Abstract
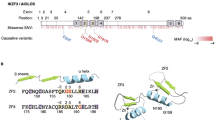
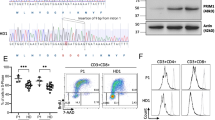
The DNA polymerase δ complex (PolD), comprising catalytic subunit POLD1 and accessory subunits POLD2, POLD3, and POLD4, is essential for DNA synthesis and is central to genome integrity. We identified, by whole exome sequencing, a homozygous missense mutation (c.1118A > C; p.K373T) in POLD3 in a patient with Omenn syndrome. The patient exhibited severely decreased numbers of naïve T cells associated with a restricted T-cell receptor repertoire and a defect in the early stages of TCR recombination. The patient received hematopoietic stem cell transplantation at age 6 months. He manifested progressive neurological regression and ultimately died at age 4 years. We performed molecular and functional analysis of the mutant POLD3 and assessed cell cycle progression as well as replication-associated DNA damage. Patient fibroblasts showed a marked defect in S-phase entry and an enhanced number of double-stranded DNA break-associated foci despite normal expression levels of PolD components. The cell cycle defect was rescued by transduction with WT POLD3. This study validates autosomal recessive POLD3 deficiency as a novel cause of profound T-cell deficiency and Omenn syndrome.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CADD:
-
Combined annotation dependent depletion
- DSB:
-
Double strand breaks
- HSCT:
-
Hematopoietic stem cell transplant
- IEI:
-
Inborn error of immunity
- IgH:
-
Immunoglobulin heavy chain
- NHEJ:
-
Non-homologous end joining
- PCNA:
-
Proliferating cell nuclear antigen
- PolD:
-
Polymerase delta complex
- Polζ:
-
Polymerase zeta complex
- TLS:
-
Translesion synthesis
- TRB:
-
T cell receptor B locus
- WES:
-
Whole exome sequencing
References
Loeb LA, Monnat RJ. DNA polymerases and human disease. Nat Rev Genet. 2008;9(8):594–604.
Johnson Robert E, Klassen R, Prakash L, Prakash S. A major role of DNA polymerase δ in replication of both the leading and lagging DNA strands. Mol Cell. 2015;59(2):163–75.
Xie B, Mazloum N, Liu L, Rahmeh A, Li H, Lee MYWT. Reconstitution and characterization of the human DNA polymerase delta four-subunit holoenzyme. Biochemistry. 2002;41(44):13133–42.
Donnianni RA, Zhou Z-X, Lujan SA, Al-Zain A, Garcia V, Glancy E, et al. DNA polymerase delta synthesizes both strands during break-induced replication. Mol Cell. 2019;76(3):371-81.e4.
Hirota K, Yoshikiyo K, Guilbaud G, Tsurimoto T, Murai J, Tsuda M, et al. The POLD3 subunit of DNA polymerase δ can promote translesion synthesis independently of DNA polymerase ζ. Nucleic Acids Res. 2015;43(3):1671–83.
Layer JV, Debaize L, Van Scoyk A, House NC, Brown AJ, Liu Y, et al. Polymerase δ promotes chromosomal rearrangements and imprecise double-strand break repair. Proc Natl Acad Sci. 2020;117(44):27566–77.
Maloisel L, Fabre F, Gangloff S. DNA polymerase δ is preferentially recruited during homologous recombination to promote heteroduplex DNA extension. Mol Cell Biol. 2008;28(4):1373–82.
Ducoux M, Urbach S, Baldacci G, Hübscher U, Koundrioukoff S, Christensen J, et al. Mediation of proliferating cell nuclear antigen (PCNA)-dependent DNA replication through a conserved p21Cip1-like PCNA-binding motif present in the third subunit of human DNA polymerase δ*. J Biol Chem. 2001;276(52):49258–66.
Lee Y-S, Gregory MT, Yang W. Human Pol ζ purified with accessory subunits is active in translesion DNA synthesis and complements Pol η in cisplatin bypass. Proc Natl Acad Sci. 2014;111(8):2954–9.
Bellido F, Pineda M, Aiza G, Valdés-Mas R, Navarro M, Puente DA, et al. POLE and POLD1 mutations in 529 kindred with familial colorectal cancer and/or polyposis: review of reported cases and recommendations for genetic testing and surveillance. Genet Med. 2016;18(4):325–32.
Palles C, Cazier J-B, Howarth KM, Domingo E, Jones AM, Broderick P, et al. Germline mutations affecting the proofreading domains of POLE and POLD1 predispose to colorectal adenomas and carcinomas. Nat Genet. 2013;45(2):136–44.
Elouej S, Beleza-Meireles A, Caswell R, Colclough K, Ellard S, Desvignes JP, et al. Exome sequencing reveals a de novo POLD1 mutation causing phenotypic variability in mandibular hypoplasia, deafness, progeroid features, and lipodystrophy syndrome (MDPL). Metabolism. 2017;71:213–25.
Pelosini C, Martinelli S, Ceccarini G, Magno S, Barone I, Basolo A, et al. Identification of a novel mutation in the polymerase delta 1 (POLD1) gene in a lipodystrophic patient affected by mandibular hypoplasia, deafness, progeroid features (MDPL) syndrome. Metabolism. 2014;63(11):1385–9.
Weedon MN, Ellard S, Prindle MJ, Caswell R, Allen HL, Oram R, et al. An in-frame deletion at the polymerase active site of POLD1 causes a multisystem disorder with lipodystrophy. Nat Genet. 2013;45(8):947–50.
Conde CD, Petronczki ÖY, Baris S, Willmann KL, Girardi E, Salzer E, et al. Polymerase δ deficiency causes syndromic immunodeficiency with replicative stress. J Clin Investig. 2019;129(10):4194–206.
Cui Y, Keles S, Charbonnier L-M, Julé AM, Henderson L, Celik SC, et al. Combined immunodeficiency caused by a loss-of-function mutation in DNA polymerase delta 1. J Allergy Clin Immunol. 2020;145(1):391-401.e8.
Nichols-Vinueza DX, Delmonte OM, Bundy V, Bosticardo M, Zimmermann MT, Dsouza NR, et al. POLD1 deficiency reveals a role for POLD1 in DNA repair and T and B cell development. J Clin Immunol. 2021;41(1):270–3.
Strobl J, Huber B, Heredia RJ, Kirnbauer R, Boztug K, Stary G. Polymerase-δ-deficiency as a novel cause of inborn cancer predisposition associated with human papillomavirus infection. Br J Dermatol. 2023;188(5):684–5.
Stirling DR, Swain-Bowden MJ, Lucas AM, Carpenter AE, Cimini BA, Goodman A. Cell Profiler 4: improvements in speed, utility and usability. BMC Bioinformatics. 2021;22(1):433.
Villarese P, Abdo C, Bertrand M, Thonier F, Giraud M, Salson M, Macintyre E. One-Step next-generation sequencing of immunoglobulin and t-cell receptor gene recombinations for mrd marker identification in acute lymphoblastic leukemia. Methods Mol Biol. 2022;2453:43–59.
Brüggemann M, Kotrová M, Knecht H, Bartram J, Boudjogrha M, Bystry V, et al. Standardized next-generation sequencing of immunoglobulin and T-cell receptor gene recombinations for MRD marker identification in acute lymphoblastic leukaemia; a EuroClonality-NGS validation study. Leukemia. 2019;33(9):2241–53.
Giraud M, Salson M, Duez M, Villenet C, Quief S, Caillault A, et al. Fast multiclonal clusterization of V(D)J recombinations from high-throughput sequencing. BMC Genomics. 2014;15(1):409.
Gu Z, Gu L, Eils R, Schlesner M, Brors B. circlize implements and enhances circular visualization in R. Bioinformatics. 2014;30(19):2811–2.
Dvorak CC, Haddad E, Heimall J, Dunn E, Buckley RH, Kohn DB, et al. The diagnosis of severe combined immunodeficiency (SCID): the primary immune deficiency treatment consortium (PIDTC) 2022 definitions. J Allergy Clin Immunol. 2023;151(2):539–46.
Tangye SG, Al-Herz W, Bousfiha A, Cunningham-Rundles C, Franco JL, Holland SM, et al. Human inborn errors of immunity: 2022 update on the classification from the international union of immunological societies expert committee. J Clin Immunol. 2022;42(7):1473–507.
Berland A, Rosain J, Kaltenbach S, Allain V, Mahlaoui N, Melki I, et al. PROMIDISα: a T-cell receptor α signature associated with immunodeficiencies caused by V(D)J recombination defects. J Allergy Clin Immunol. 2019;143(1):325-34.e2.
Frugoni F, Dobbs K, Felgentreff K, Aldhekri H, Al Saud BK, Arnaout R, et al. A novel mutation in the POLE2 gene causing combined immunodeficiency. J Allergy Clin Immunol. 2016;137(2):635-8.e1.
Pachlopnik Schmid J, Lemoine R, Nehme N, Cormier-Daire V, Revy P, Debeurme F, et al. Polymerase ε1 mutation in a human syndrome with facial dysmorphism, immunodeficiency, livedo, and short stature (“FILS syndrome”). J Exp Med. 2012;209(13):2323–30.
Murga M, Lecona E, Kamileri I, Díaz M, Lugli N, Sotiriou SK, et al. POLD3 is haploinsufficient for DNA replication in mice. Mol Cell. 2016;63(5):877–83.
Ogi T, Limsirichaikul S, Overmeer RM, Volker M, Takenaka K, Cloney R, et al. Three DNA polymerases, recruited by different mechanisms, carry out NER repair synthesis in human cells. Mol Cell. 2010;37(5):714–27.
Richards F, Llorca-Cardenosa MJ, Langton J, Buch-Larsen SC, Shamkhi NF, Sharma AB, et al. Regulation of Rad52-dependent replication fork recovery through serine ADP-ribosylation of PolD3. Nat Commun. 2023;14(1):4310.
Mehawej C, Chouery E, Azar-Atallah S, Shebaby W, Delague V, Mansour I, et al. POLD3 deficiency is associated with severe combined immunodeficiency, neurodevelopmental delay, and hearing impairment. Clin Immunol. 2023;251:109326.
Acknowledgements
We acknowledge the Centre for Multimodal Oncology Research (C-MORE) at KU Leuven for the use of the ImageStream.We thank Dr Rebeca Perez-De Diego for fruitful discussion. We thank the parents of the patient for their trust.
Funding
This work was supported by UZ Leuven Klinische Onderzoeksraad Funding – KOOR. MRR was supported by an ESID fellowship. LE is supported by a PhD Fellowship from the Research Foundation – Flanders (FWO) (grant 11E0123N). SD is supported by a PhD Fellowship from the Research Foundation – Flanders (FWO) (grant 11F4421N). LDN is supported by the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health (grant AI001222). IM is supported by the Jeffrey Modell Foundation.
Author information
Authors and Affiliations
Contributions
Experiments, data collection, and analysis were performed by Maria Rodrigo Riestra, Bethany Pillay, Giorgia Bucciol, Xavier Bossuyt, Kate Sauer, and Leen Moens with assistance from Mathijs Willemsen, Verena Kienapfel, Lisa Ehlers, Selket Delafontaine, Marjon Wouters, Anneleen Hombrouck, Stephanie Humblet-Baron, Frédéric Rieux-Laucat, Cecilia Dominguez Conde, Kaan Boztug, and Luigi D. Notarangelo. Antoine Pinton and Aurore Touzart analyzed TRB and IgH data. Arnout Voet provided structural data. Stefaan J Soenen provided Imagestream facilities. Isabelle Meyts conceptualized and funded the study. The manuscript was written by Maria Rodrigo Riestra, Bethany Pillay, Giorgia Bucciol, and Isabelle Meyts, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The Ethics Committee of the University Hospitals Leuven (UZLeuven) approved this study (S66910, S63807). The study was performed in compliance with the Declaration of Helsinki for research involving human subjects.
Consent to Participate
Written informed consent was obtained from the parents prior to DNA, PBMCs, and fibroblast isolation.
Consent for Publication
Informed consent was provided by the parents for the publication of the images in Fig. 1a and b.
Competing Interests
IM receives funding from CSL Behring and consultancy fees from Boehringer-Ingelheim, unrelated to this submission, paid to KU Leuven.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Riestra, M.R., Pillay, B.A., Willemsen, M. et al. Human Autosomal Recessive DNA Polymerase Delta 3 Deficiency Presenting as Omenn Syndrome. J Clin Immunol 44, 2 (2024). https://doi.org/10.1007/s10875-023-01627-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10875-023-01627-z