Abstract
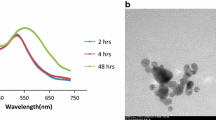
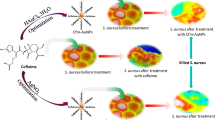
In the present study, we report a simple, robust, and eco-friendly one pot synthesis of gentamicin conjugated gold nanoparticles (G-GNPs), where gentamicin behaves dually as a reducing as well as a stabilizing agent. The resultant nanoparticles were characterized through different microscopic and spectroscopic techniques and found to be almost spherical in shape with hydrodynamic diameter of ~ 15 nm along with excellent stability. The antibacterial potential was evaluated by well diffusion assay and showed that G-GNPs effectively inhibit the growth of gram-positive and gram-negative bacteria viz. Escherichia coli DH5α, Escherichia coli ATCC 25922 and Staphylococcus aureus MTCC 3160 (p < 0.05). Results also revealed that G-GNPs exhibit excellent antibacterial activity as compared with pure gentamicin, interestingly G-GNPs also showed excellent activity against gentamicin resistant Escherichia fergusonii ATCC 35469. A sustained release of gentamicin molecules from nanoparticles was observed. Furthermore, when we tested the effect of G-GNPs on mouse myoblast C2C12 cell line, G-GNPs exhibited minimal cytotoxicity. Consequently, the developed G-GNPs can be considered as safe based on minimal cytotoxicity of G-GNPs, and hold a great potential against gram-positive, gram-negative and drug resistance bacteria.





Similar content being viewed by others
References
P. V. Baptistal, M. P. McCusker, A. Carvalho, D. A. Ferreira, N. M. Mohan, M. Martins, and A. R. Fernandes (2018). Front. Microbiol. 9, 1441.
R. S. McInnes, G. E. McCallum, L. E. Lamberte, and W. van Schaik (2020). Curr. Opin. Microbiol. 53, 35.
World Health Organisation. Antibiotic resistance. 2018.
J. M. Munita and C. A. Arias (2016). Microbiol. Spectr. 4, 1.
J. J. Farmer, G. R. Fanning, B. R. Davis, C. M. O’hara, C. Riddle, F. W. Hickman-Brenner, M. A. Asbury, V. A. Lowery, and D. J. Brenner (1985). J. Clin. Microbiol. 21, 77.
T. Adesina, O. Nwinyi, N. De, O. Akinnola, and E. Omonigbehin (2019). Pathogens. 8, 164.
S. Onoue, S. Yamada, and H. K. Chan (2014). Int. J. Nanomed. 9, 1025.
M. Salouti, Z. Heidari, A. Ahangari, and S. Zare (2016). Drug Deliv. 23, 49.
A. Ahangari, M. Salouti, Z. Heidari, A. R. Kazemizadeh, and A. A. Safari (2013). Drug Deliv. 20, 34.
Clinical and Laboratory Standards Institute (CLSI) (2012). CLSI Document M07-A9. Approved Standard. 9th Edition by Wayne, P. A.
H. Sim Choi, J. Woo Kim, Y. N. Cha, and C. Kim (2006). J. Immunoassay Immunochem. 27, 31.
R. S. Thombre, V. Shinde, E. Thaiparambil, S. Zende, and S. Mehta (2016). Front. Microbiol. 7, 1424.
Y. Zhao, Y. Tian, Y. Cui, W. Liu, W. Ma, and X. Jiang (2010). J. Am. Chem. Soc. 132, 12349.
S. Perni and P. Prokopovich (2014). RSC Advances. 4, 51904.
A. Bahuguna, I. Khan, V. K. Bajpai, and S. C. Kang (2017). Bangladesh J. Pharmacol. 12, 8.
J. D. S. Newman and G. J. Blanchard (2006). Langmuir. 22, 5882.
T. Junqi and M. Shiqing (2013). Rare Metal Mat. Eng. 42, 2232.
W. Haiss, N. T. K. Thanh, J. Aveyard, and D. G. Fernig (2007). Anal. Chem. 79, 4215.
P. Yang, P. Pageni, M. A. Rahman, M. Bam, T. Zhu, Y. P. Chen, M. Nagarkatti, A. W. Decho, and C. Tang (2019). Adv. Healthc. Mater. 8, 1800854.
T. P. Shareena Dasari, Y. Zhang, and H. Yu (2015). Biochem. Pharmacol. 4.
M. Y. Memar, R. Ghotaslou, M. Samiei, and K. Adibkia (2018). Infect. Drug Resist. 11, 567.
V. Tiwari, N. Mishra, K. Gadani, P. S. Solanki, N. A. Shah, and M. Tiwari (2018). Front. Microbiol. 9, 1218.
J. N. Payne, H. K. Waghwani, M. G. Connor, W. Hamilton, S. Tockstein, H. Moolani, F. Chavda, V. Badwaik, M. B. Lawrenz, and R. Dakshinamurthy (2016). Front. Microbiol. 7, 607.
R. Shukla, V. Bansal, M. Chaudhary, A. Basu, R. R. Bhonde, and M. Sastry (2005). Langmuir. 21, 10644.
X. Li, S. M. Robinson, A. Gupta, K. Saha, Z. Jiang, D. F. Moyano, A. S. M. A. Riley, and V. M. Rotello (2014). ACS Nano. 8, 10682.
M. G. Ma in V. Basiuk and E. Basiuk (eds.), Green Processes for Nanotechnology (Springer, Cham, 2015), p. 119.
Acknowledgements
The work carried out in the present manuscript is supported by Jaypee University of Information Technology, Solan, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
There are no conflicts between the authors to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, D., Chaudhary, A. One Pot Synthesis of Gentamicin Conjugated Gold Nanoparticles as an Efficient Antibacterial Agent. J Clust Sci 32, 995–1002 (2021). https://doi.org/10.1007/s10876-020-01864-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-020-01864-x