Abstract
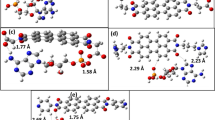
The 2D hybrid compound bis(2-amino-4-methoxy-6-methylpyrimidinium) bis(μ2-chloro)-tetrachloro-di-copper(II), (C6H10N3O)2Cu2Cl6, is successfully synthesized by slow solvent evaporation at room temperature. Structural properties have been investigated through single-crystal X-ray diffraction and reveal that the structure contains a centrosymmetric hexachlorodicuprate group where each Cu atom is coordinated to four Cl atoms in a slightly distorted square planar geometry. There are short contacts between neighboring [Cu2Cl6]2− dimer units. The crystalline building stability is ensured by N–H⋯Cl and C–H⋯O hydrogen bonding as well as weak C–H···π intermolecular interactions. From the infrared spectroscopy analysis, the functional groups were identified. Simultaneously, the electrical properties and Hirshfeld surface analyses were also elucidated. Furthermore, the molecular docking study of 2D hybrid compound bis(2-amino-4-methoxy-6-methylpyrimidinium) bis(2-chloro)-tetrachloro-di-copper(II) ligand with an HSP90/PDB: 5LRZ was performed by Autodock Vina. Additionally, drug-likeness and ADME properties and evaluations of the newly synthesized molecule were performed in detail. FT-IR was used to explore the modes of vibration of the different functional groups present in the studied compound.










Similar content being viewed by others
Data Availability
All data are available within the manuscript and associated supporting information file.
References
F. Girardi, E. Cappelletto, J. Sandak, G. Bochicchio, B. Tessadri, S. Palanti, E. Feci, and R. D. Maggio (2014). Prog. Org. Coat. 77, 449–457.
M. Szafranski (2016). Cryst. Growth Des. 16, 3771–3776.
H. B. Duan, X. M. Ren, and Q. J. Meng (2010). Coord. Chem. Rev. 254, 1509–1522.
C. Bellitto, E. M. Bauer, and G. Righini (2015). Coord. Chem. Rev. 289–290, 123–136.
J. M. Lei, Q. X. Peng, S. P. Luo, Y. Liu, S. Z. Zhan, and C. L. Ni (2018). Mol. Catal. 448, 10–17.
K. Pradeesh, M. Agarwal, K. K. Rao, and G. Vijaya Prakash (2010). Solid State Sci. 12, 95–98.
Q. Chen, N. De Marco, Y. Yang, T.-B. Song, C.-C. Chen, H. Zhao, Z. Hong, H. Zhou, and Y. Yang (2015). Nano Today 10, 355–396.
Z. Czapla, Yu. Eliyashevskyy, and S. Dacko (2006). Ferroelectr. Lett. Sect. 33, 1–6.
A. O. Polyakov, A. H. Arkenbout, J. Baas, G. R. Blake, A. Meetsma, A. Caretta, and T. T. M. Palstra (2011). Chem. Mater. 24, 133–139.
C. Jiang, Q. Luo, H. Fu, H. Lin, C. Luo, J. Wang, X. Meng, H. Peng, C.-G. Duanb, and J. Chu (2020). CrystEngComm 22, 587–592.
C. Peng (2011). Acta Cryst. E67.
S. Bouacida, R. Bouchene, A. Khadri, R. Belhouas, and H. Merazig (2013). Acta Crystallogr. Sect. E 69, 610.
A. Kessentini, M. Belhouchet, J. J. Suñol, Y. Abid, and T. Mhiri (2015). Spectrochim. Acta A 134, 28–33.
P. Arularasan, B. Sivakumar, G. Chakkaravarthi, and R. Mohana (2013). Acta Cryst. E 69, 583.
T. Dammak, H. Boughzala, A. Mlayah, and Y. Abid (2016). J. Lumin. 173, 213–217.
N. Karaa, B. Hamdi, A. Oueslati, A. Ben Salah, and R. Zouari (2010). J. Inorg. Organomet. Polym. Mater. 20, 746–754.
A. K. Vishwakarma, R. Kumari, P. S. Ghalsasi, and N. Arulsamy (2017). J. Mol. Struct. 1141, 93–98.
S. Walha, H. Naïli, S. Yahyaoui, B. Fares Ali, M. M. Turnbull, T. Mhiri, and S. W. Ng (2013). J. Supercond. Nov. Magn. 26, 437–442.
A. K. Vishwakarma, P. S. Ghalsasi, A. Navamoney, Y. Lan, and A. K. Powell (2011). Polyhedron 30, 1565–1570.
W. E. Hunt, C. H. Schwalbe, K. Bird, and P. D. Mallinson (1980). Biochem. J. 187, 533–536. https://doi.org/10.1042/bj1870533.
M. Jeevaraj, P. Sivajeyanthi, B. Edison, K. Thanigaimani, and K. Balasubramani (2018). Acta Crystallogr. Sect. E Crystallogr. Commun. 74, 656–659.
N. V. Rao, B. Vaizalini, B. Mounika, L. V. Harika, P. K. Desu, and S. Nama (2013). J. Pharm. Chem. Res. 2, 14–22.
Z. Afroz, M. Faizan, M. J. Alam, V. H. N. Rodrigues, S. Ahmad, and A. Ahmad (2018). J. Mol. Struct. 1171, 438–448.
E. A. Alodeani, M. A. Izhari, and M. Arshad (2014). Eur. J. Biomed. Pharm. Sci. 1, 504–527.
S. Q. Wang, L. Fang, X. J. Liu, and K. Zhao (2004). Chin. Chem. Lett. 15, 885–888.
V. Yerragunta, P. Patil, V. Anusha, T. Kumaraswamy, D. Suman, and T. Samhitha (2013). PharmaTutor 1, 39–44.
M. Kurono, M. Hayashi, K. Miura, Y. Isogawa, and K. Sawai (1988). Chem. Abstr. 109, 37832.
S. N. Pandeya, D. Sriram, G. Nath, and E. D. Clercq (1999). Farmaco 54, 624–628.
P. Sharma, N. Rane, and V. K. Gurram (2004). Bioorg. Med. Chem. Lett. 14, 4185–4190.
E. Wagner, K. A. Kadasi, M. Zimecki, and W. S. Dobrowolska (2008). Eur. J. Med. Chem. 43, 2498–2504.
T. P. Selvam, C. R. James, P. V. Dniandev, and S. K. Valzita (2012). Res. Pharm. 2, 01–09.
M. Asif (2014). SOP Trans. Org. Chem. 1, 1–10.
S. Lahmidi, E. H. Anouar, M. El Hafi, M. Boulhaoua, A. Ejjoumamany, M. El Jemli, E. M. Essassi, and J. T. Mague (2019). J. Mol. Struct. 1177, 131–142.
Stoe and Cie, X-RED and X-AREA (Stoe and Cie, Darmstadt, 2009).
G. M. Sheldrick (2008). Acta Crystallogr. Sect. A 64, 112–122.
G. M. Sheldrick (2015). Acta Cryst. C 71, 3–8.
J. J. McKinnon, M. A. Spackman, and A. S. Mitchell (2004). Acta Cryst. B 60, 627–668.
J. J. McKinnon, D. Jayatilaka, and M. A. Spackman (2007). Chem. Commun. 37, 3814–3816.
M. A. Spackman and J. J. McKinnon (2002). CrystEngComm 4, 378–392.
M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, P. R. Spackman, D. Jayatilaka, and M. A. Spackman. CrystalExplorer17 (University of Western Australia, 2017).
R. Dennington, T. Keith, and J. Millam, GaussView, Version 5 (Semichem Inc., Shawnee Mission, 2009).
M. J. Frisch, G. Trucks, H. Schlegel, G. Scuseria, M. Robb, J. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, and G. Petersson, Gaussian 09, Revision D. 01 (Gaussian Inc., Wallingford, 2009).
O. Trott and A. J. Olson (2010). J. Comput. Chem. 31, 455–461.
A. Daina, O. Michielin, and V. Zoete (2017). Sci. Rep. 7, 42717.
B. Bremer and A. A. Pinkerton (1990). Inorg. Chim. Acta 174, 21–32.
G. Hu and E. M. Holt (1994). Acta Cryst. C 50, 1212–1214.
V. V. Sharutin, V. S. Senchurin, O. K. Sharutina, A. P. Pakusina, and O. A. Fastovets (2010). Russ. J. Inorg. Chem. 55, 1415–1420.
J. Dai and J. Xu (2011). Acta Cryst. E67.
L. Yang, D. R. Powell, and R. P. Houser (2007). Dalton Trans. 9, 955–964.
R. D. Willett (1966). J. Chem. Phys. 44, 39.
R. L. Harlow, W. J. Wells, G. W. Watt, and S. H. Simonsen (1974). Inorg. Chem. 13, 2860–2863.
K. Kaabi, M. El Glaoui, V. Ferretti, M. Zeller, and C. Ben Nasr (2011). Acta Cryst. E 67, o2507–o2508.
S. Suganya, A. Karthikeyan, V. Srimathi, and P. Kumaradhas (2020). Chem. Data Collect. 29.
E. Jaziri, L. Khedhiri, S. Soudani, V. Ferretti, F. Lefebvre, W. Fujita, and C. Ben Nasr (2021). J. Clust. Sci. 32, 145–153.
M. Jeevaraj, P. Sivajeyanthi, B. Edison, K. Thanigaimani, and K. Balasubramani (2018). Acta Cryst. E 74, 656–659.
Fleming, Frontier Orbitals and Organic Chemical Reactions. (Wiley, New York, 1976), pp. 5–27.
M. D. Diener and J. M. Alford (1998). Nature 393, 668–671.
E. Scrocco and J. Tomasi (1978). Adv. Quantum Chem. 11, 115–193.
F. J. Luque, J. M. Lopez, and M. Orozco (2000). Theor. Chem. Acc. 103, 343–345.
B. J. McConkey, V. Sobolev, and M. Edelman (2002). Curr. Sci. 83, 845–856.
R. R. Nadendla (2004). Resonance 9, 51–60.
X.-Y. Meng, H.-X. Zhang, M. Mezei, and M. Cui (2011). Curr. Comput. Aided Drug Des. 7, 146–157.
J. Polanski (2009). Curr. Med. Chem. 16, 3243–3257.
A. Mettu, V. Talla, D. M. Bajaj, and N. J. P. Subhashini (2019). Arch. Pharm. 352, 1900063.
C. A. Lipinski, F. Lombardo, B. W. Dominy, and P. J. Feeney (1997). Adv. Drug Deliv. Rev. 23, 3–25.
A. Daina, O. Michielin, and V. Zoete (2017). Sci. Rep. 7, 1–13.
A. Daina and V. Zoete (2016). Chem. Med. Chem. 11, 1117–1121.
Acknowledgements
The authors especially thanks to Prof. Dr. Fatih UCUN from the Suleyman Demirel University for his helpful contribution for Gaussian calculations.
Funding
Not funded.
Author information
Authors and Affiliations
Contributions
CBN and LK: Conceptualization, design, resources, editing and supervision. CG: Synthesis, Analysis, writing and manuscript draft. UB: X-ray data collections and structure resolution. YS, EBÇ and ND: DFT calculations, writing and proofreading.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare zero conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gharbi, C., Sert, Y., Çınar, E.B. et al. Synthesis, Crystal Structure, Hirshfeld Surface Analysis, Molecular Docking, IR Spectroscopy and DFT Calculations of a Novel 2D Layered Hybrid Compound (C6H10N3O)2Cu2Cl6. J Clust Sci 34, 1423–1435 (2023). https://doi.org/10.1007/s10876-022-02314-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-022-02314-6