Abstract
Continuous measurement of resting energy expenditure (REE) in critically ill patients remains challenging but is required to prevent malnutrition. SenseWear Pro 3 Armband (SWA) is a research grade accelerometer for assessment of REE with the advantage of easy handling. In a prospective study we compared SWA with indirect calorimetry (IC) and predictive equations in critically ill, ventilated patients. REE was measured by SWA, IC and calculated by predictive formulas. Potential confounding factors that influence REE were also recorded. Results of SenseWear Armband and indirect calorimetry were compared using the Bland–Altman method. 34 ICU patients were investigated. SWA underestimated resting energy expenditure compared to IC with a mean bias of ΔREE = −253.6 ± 333.2 kcal, equivalent to −11.7 % (p = 0.025). This underestimation was seen in both, medical (−14.9 %) and surgical (−12.9 %) patients and the bias was greater in patients with fever (−19.0 %), tachycardia (−18.7 %) or tachypnea (−26.2 %). Differences were also noted when SWA was compared to predictive formulas. At present, SWA cannot be regarded as an alternative to indirect calorimetry. Individual measurements are often inaccurate and should be used with caution until improved algorithms, based on the results of this study, have been implemented.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Critically ill patients have a high risk of developing malnutrition [1, 2]. Their energy consumption varies greatly depending on the severity of the underlying condition and several other factors such as fever, concurrent infection, pain or surgical procedures [3]. Resting energy expenditure in patients with sepsis, burns, head trauma, mechanical ventilation or after surgery is increased of up to 50 % of normal energy requirements and omission of adjustments in substrate supply may lead to rapid malnutrition in these patients [4].
Malnutrition is associated with multiple complications as it results in impaired immune function, poor wound healing, decreased renal function, hospital-acquired infections, reduced respiratory capacity and consequently a longer ventilation time [5]. These factors contribute to increased morbidity and mortality, a prolonged hospital stay and increased costs [6–10]. Early overfeeding can also induce complications, such as hyperlipidemia, liver dysfunction, hyperglycemia and immunosuppression [5, 11–13].
Therefore both, hypocaloric and hypercaloric nutrition should be avoided [14]. Unfortunately, data regarding the adjustment of daily energy delivery to the patient’s needs are scarce [4, 15]. There is evidence that energy requirements are dynamic during ICU stay. Energy needs were first increasing and later decreasing in sepsis and trauma and both sepsis and trauma patients [16]. In order to reduce malnutrition methods are required that allow an optimal nutrient supply adjusted to different stages of disease [1, 4]. Indirect calorimetry (IC) and the double-labelled-water method are the gold standard for determining energy requirements. Technically indirect calorimetry is based on measurements of respiratory gases, i.e. consumption of oxygen (VO2) and production of carbon dioxide (VCO2). By using these parameters heat, which is generated during oxygenation of (carbon-based) substrates and an equivalent to human energy, can be assessed. Indirect calorimetry is suitable in both spontaneously breathing and mechanically ventilated patients [17]. Moreover by determination of the in- and expiratory O2 concentrations, and the CO2 concentration the respiratory quotient (RQ) can be calculated based on the Haldane transformation. However, these methods are not always available, time-consuming, expensive and often cumbersome in routine practice. VCO2-based calorimetry is an alternative non-invasive method for the calculation of energy expenditure. Normally VCO2 is measured continuously by most mechanical ventilators and is the starting point for energy measurement using a modified Weir formula [18, 19].
In recent years technological advancements contributed to the development of portable, easy to handle electronic motion detectors. One of them is the SenseWear® Pro Armband (SMT medical GmbH&Co. KG, Würzburg, Germany), a multidimensional device, which detects different physiological parameters of the human body by dedicated sensors. These include a continuous registration of skin temperature and galvanic skin resistance of heat flow. In addition a 2-axis accelerometer is localized in the armband, which records movements of the patient [20]. The SenseWear® Pro Armband is placed on the triceps of the right upper arm (Fig. 1). The upper arm allows for the best skin contact of the device and meets the requirements of the individual sensors. Once skin contact is achieved the armband automatically starts the measurement and stops with loss of skin contact [21].
Three different versions of the armband are commercially available: the SenseWear Pro Armband (sold from 2001 to 2003), the SenseWear Pro2 and SenseWear Pro3 Armband, for which a detailed assessment algorithm has not been published [20]. Due to its easy manageability and its capability for continuous data acquisition it was proposed to be suitable to determine resting energy expenditure in intensive care patients as well. SWA has shown promising results for calculating energy expenditure in non-critically ill patients [22–24] and various diseases such as obesity [25], cancer [26], COPD [27], cystic fibrosis [28], rheumatoid arthritis [29] or renal insufficiency [30]. Acceptable levels of agreement were also found in older individuals [31, 32]. Data for ICU patients are sparse and only one small study investigated patients under mechanical ventilation [33]. In this study we aimed to clarify the role of SenseWear® armband in critically ill patients on intensive care units and investigated whether SWA would be a suitable alternative for REE assessment. Therefore SenseWear Pro 3 Armband was compared to indirect calorimetry, the gold standard for REE measurement. Secondly we investigated the usability of predictive equations for REE by comparing to indirect calorimetry and SenseWear Armband.
2 Methods
34 patients from two intensive care units of the University Medicine Greifswald were recruited over a period of 7 month including both surgical and medical patients. Eligible were white Caucasians fulfilling the following criteria: (1) age >18 years, (2) ICU patient (3) and mechanical ventilation of at least 24 h. Patients without mechanical ventilation, pregnant women, patients under haemodialysis, patients with MRSA infection or FiO2 > 60 % were excluded. Severity of disease was estimated using the Simplified Acute Physiology Score (SAPS II) [34]. The investigation was approved by the ethics committee of the University of Greifswald and informed consent was obtained by proxy (BB 14/10).
2.1 Anthropometry, vital signs and nutrition status
The patients were studied by anthropometry (height, weight) and BMI (kilograms per height in meters squared). Heart rate, respiratory rate and body temperature were recorded as well. All patients received enteral, parenteral or combined enteral and parenteral nutrition. The difference between estimated REE by IC and effective daily energy intake was calculated (=energy balance). Subgroups were formed according to the energy balance (>−200, −200 to +200 and >+200 kcal/day).
2.2 Indirect calorimetry (IC)
REE was measured using the Deltatrac® TM U, MBM-200 system (Datex Ohmeda, Bremen, Germany). Before each measurement CO2 and O2 analyzers were calibrated with a standard gas mixture. The Deltatrac® device was connected to the ventilator in mechanically ventilated patients to calculate the energy expenditure and the measurements were performed for a minimum of 30 min in resting conditions. The first 5–10 min were discarded to achieve steady state (VO2 and VCO2 variation of ≤10 %/min). The ratio between volume of carbon dioxide and volume of oxygen is called respiratory quotient (RQ). Normal range of RQ was between 0.7 and 1.0.
The volumes of oxygen consumption and carbon dioxide production were determined continuously (both in l/min). By using the modified Weir formula the metabolic rate was estimated as follows:
REE (kcal/day) = [3.941 × VO2 (ml/min) + 1.1xVCO2 (ml/min)] × 1.440 [35].
2.3 SenseWear Pro 3 Armband (SWA)
The SWA was applied to the upper arm 10 min before indirect calorimetry started. This was done for each measurement by the same investigator. Over a 24 h period, patients wore the SWA continuously. During this period the nursing staff recorded factors that could affect energy consumption such as physical examination, washing, diagnostic work up and vegetative parameters such as sweating, increased or decreased body temperature. After 24 h the armband was removed and the data were evaluated with the SenseWear® software (lnnerview Professional Software 6.1 BodyMedia Inc., Pittsburgh, PA, USA). Energy consumption was calculated considering “age”, “size”, “weight”, “BMI” and “gender” according to the manufacturer’s protocol. Exact time for IC and measurement by SWA was recorded to rule out a detection bias.
2.4 Predictive equations
Many equations have been developed for estimating energy consumption in critically ill patients. There are two categories of equations: first, those using static variables like height, weight, age and sex and those that use dynamic variables which varies like body temperature, respiratory rate and tidal volume [36]. The equations that have been used in the present study are shown in Supplementary Table 1.
2.5 Statistical analysis
Data were analyzed using SPSS (Inc., Chicago, II, USA). Means of two methods were compared using the paired t test. Relationship between two parameters was performed by a simple Pearson correlation. Mann–Whitney U-test was used to test significant differences between mean values for two independent samples. For comparison of three groups and more, the Kruskal–Wallis test was used. Residual plots according to Bland and Altman were performed to compare two methods. A deviation of 10 % was determined to be acceptable (accuracy in %).
3 Results
3.1 Study population
34 patients were studied; among them 20 were medical and 14 surgical patients. Baseline characteristics of the participants are listed in Table 1. The mean age was 67 ± 12.4 years. Pulmonary disorders were the most frequent causes for ICU admission (29.4 %), followed by cardiovascular diseases and post-surgical surveillance (20.6 %). Disease severity was objectified using the SAPS II score that provides an estimation of the risk of death independently of a specific primary diagnosis [34]. Both groups showed similar mean SAPS II of around 32–34. The mean BMI was 27.6 ± 5.7 kg/m2. 44.1 % of patients had a normal weight and 55.9 % were overweight or obese. Mean daily energy intake was 1681 ± 333 kcal/day.
3.2 Correlation between estimated REE by IC and estimated REE by SWA
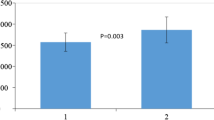
Energy consumption measured by IC was 1805.9 ± 413.6 and 1552.3 ± 284.9 kcal/day when measured by SWA. Results of IC and SWA showed a linear correlation (r = 0.598, p < 0.001, Fig. 2a) but also demonstrate that SWA underestimates REE compared to the gold standard. In addition root means squared error (RMSE) was 35.8 %, indicating still a poor quality of prediction. In order to evaluate measurement errors by SWA we performed a Bland–Altman analysis. Bias (ΔREE) was −253.6 ± 333.2 kcal/day, equivalent to −11.7 % (Fig. 2b). Bland–Altman plot also indicated that REE differences between indirect calorimetry and SenseWear armband are dependent on the amount of the patient’s energy intake: the higher the energy consumption, the larger differences were found between these methods.
3.3 Effects on measurement error
We were interested whether specific subgroups might predispose for a higher measurement error. Neither gender nor patient group (medical or surgical) showed any influence on REE measurement bias (Table 2). In females there was a trend towards higher deviations. Also, there weren’t any significant differences between individuals who underwent energy consuming interventions (physical therapy or diagnostic procedures) or those who were without interventions. The results indicate that energy balance appears to affect REE measurements. In the presence of a higher energy balances (the patient effectively received more calories than determined by indirect calorimetry) the differences between IC and SWA were greater. No significant differences were found in patients with an energy balance of less than −200 kcal (Supplementary Table 2).
When patients were analyzed according to their vital signs significant differences of REE measurements were observed irrespective of heartbeat and respiration rate. The discrepancy was greatest in tachycardic (>100 beats/min) and tachypnoeic patients (>30 breaths/min). Even in normofrequent patients (heart rate 70–99 beats/min) who account for the majority of the study group, bias was still around 15 % (Table 3). A significant discrepancy was also seen in patients with fever (>38 °C). In non-febrile ICU patients REE measurements by SWA and IC showed comparable results, with the highest concordance in individuals with normal body temperature (<37.4 °C).
3.4 Comparison of estimated REE by indirect calorimetry with predictive equations
We compared energy consumption determined by IC or SWA with frequently used estimation formulas. Except for the formula of Ireton Jones (1992), all equations underestimated the energy consumption compared to indirect calorimetry (Supplementary Table 3). The equation of Mifflin St Jeor showed the worst correlation with an average bias of −324 kcal/24 h (−15.5 %). Significant differences were also detected when REE was assessed by Harris–Benedict, Müller, Schofield, Ireton Jones (1992) and Swinamer. There was a good correlation between indirect calorimetry and estimated REE by Penn State, Ireton Jones (1997) and Fusco formulas.
Comparable results between REE values from SWA and predictive equations were only found for Harris–Benedict (ΔREE = +27.3 kcal/day; +2.7 %) and Schofield (ΔREE = −16.8 kcal/day; −1.1 %) formula. All other equations showed significantly different results. Greatest differences were recorded when using the Ireton Jones equation (1992) with an average deviation of −412 kcal/day (−27.3 %) (Supplementary Table 4).
Results of REE measurements by indirect calorimetry, SenseWear Armband and predictive equations are summed up in Table 4. A deviation of 10 % from REE by IC was considered to be acceptable. Formulas of Penn State and Fusco showed a relative good correlation to indirect calorimetry in 41.2 and 47.1 % of patients respectively.
4 Discussion
This study was designed to compare three REE assessment tools, indirect calorimetry, SenseWear® armband and predictive equations for their usability in ventilated intensive care patients. This group of patients is critically ill, needs close monitoring of vital functions and metabolism, and both over- and underfeeding should be avoided. Up till now indirect calorimetry is the gold standard for the estimation of energy expenditure but it is time consuming and cumbersome. Only one single study exists that compared IC and SWA in intensive care patients under mechanical ventilation for at least 24 h [33]. That cohort consisted of 23 patients and there was an underestimation of 6.8 % for daily energy expenditure when using SWA with a mean bias of 135.0 ± 417.2 kcal/day. In our study we found an even greater discrepancy of 11.7 % (−253 kcal/day), but a better correlation between the two methods (r = 0.598), possibly due to the higher number of patients included.
Measurement of REE by SenseWear® armband is a reliable method in healthy individuals. Malavolti and collaborators investigated normal-weight healthy subjects and found no systematic measurement error between SWA and IC [37]. The correlation factor was high with r = 0.86 (p < 0.0001) in this group. Similar results were obtained by Fruin and Rankin who additionally found that deviation of SWA from indirect calorimetry varies depending on the intensity of physical activity. While SenseWear Armband provides valid results for energy expenditure in resting subjects it overestimated flat walking activity by 13–27 % but underestimated inclined walking energy expenditure (by 22 %) [22]. Effects of exercise intensity on energy expenditure assessment by SWA and IC were further evaluated in cystic fibrosis patients compared to healthy adults who underwent treadmill walking [28]. There was an underestimation of energy consumption by SWA individuals with high physical activity but an overestimation at very low physical activity.
Our study demonstrated that SWA measurements in ICU patients generally underestimates REE irrespective of physical or diagnostic examinations (Table 2) or physiotherapy (data not shown). However it should be noted that any comparison of energy expenditures between healthy and physically active individuals and ICU patients, who are mostly confined to bed, is rather difficult. Quantification of physical activity in intensive care patients under mechanical ventilation is even more challenging. While SWA has been established as a valid tool for healthy and normal weight persons its usability has limitations for obese individuals: while SWA underestimates REE in obese people at rest it highly overestimates energy expenditure under physical exercise [25]. In cancer patients who are often confronted with nutritional problems and metabolic derangements the practicability of SWA compared to IC was also investigated. In a small study an acceptable accuracy for SWA-based energy expenditure measurement was found in patients with acute myelogenous leukemia undergoing induction chemotherapy. Bland–Altman analysis revealed no significant differences and correlation was high (r = 0.84, p < 0.0001) [26]. Further validation studies on larger cohorts and different tumor types will be necessary for a more conclusive evaluation of the usefulness of SWA. To the best of our knowledge this study is the first one that investigates measurement errors between SWA and IC in various subgroups of ICU patients. Differences were noted irrespective of sex, medical discipline (internal medicine, surgery) and deviations tended to be higher in females and medical patients. Moreover, our study considers other potential confounders such as heart and respiratory rate and body temperature.
4.1 Predictive equations
Caloric requirements for intensive care patients are often assessed by predictive equations. We compared REE calculations by a set of predictive formulas with both indirect calorimetry and SenseWear Armband. Only a few predictive equations are useable for critically ill and ventilated ICU patients and most consistent results were obtained with equations of Ireton-Jones (1997), Fusco and Penn State. Our observations are partly in line with the results from Frankenfield and co-workers who found an accurate assessment of the metabolic rate in critically ill patients with the Penn State equation, whereas the equation by Ireton-Jones turned out to be less accurate [36]. Predictive equations by Müller and Harris–Benedict have been derived from healthy subjects and may therefore be less reliable in critically ill and ventilated patients. Notably our study showed an underestimation of energy consumption by most equations implying a risk for underfeeding and malnutrition. Prediction accuracy of formulas has been reported with an error range of 7–55 % [38]. In addition, ICU patients may fluctuate in energy needs, which restrict the usability of predictive equations. In a recently published study REE estimated by Harris–Benedict equation (HBE) was compared to SWA measurements in postoperative patients with a good agreement between HBE and SWA (mean bias: −0.569 ± 0.378 kcal/24 h, underestimation of −2.9 %) [39]. When we compared REE results by SWA with predictive equations there was a strong correlation between SWA and the formulas of Harris–Benedict and Schofield. Since SWA underestimates energy needs when compared to IC, results from predictive equations were more frequently higher than SWA results. As recently published calculation of energy expenditure on only VCO2-based methods showed accurate results and were significantly better than predictive equations [19]. Using a fixed respiratory quotient of 0.81–0.89 energy expenditure was measured by the modified Weir equation that was between 1830 and 1973 kcal/day. These results were quite close to measured REE by indirect calorimetry (1805.9 ± 413.6 kcal/day).
There are limitations of our study. Since we included patients from medical and a surgical intensive care wards, the patient cohort was more heterogeneous than in other studies and a subgroup analysis more challenging. However, our results show that the measurement bias was quite similar the surgical and medical patients. This study was designed as a feasibility and pilot study for the applicability of a new device in intensive care units since no prior data were available to base a power calculation on. We intended to evaluate a measuring tool that is practicable and inexpensive and thus could be useful for primary care hospitals that usually are not equipped with indirect calorimetry and do not have separate intensive care wards for surgical or medical patients. For a definitive assessment of the role of SWA in daily practice on ICUs a confirmatory investigation would be warranted, which can base its predictions and algorithms on our data. Application of one easy-to-use method would be more convenient for routine clinical practice.
5 Conclusions
SenseWear Pro 3 Armband is not an appropriate alternative to indirect calorimetry in intensive care patients under mechanical ventilation because it underestimates REE. Individual measurements should be interpreted with caution. There is a necessity of an improved algorithm for REE assessment based on the data obtained in this study, which will ultimately allow a more accurate measurement of energy expenditure in critically ill patients using the device.
Abbreviations
- HBE:
-
Harris–Benedict equation
- IC:
-
Indirect calorimetry
- ICU:
-
Intensive care unit
- REE:
-
Resting energy expenditure
- RQ:
-
Respiratory quotient
- SWA:
-
SenseWear Pro 3 Armband
References
Weekes CE. Controversies in the determination of energy requirements. Proc Nutr Soc. 2007;66:367–77.
Giner M, Laviano A, Meguid MM, Gleason JR. In 1995 a correlation between malnutrition and poor outcome in critically ill patients still exists. Nutrition. 1996;12(1):23–9. doi:10.1016/0899-9007(95)00015-1.
Lerch MM, Braun J, Harder M, Hofstadter F, Schumpelick V, Matern S. Postoperative adaptation of the small intestine after total colectomy and J-pouch-anal anastomosis. Dis Colon Rectum. 1989;32(7):600–8.
Battezzati A, Vigano R. Indirect calorimetry and nutritional problems in clinical practice. Acta Diabetol. 2001;38(1):1–5.
McClave SA, Lowen CC, Kleber MJ, Nicholson JF, Jimmerson SC, McConnell JW, Jung LY. Are patients fed appropriately according to their caloric requirements? JPEN. 1998;22(6):375–81.
Villet S, Chiolero RL, Bollmann MD, Revelly JP, Cayeux RNM, Delarue J, Berger MM. Negative impact of hypocaloric feeding and energy balance on clinical outcome in ICU patients. Clin Nutr. 2005;24(4):502–9. doi:10.1016/j.clnu.2005.03.006.
Chima CS, Barco K, Dewitt ML, Maeda M, Teran JC, Mullen KD. Relationship of nutritional status to length of stay, hospital costs, and discharge status of patients hospitalized in the medicine service. J Am Diet Assoc. 1997;97(9):975–8. doi:10.1016/S0002-8223(97)00235-6 (quiz 979–980).
Reinhardt GF, Myscofski JW, Wilkens DB, Dobrin PB, Mangan JE Jr, Stannard RT. Incidence and mortality of hypoalbuminemic patients in hospitalized veterans. JPEN. 1980;4(4):357–9.
Robinson G, Goldstein M, Levine GM. Impact of nutritional status on DRG length of stay. JPEN. 1987;11(1):49–51.
Correia MI, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22(3):235–9. doi:10.1016/S0261-5614(02)00215-7.
Mechanick JI, Brett EM. Nutrition support of the chronically critically ill patient. Crit Care Clin. 2002;18(3):597–618.
Klein CJ, Stanek GS, Wiles CE 3rd. Overfeeding macronutrients to critically ill adults: metabolic complications. J Am Diet Assoc. 1998;98(7):795–806. doi:10.1016/S0002-8223(98)00179-5.
Plank LD, Hill GL. Energy balance in critical illness. Proc Nutr Soc. 2003;62(2):545–52.
Thibault R, Pichard C. Nutrition and clinical outcome in intensive care patients. Curr Opin Clin Nutr Metab Care. 2010;13(2):177–83. doi:10.1097/MCO.0b013e32833574b9.
Kan MN, Chang HH, Sheu WF, Cheng CH, Lee BJ, Huang YC. Estimation of energy requirements for mechanically ventilated, critically ill patients using nutritional status. Crit Care. 2003;7(5):R108–15. doi:10.1186/cc2366.
Pielmeier U, Rousing ML, Andreassen S. A model of changes in energy expenditure to specify daily caloric intake targets in sepsis and trauma patients. Intensive Care Med. 2014;40(Suppl. 1):83, no. 0279.
Haugen HA, Chan LN, Li F. Indirect calorimetry: a practical guide for clinicians. Nutr Clin Pract. 2007;22(4):377–88. doi:10.1177/0115426507022004377.
Stapel SN, de Grooth HJ, Alimohamad H, Elbers PW, Girbes AR, Weijs PJ, Oudemans-van Straaten HM. Ventilator-derived carbon dioxide production to assess energy expenditure in critically ill patients: proof of concept. Crit Care. 2015;19:370. doi:10.1186/s13054-015-1087-2.
Rousing ML, Hahn-Pedersen MH, Andreassen S, Pielmeier U, Preiser JC. Energy expenditure in critically ill patients estimated by population-based equations, indirect calorimetry and CO2-based indirect calorimetry. Ann Intensive Care. 2016;6(1):16. doi:10.1186/s13613-016-0118-8.
André D, Pelletier R, Farringdon J, Safier S, Talbott W, Stone R, Vyas N, Trimble J, Wolf D, Vishnubhatla S, Boehmke S, Stivoric J, Teller A. The development of the SenseWear® armband, a revolutionary energy assessment device to assess physical activity and lifestyle. Body Media Inc. 2006;1–19.
LaMonte MJ, Ainsworth BE, Reis JP. Measuring physical activity. In: Wood TM, Zhu W, editors. Measurement theory and practice in kinesiology. Leeds: Human Kinetics; 2006. p. 237–62.
Fruin ML, Rankin JW. Validity of a multi-sensor armband in estimating rest and exercise energy expenditure. Med Sci Sports Exerc. 2004;36(6):1063–9. doi:10.1249/01.MSS.0000128144.91337.38.
St-Onge M, Mignault D, Allison DB, Rabasa-Lhoret R. Evaluation of a portable device to measure daily energy expenditure in free-living adults. Am J Clin Nutr. 2007;85(3):742–9.
Bertoli S, Posata A, Battezzati A, Spadafranca A, Testolin G, Bedogni G. Poor agreement between a portable armband and indirect calorimetry in the assessment of resting energy expenditure. Clin Nutr. 2008;27(2):307–10. doi:10.1016/j.clnu.2007.11.005.
Papazoglou D, Augello G, Tagliaferri M, Savia G, Marzullo P, Maltezos E, Liuzzi A. Evaluation of a multisensor armband in estimating energy expenditure in obese individuals. Obesity (Silver Spring). 2006;14(12):2217–23. doi:10.1038/oby.2006.260.
Cereda E, Turrini M, Ciapanna D, Marbello L, Pietrobelli A, Corradi E. Assessing energy expenditure in cancer patients: a pilot validation of a new wearable device. JPEN. 2007;31(6):502–7. doi:10.1177/0148607107031006502.
Patel SA, Benzo RP, Slivka WA, Sciurba FC. Activity monitoring and energy expenditure in COPD patients: a validation study. COPD. 2007;4(2):107–12. doi:10.1080/15412550701246658.
Dwyer TJAJA, McKeough ZJ, Elkins MR, Bye PTP. Evaluation of the SenseWear activity monitor during exercise in cystic fibrosis and in health. Respir Med. 2009;103:1511–7.
Tierney M, Fraser A, Purtill H, Kennedy N. Study to determine the criterion validity of the SenseWear Armband as a measure of physical activity in people with rheumatoid arthritis. Arthritis Care Res (Hoboken). 2013;65(6):888–95. doi:10.1002/acr.21914.
Avesani CM, Trolonge S, Deleaval P, Baria F, Mafra D, Faxen-Irving G, Chauveau P, Teta D, Kamimura MA, Cuppari L, Chan M, Heimburger O, Fouque D. Physical activity and energy expenditure in haemodialysis patients: an international survey. Nephrol Dial Transplant. 2012;27(6):2430–4. doi:10.1093/ndt/gfr692.
Mackey DC, Manini TM, Schoeller DA, Koster A, Glynn NW, Goodpaster BH, Satterfield S, Newman AB, Harris TB, Cummings SR. Validation of an armband to measure daily energy expenditure in older adults. J Gerontol A Biol Sci Med Sci. 2011;66(10):1108–13. doi:10.1093/gerona/glr101.
Heiermann S, Khalaj Hedayati K, Muller MJ, Dittmar M. Accuracy of a portable multisensor body monitor for predicting resting energy expenditure in older people: a comparison with indirect calorimetry. Gerontology. 2010;57(5):473–9. doi:10.1159/000322109.
Rokuss K, Kalenka A, Bender HJ, Hinkelbein J. Intensive care patients. Determining daily energy expenditure—a comparison of two methods. Anaesthesist. 2009;58(8):787–94. doi:10.1007/s00101-009-1552-x.
Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;270(24):2957–63.
Weir JB. New methods for calculating metabolic rate with special reference to protein metabolism. J Physiol. 1949;109(1–2):1–9.
Frankenfield DC, Coleman A, Alam S, Cooney RN. Analysis of estimation methods for resting metabolic rate in critically ill adults. JPEN. 2009;33(1):27–36. doi:10.1177/0148607108322399.
Malavolti M, Pietrobelli A, Dugoni M, Poli M, Romagnoli E, De Cristofaro P, Battistini NC. A new device for measuring resting energy expenditure (REE) in healthy subjects. Nutr Metab Cardiovasc Dis. 2007;17(5):338–43. doi:10.1016/j.numecd.2005.12.009.
Walker RN, Heuberger RA. Predictive equations for energy needs for the critically ill. Respir Care. 2009;54(4):509–21.
Dummler R, Zittermann A, Schafer M, Emmerich M. Postoperative assessment of daily energy expenditure. Comparison of two methods. Anaesthesist. 2013;62(1):20–6. doi:10.1007/s00101-012-2120-3.
Funding
JK, SG and LJV received a Gerhard-Domagk-Scholarship from University Medicine Greifswald made possible through unrestricted educational grants from Nutricia GmbH (Erlangen, Germany), Baxter Deutschland GmbH (Unterschleissheim, Germany) and Gesundheits GmbH (Ahrensburg).
Author’s contribution
JK and MK are equal contributors. JK, MK, MML and AAA concepted and designed the study and wrote the manuscript. All authors substantially contributed to conception, design, acquisition and analysis of data and interpretation of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare to have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Krüger, J., Kraft, M., Gründling, M. et al. Evaluation of a non-invasive multisensor accelerometer for calculating energy expenditure in ventilated intensive care patients compared to indirect calorimetry and predictive equations. J Clin Monit Comput 31, 1009–1017 (2017). https://doi.org/10.1007/s10877-016-9934-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-016-9934-5