Abstract
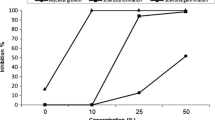
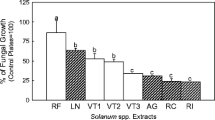
In a previous study, we observed that bract and corolla extracts from a Sclerotinia sclerotiorum-resistant sunflower contained high amounts of the known coumarins scopoletin, scopolin, and ayapin. There was a correlation between coumarin concentration and disease resistance. Thin layer chromatography showed higher concentrations of three other compounds in the resistant genotype when compared to the susceptible. A bioassay-directed purification that used column chromatography and HPLC allowed the isolation of a new compound, 3-acetyl-4-acetoxyacetophenone, and known compounds, demethoxyencecalin and 3-acetyl-4-hydroxyacetophenone. Structures were assigned from spectral data, and bioactivities were characterized by in vitro bioassays against S. sclerotiorum. The new compound, 3-acetyl-4-acetoxyacetophenone, had an antifungal activity similar to the coumarin ayapin, previously described as a potent Sclerotinia inhibitor. The speed and simplicity by which these compounds can be detected make them suitable for use in screening procedures that may identify genotypes with valuable levels of resistance. A screening of seven sunflower genotypes in a field experiment showed a correlation between these compounds and resistance to Sclerotinia.


Similar content being viewed by others
References
Acimovic, M. 1985. Sunflower disease mapping in Europe and some countries outside Europe in the period 1984–1986. Helia 11:41–49.
Bate, N. J., Orr, J., Ni, W. T., Meromi, A., Nadlerhassar, T., Doerner, P. W., Dixon, R. A., Lamb, C. J., and Elkind, Y. 1994. Quantitative relationship between phenylalanine ammonia-lyase levels and phenylpropanoid accumulation in transgenic tobacco identifies a rate-determining step in natural product synthesis. Proc. Natl. Acad. Sci. U.S.A. 91:7608–7612.
Becerra, J., Silva, M., Delle Monache, G., and Delle Monache, F. 1983. Two new chromenes from Eupatorium glechonophyllum. Less. Rev. Latinoam. Quim. 14:92–94.
Cabello-Hurtado, F., Durst, F., Jorrín, J. V., and Werck-Reichhart, D. 1998. Coumarins in Helianthus tuberosus: Characterization, induced accumulation and biosynthesis. Phytochemistry 49:1029–1036.
De Pascual, J., Bellido, I., González, M., Muriel, M., and Hernández, J. 1981. Aromatic compounds from Artemisia campestris subsp glutinosa. Phytochemistry 20:2417–2420.
Ding, H., Lamb, R. J., and Ames, N. 2000. Inducible production of phenolic acids in wheat and antibiotic resistance to Sitodiplosis mosellana. J. Chem. Ecol. 26:969–985.
Dixon, R. A., Achnine, L., Kota, P., Liu, C. J., Reddy, M. S. S., and Wang, L. J. 2002. The phenylpropanoid pathway and plant defense: A genomics perspective. Mol. Plant Pathol. 3:371–390.
Kulkarni, M., Nagasampagi, B., Deshpande, S., and Sharma, R. 1987. 5 chromenes from Blepharispermum subsessile. Phytochemistry 26:2969–2971.
Macías, F., Varela, R., Torres, A., Oliva, R., and Molinillo, J. 1998. Allelopathic studies in cultivar, part 10. Bioactive norsesquiterpenes from Helianthus annuus potential allelopathic activity. Phytochemistry 48:631–636.
Macías, F. A., Molinillo, J. M. G., Varela, R. M., Torres, A., and Galindo, J. C. G. 1999. Bioactive Compounds from the Genus Helianthus, pp. 121–148, in: F. A. Macías, Galindo, J. C. G., Molinillo, J. M. G., Cutler, H. G., (ed.). Recent Advances in Allelopathy, vol. I. A Science for the Future. Servicio de Publicaciones de la Universidad de Cádiz, Spain.
Mullin, C. A., Alfatafta, A. A., Harman, J. L., Serino, A. A., and Everett, S. L. 1991. Corn rootworm feeding on sunflower and other compositae: Influence of floral terpenoid and phenolic factors, pp. 278–292, in: P. A. Hedin, (ed.). Naturally Occurring Pest Bioregulators, ACS Symposium Series No. 449. American Chemical Society, Washington, DC.
Okunade, A., Hufford, C., Clark, A., and Lentz, D. 1997. Antimicrobial properties of the constituents of Piper aduncum. Phytotherapy Res. 11:142–144.
Pereyra, V. R., Bazzalo, M. E., and Sala, C. 1991. Variabilidad y estabilidad entre hibridos comerciales de girasol para la resistencia a la podredumbre del capitulo provocada por el hongo Sclerotinia sclerotiorum (Lib.) de Bary. 1° R. Nac. Oleagin.
Picman, A., and Schneider, E. 1993. Inhibition of fungal growth by selected sesquiterpene lactones. Biochem. Sys. Ecol. 21:307–314.
Prats, E., Rubiales, D., and Jorrín, J. 2002. Acibenzolar-S-methyl-induced resistance to sunflower rust (Puccinia helianthi) is associated with an enhancement of coumarins on foliar surface. Physiol. Mol. Plant Pathol. 60:155–162.
Prats, E., Bazzalo, M. E., León, A., and Jorrín, J. V. 2003. Accumulation of soluble phenolic compounds in sunflower capitula correlates with resistance to Sclerotinia sclerotiorum. Euphytica 132:321–329.
Prats, E., Bazzalo, M. E., León, A., and Jorrín, J. 2006. Fungitoxic effect of scopolin and related coumarins on Sclerotinia sclerotiorum. A way to overcome sunflower head rot. Euphytica 147:451–460.
Prats, E., Llamas, M. J., Jorrín, J., and Rubiales, D. 2007. Constitutive coumarin accumulation on sunflower leaf surface prevents rust germ tube growth and appressorium differentiation. Crop Sci. 47:1–6.
Purdy, L. H. 1979. History, disease and symptomatology, host range, geographic distribution and impact. Symposium on Sclerotinia taxonomy, biology and pathology. Phytopathology 69:875–880.
Satoh, A., Utamura, H., Ishizuka, M., Endo, N., Tsuji, M., and Nishimura, H. 1996. Antimicrobial benzopyrans from the receptacle of sunflower. Biosci. Biotech. Biochem. 60:664–665.
Serghini, K. 2000. Papel defensivo de las 7-hidroxicumarinas simples de girasol (Helianthus annuus) frente a hongos (Plasmopara halstedii) y plantas parasitas (Orobanche cernua). Ph.D. Thesis. University of Cordoba, Spain.
Spring, O., Rodon, U., and Macías, F. A. 1992. Sesquiterpenes from noncapitate glandular trichomes of Helianthus annuus. Phytochemistry 31:1541–1544.
Spring, O., Zipper, R., Conrad, J., Vogler, B., Klaiber, I., and Da Costa, F. B. 2003. Sesquiterpene lactones from glandular trichomes of Viguiera radula (Heliantheae; Asteraceae). Phytochemistry 62:1185–1189.
Srivastava, R. P., and Proksch, P. 1993. Insecticidal and antifeedant effects of compounds of plant origin against insect pests. Indian J. Plant Prot. 21:234–239.
Tsuda, H., Ishitani, Y., Takemura, Y., Suzuki, Y., and Kato, T. 1997. 6-acetyl-8-hydroxy-2,2-dimethylchromene, an antioxidant in sunflower seeds; Its isolation and synthesis and antioxidant activity of its derivatives. Heterocycles 44:139–142.
Urdangarin, C., Regente, M. C., Jorrín, J., and De La Canal, L. 1999. Sunflower coumarin phytoalexins inhibit the growth of the virulent pathogen Sclerotinia sclerotiorum. J. Phytopathol. 147:441–443.
Varga, E., Szendrei, K., Dinya, Z., and Reisch, J. 1984. Aromatic compounds from the stalk of Helianthus annuus. Fitoterapia 55:307–309.
Waiss, A. C., Chan, B. G., Elliger, C. A., Dreyer, D. L., Binder, R. G., and Gueldner, R. C. 1981. Insect growth inhibitors in crop plants. Ent. Soc. Am. Bull. 27:217–221.
Acknowledgement
This work was supported by the Spanish Ministry of Science and Technology, Project AGL 2001-2420.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prats, E., Galindo, J.C., Bazzalo, M.E. et al. Antifungal Activity of a New Phenolic Compound from Capitulum of a Head Rot-resistant Sunflower Genotype. J Chem Ecol 33, 2245–2253 (2007). https://doi.org/10.1007/s10886-007-9388-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-007-9388-9