Abstract
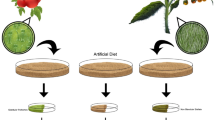
Plant trichomes are known for their capability to produce and store secondary metabolites that protect plants from biotic and abiotic stresses. 1H NMR studies on intact individual trichomes located on the leaf surface of Nicotiana attenuata revealed the presence of two major secondary metabolites: nicotine, the signature metabolite of the genus, and phaseoloidin, a homogentisic acid glucoside. This glucoside was reported originally from the seeds of Entada phaseoloides, and this is the first report of its occurrence in a Solanaceous plant. Artificial diet feeding bioassays with Manduca sexta and Spodoptera littoralis larvae, two important herbivores of N. attenuata, revealed that the ingestion of phaseoloidin negatively influenced caterpillar performance. This effect was more pronounced for the generalist, S. littoralis, than for the specialists, M. sexta.




Similar content being viewed by others
References
Arrendale, R. F., Severson, R. F., Sisson, V. A., Costello, C. E., Leary, J. A., Himmelsbach, D. S., and Vanhalbeek, H. 1990. Characterization of the sucrose ester fraction from Nicotiana glutinosa. J. Agric. Food Chem. 38:75–85.
Baldwin, I. T. 1989. Mechanism of damage-induced alkaloid production in wild tobacco. J. Chem. Ecol. 15:1661–1680.
Barua, A. K., Chakrabarty, M., Datta, P. K., and Ray, S. 1988. Phaseoloidin, a homgentisic acid glucoside from Entada phaseoloides. Phytochemistry 27:3259–3261.
Cardoso, M. Z. 2008. Herbivore handling of a plants trichome: The case of Heliconius charithonia (L.) (Lepidoptera: Nymphalidae) and Passiflora lobata (Killip) Hutch. (Passifloraceae). Neotrop. Entomol. 37:247–252.
Chortyk, O. T., Kays, S. J., and Teng, Q. 1997. Characterization of insecticidal sugar esters of Petunia. J. Agric. Food Chem. 45:270–275.
Dai, X., Wang, G., Yang, D. S., Tang, Y., Broun, P., Marks, M. D., Sumner, L. W., Dixon, R. A., and Zhao, P. X. 2010. TrichOME: A comparative omics database for plant trichomes. Plant Physiol. 152:44–54.
Duke, M. V., Paul, R. N., Elsohly, H. N., Sturtz, G., and Duke, S. O. 1994. Localization of artemisinin and artemisitene in foliar tissues of glanded and glandless biotypes of Artemisia annua. Int. J. Plant Sci. 155:365–372.
Duke, S. O., Canel, C., Rimando, A. M., Telle, M. R., Duke, M. V., and Paul, R. N. 2000. Current and potential exploitation of plant glandular trichome productivity. Adv. Bot. Res.: Academic Press. p 121–151.
Goodspeed, T. H. 1955. The Genus Nicotiana. New York, Stechert-Hafner, Inc.
Heiling, S., Schuman, M. C., Schoettner, M., Mukerjee, P., Berger, B., Schneider, B., Jassbi, A. R., and Baldwin, I. T. 2010. Jasmonate and ppHsystemin regulate key malonylation steps in the biosynthesis of 17-Hydroxygeranyllinalool diterpene glycosides, an abundant and effective direct defense against herbivores in Nicotiana attenuata. Plant Cell 22:273–292.
Jones, D., Jones, G. A., Hagen, T., and Creech, E. 1985. Wild species of Nicotiana as a new source of tobacco resistance to the tobacco hornworm Manduca sexta. Entomol. Exp. Appl. 38:157–164.
Krügel, T., Lim, M., Gase, K., Halitschke, R., and Baldwin, I. T. 2002. Agrobacterium-mediated transformation of Nicotiana attenuata, a model ecological expression system. Chemoecology 12:177–183.
Laue, G., Preston, C. A., and Baldwin, I. T. 2000. Fast track to the trichome: Induction of N-acyl nornicotines precedes nicotine induction in Nicotiana repanda. Planta 210:510–4.
McKenzie, C. L. and Puterka, G. J. 2004. Effect of sucrose octanoate on survival of nymphal and adult Diaphorina citri (Homoptera: Psyllidae). J. Econ. Entomol. 97:970–975.
Moran, G. R. 2005. 4-Hydroxyphenylpyruvate dioxygenase. Arch. Biochem. Biophysics 433:117–128.
Morita, M., Shitan, N., Sawada, K., Van Montagu, M. C. E., Inzé, D., Rischer, H., Goossens, A., Oksman-Caldentey, K.-M., Moriyama, Y., and Yazaki, K. 2009. Vacuolar transport of nicotine is mediated by a multidrug and toxic compound extrusion (MATE) transporter in Nicotiana tabacum. Proc. Natl. Acad. Sci. USA 106:2447–2452.
Puterka, G. J., Farone, W., Palmer, T., and Barrington, A. 2003. Structure-function relationships affecting the insecticidal and miticidal activity of sugar esters. J. Econ. Entomol. 96:636–44.
Roda, A. L., Oldham, N. J., Svatos, A., and Baldwin, I. T. 2003. Allometric analysis of the induced flavonols on the leaf surface of wild tobacco (Nicotiana attenuata). Phytochemistry 62:527–36.
Schilmiller, A. L., Last, R. L., and Pichersky, E. 2008. Harnessing plant trichome biochemistry for the production of useful compounds. Plant J. 54:702–11.
Schwachtje, J., Minchin, P. E., Jahnke, S., Van Dongen, J. T., Schittko, U., and Baldwin, I. T. 2006. SNF1-related kinases allow plants to tolerate herbivory by allocating carbon to roots. Proc. Natl. Acad. Sci. USA 103:12935–40.
Shoji, T., Yamada, Y., and Hashimoto, T. 2000. Jasmonate induction of putrescine N-methyltransferase genes in the root of Nicotiana sylvestris. Plant Cell Physiol. 41:831–839.
Sirikantaramas, S., Taura, F., Tanaka, Y., Ishikawa, Y., Morimoto, S., and Shoyama, Y. 2005. Tetrahydrocannabinolic acid synthase, the enzyme controlling marijuana psychoactivity, is secreted into the storage cavity of the glandular trichomes. Plant Cell Physiol. 46:1578–1582.
Sirikantaramas, S., Yamazaki, M., and Saito, K. 2008. Mechanisms of resistance to self-produced toxic secondary metabolites in plants. Phytochem. Rev. 7:467–477.
Van Schie, C. C., Haring, M. A., and Schuurink, R. C. 2007. Tomato linalool synthase is induced in trichomes by jasmonic acid. Plant Mol. Biol. 64:251–63.
Waldbauer, G. P., Yamamoto, R. T., and Bowers, W. S. 1964. Laboratory rearing of tobacco hornworm Protoparce sexta (Lepidoptera: Sphingidae). J. Econ. Entomol. 57:93-95.
Weinhold, A. and Baldwin, I. T. 2011. Trichome-derived O-acyl sugars are a first meal for caterpillars that tags them for predation. Proc. Natl. Acad. Sci. USA 108:7855–7859.
Zador, E. and Jones, D. 1986. The biosynthesis of a novel nicotine alkaloid in the trichomes of Nicotiana stocktonii. Plant Physiol. 82:479–84.
Acknowledgments
We thank three anonymous reviewers for their comments and the Max Planck Society for funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Alexander Weinhold, Kamel Shaker, and Michael Wenzler contributed equally to the results presented in this paper.
Electronic Supplementary Materials
Below is the link to the electronic supplementary material.
Supplemental Figure S1
Light micrographs of intact glandular trichomes of Nicotiana attenuata WT leaves. A Two different trichome types are found on the leaf surface, short trichomes with a stem consisting of two to four cells and a multi cellular head (type D), as well as tall trichomes with a multi cellular base, a stem (six to ten cells) and a small head (type C). The short trichomes are found mainly on the leaf surface whereas the tall ones are mostly located on the leaf veins shown in C, where they are even larger than on the surface. Magnification (200x) of a type D trichome B and of the head of a type C trichome (100×) D reveal green structures in the heads, most likely chloroplasts, as described for N. tabacum. E Trichomes of the insertional mutant irGAL83 are characterized by their enlarged basal cells. (JPEG 0 kb)
Supplemental Figure S2
Phaseoloidin contents in the trichomes of the insertional mutant irGAL83 and the Nicotiana attenuata wild type line do not differ. Two excised leaves were gently frozen over liquid nitrogen. The trichomes were harvested by gently scratching the leaf surface with a microscope cover slip. The plate was rinsed with 10 ml methanol. Afterwards the extract was concentrated to 1 ml under a constant flow of nitrogen. After centrifuging, the supernatant was transferred to HPLC vials. Phaseoloidin content was analyzed by LC-TOF-MS. Error bars indicate standard errors (WT N = 4; insertional mutant N = 3). (PDF 70.3 kb)
Supplemental Figure S3
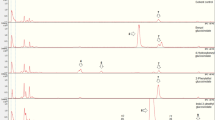
Aliphatic part of the NMR spectra from the type C and D trichome samples. The spectrum of D Head contains much more sugar and fatty acid signals, belonging to acyl sugars, than the spectra of C Tip and C Middle. (PDF 96.0 kb)
Rights and permissions
About this article
Cite this article
Weinhold, A., Shaker, K., Wenzler, M. et al. Phaseoloidin, a Homogentisic Acid Glucoside from Nicotiana Attenuata Trichomes, Contributes to the Plant’s Resistance against Lepidopteran Herbivores. J Chem Ecol 37, 1091–1098 (2011). https://doi.org/10.1007/s10886-011-0012-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-011-0012-7