support providing complementary data and literature and an anonymous reviewer for their helpful comments on an earlier version of the manuscript. We also thank to Dr. S. Foster for helping to improve the quality of the manuscript.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
JRV and VC conceived and designed the study. Data collection during the laboratory bioassays were performed by VC and MU. Data analysis was performed by JRV and MU. The final draft of the manuscript was written by all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest/Competing Interests
The authors declare no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Urrutia, M.A., Cortez, V. & Verdú, J.R. Links Between Feeding Preferences and Electroantennogram Response Profiles in Dung Beetles: The Importance of Dung Odor Bouquets. J Chem Ecol 48, 690–703 (2022). https://doi.org/10.1007/s10886-022-01383-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-022-01383-1