Abstract
Euphausiids are a vital component of global marine micronekton. To reveal the primary environmental factors influencing euphausiid distribution patterns in the previously overlooked low-latitude ecosystems, we investigated a large-scale community structure of euphausiids covering the North Pacific subtropical gyre (NPSG) and low-latitude eastern Indian (EI), and South Pacific Oceans (SP). A total of 41 euphausiid species from six genera were identified. Integrated primary production (PP) correlated significantly with the euphausiid abundance and species diversity and displayed the most critical influence on the variations in euphausiid community structure in low latitudes. Dissolved oxygen (DO) was the second significant environmental driver. Due to the distinct distribution patterns of euphausiid species in response to different PP and DO levels, the low-latitude euphausiid assemblages were mainly distinguished into subtropical and tropical communities. The subtropical euphausiid community associated with lower PP and higher DO demonstrated significantly lower euphausiid abundance but higher diversity than the tropical community. Euphausia brevis, E. mutica, and Stylocheiron abbreviatum, which characterize the subtropical community, appear to depend less on the phytoplanktonic prey. Euphausia diomedeae and Hansarsia gracilis, typifying the tropical population, showed stronger hypoxia tolerance. Additionally, each community was further divided into three subgroups under the influence of surface PP (subtropical: marginal NPSG, central NPSG, and austral EI-SP subgroups; tropical: the Bay of Bengal, equatorial EI, and equatorial EI-SP subgroups). These results suggested that food resource is the most important in shaping euphausiids’ community structure in an oligotrophic ecosystem with subtle hydrography gradients.
Similar content being viewed by others
1 Introduction
Euphausiids (Order Euphausiacea) are shrimp-like crustaceans that inhabit oceanic basins worldwide. They are vital pelagic ecosystem components, contributing to approximately 5–10% of total zooplankton biomass (Everson 2008). Through diel vertical migration, many euphausiid species accelerate the transport of organic matter produced by phytoplankton photosynthesis from the euphotic zone to the deeper sea (Bianchi et al. 2013). Euphausiids are also significant links between lower and higher trophic levels in marine ecosystems as they feed on marine snow, phytoplankton, and small zooplankton (Dilling et al. 1998; Park et al. 2011) and function as frequent prey for commercially important fishes, marine mammals, and birds (Roger 1994). Thus, ascertaining the community structure and biomass of euphausiids is important for determining the fate of primary production and flux of organic matter to the deep ocean through the food web (Steinberg et al. 2008).
The worldwide distribution of the present 86 nominal euphausiid species has been systematically reported by Brinton et al. (2000). Euphausiid assemblages exhibit an apparent affinity for water masses, since they quickly react to changing hydrographical conditions, leading to a different community structure (Brinton 1962; Gibbons et al. 1995). Seawater temperature has been well-documented as the most crucial environmental factor affecting the euphausiid community structure. A summary of euphausiid standing stocks is available for the Atlantic Ocean (Letessier et al. 2009; 2011b) as well as for the Pacific Ocean (Letessier et al. 2011a). These studies confirm that the abundance and species richness of euphausiids in the two oceans were most influenced by sea surface temperature, linking higher euphausiid species richness with warmer seawater based on the species-energy hypothesis (Rohde 1992). Rutherford et al. (1999) and Rombouts et al. (2009) reached a similar conclusion in the global biodiversity pattern across other marine taxa.
Notable in the aforementioned studies are observations of considerably weaker impacts of seawater temperature on zooplankton community variation above 25 ℃. This implies that other environmental factors should appear more significant in determining euphausiids’ community structure in low-latitude open-water ecosystems which are generally characterized by higher temperatures. For instance, it has been shown that food resource availability might influence euphausiid distribution in the southeastern Indian Ocean (9–32° S, 110° E; McWilliam 1977) and may also be responsible for the differences in euphausiid species composition between the eastern and western North Pacific subtropical gyre (Zhou et al. 2021). Low dissolved oxygen levels were shown to prevent the existence of some euphausiids without good hypoxia tolerance (Tremblay et al. 2010, 2020; Seibel et al. 2016; Sutton and Beckley 2017), whereas surface circulations could disperse euphausiids beyond their normal distribution limits (Sutton and Beckley 2016). A comprehensive survey of euphausiid communities across subtropical and tropical zones covering different open oceans is required to gain deeper insight into a factor controlling community structures of euphausiids in low-latitude ecosystems.
The Pacific Ocean is the oldest and largest ocean basin (Barkley 1968). It hosts 59 of the 86 species of euphausiids (Brinton 1962; Brinton et al. 2000), with the North Pacific subtropical gyre (NPSG) being one of the hotspots of euphausiid species diversity (Letessier et al. 2011a). However, investigations on euphausiid spatial displacements, particularly the species-specific responses caused by changing environments, have been largely focused on mid-high latitudes or productive systems (Brinton and Townsend 2003; Yoon et al. 2006; Ambriz-Arreola et al. 2012; Rivera-Gómez et al. 2019; Lilly and Ohman 2021). The Indian Ocean contains two-thirds of the world’s euphausiid species (Brinton and Gopalakrishnan 1973). However, the Indian Ocean has received less attention than the Pacific and Atlantic Oceans due to its dynamic hydrography and limited accessibility. To fill this knowledge gap, there is a need to investigate the distribution of euphausiids species in low-latitude regions of the Pacific and Indian Oceans.
In the present study, euphausiids were collected mainly from the subtropical/tropical regions in the Pacific and eastern Indian Oceans. We posited that primary production, as a proxy for food available in the environment, is a significant driver for structuring the distribution and association of euphausiid species in low latitudes. To test this hypothesis, we present (1) the euphausiid abundance, species diversity, and the representative euphausiid species; (2) the primary drivers of the euphausiid distribution variations; and (3) how the representative species of euphausiids associate with these drivers in the low-latitude Indian and Pacific Oceans.
2 Materials and methods
2.1 Field collection
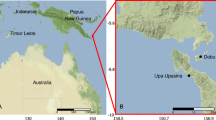
Sampling was conducted onboard the R/V Hakuho-Maru at 25 stations during the KH17-4, KH18-6, and KH19-6 cruises (Fig. 1, Table 1). The study sites are located mainly in the subtropical and tropical areas of the Indian and Pacific Oceans (except NP3 located in the California Current [CC] and NP4 in the subarctic-subtropical transition zone of the eastern North Pacific [Roden 1971]). At each station, zooplankton samples were collected at night from approximately 500 m depth to the surface using oblique tows of Matsuda-Oozeki-Hu Trawl (MOHT; Oozeki et al. 2007) at 3–4 knots of the ship speed. The MOHT in this study had a square mouth of 2 m2 mouth area (1.43 × 1.43 m) and 1.95 mm mesh size. A flow meter was attached to the net to measure the filtered water volume. Samples were promptly split after landing of the net onboard, and one-eighth or one-fourth of the aliquots were fixed with 5% buffered formaldehyde for community structure analysis. Vertical profiles of water temperature, salinity, and dissolved oxygen (DO; mL O2 L−1) between 0 and 500 m at all stations were recorded using a conductivity-temperature-depth (CTD) system (SBE-911 plus; Sea-Bird Electronics) and dissolved oxygen sensor (SBE-43; Sea-Bird Electronics). Mixed layer depth (MLD) was determined using a ΔT = 0.2 ℃ criterion relative to a 10-m reference level on individual profiles (de Boyer et al. 2004). For chlorophyll a (Chl-a) measurements, water samples were collected using Niskin bottles attached to the CTD system at 13 intervals from 0 to 250 m at each station. Chl-a was extracted with N, N-dimethylformamide, and its concentration was analyzed using a Turner fluorometer (Turner Designs). Additionally, nitrogen fixation rate and primary production were measured at depths corresponding to 100%, 10%, 1%, and 0.1% of surface light density using the 15N-13C dual inlet technique that combines 15N2 gas dissolution (Mohr et al. 2010) with a 13C primary production assay (Hama et al. 1983) according to Sato et al. (2022) and Horii et al. (2023). Depth-integrated nitrogen fixation and primary production were calculated using trapezoidal integration. Details of the sampling and water properties are presented in Table 1 and S1, respectively.
2.2 Euphausiid community structure analysis
All adult euphausiids at each station were sorted in the laboratory and identified to the lowest possible taxonomic level according to Baker et al. (1990) and Brinton et al. (2000) under a stereomicroscope (Nikon SMZ-745 T). The genus Nematoscelis is described as Hansarsia in the present study, according to Shaw et al. (2023). Thysanopoda astylata and T. aequalis were merged as one species because of the absence of subtle morphological differences between the two species. The total length of euphausiids was measured from the tip of the rostrum to the end of the telson. The abundance of each species was standardized as individuals (ind.) 1000 m–3. Species richness (N, total number of species), Shannon index (H’; Shannon 1948), and Simpson’s diversity index (D [compliment]; Simpson 1949) were computed to evaluate euphausiid species diversity. To detect the regional differences in euphausiid abundance and species diversity, the Kruskal–Wallis test with Dunn post hoc test or Mann–Whitney U test was conducted when data sets were non-normally distributed. Otherwise, a one-way analysis of variance (ANOVA) with Fisher’s least significant difference post-hoc test was used. To investigate the relationship between euphausiid abundance and species diversity and the environment, correlations among euphausiid abundance, species diversity, and environmental parameters were tested using Spearman’s rank correlation. All analyses were performed using the IBM SPSS Statistics 23 (Zar 1999).
For further community analyses, to avoid spurious associations among rare species, only species comprising more than 3% of the total euphausiid abundance at any station were included. To evaluate the relationship among euphausiid assemblages in the study area, cluster analysis (group average) and non-metric multidimensional scaling ordination based on the Bray–Curtis similarity index were employed on log(x + 1)-transformed euphausiid abundance data. To assess the similarity within the potential clustered groups and the dissimilarity between groups and verify the “representative species” that typified every clustered group, similarity percentages analysis (SIMPER) was executed (Clarke 1993). Euphausiid species that contributed to the similarity/dissimilarity within/between groups majorly (displaying higher Av.Sim or Av.Diss values) and consistently (larger Sim/SD or Diss/SD ratios) were determined as “representative species” of the defined clustered groups.
To explore the effects of environmental drivers on euphausiid community structure in the low latitudes (except NP3), sea surface temperature (SST), sea surface salinity (SSS), depth-averaged (0–500 m) temperature and salinity (T500; S500), depth-averaged (0–250 m) DO (DO250), MLD, surface Chl-a, integrated Chl-a from 0 to 250 m, integrated nitrogen fixation, surface primary production, and integrated primary production were assessed using distance-based linear model (DistLM) analysis. To determine the best combination of these predictor variables to explain the variation in the euphausiid community, models were constructed using the BEST selection procedure (all possible variable combinations) under the Akaike Information Criterion (AIC) with 9999 permutations. Finally, distance-based redundancy analysis (dbRDA) was used to visualize the impact of the fitted environmental parameters ascertained by DistLM on the representative euphausiid species. All analyses were performed using PRIMER version 7 with the PERMANOVA + add-on package (Anderson et al. 2008).
3 Results
3.1 Environmental conditions
Located at relatively higher latitudes, NP3–4 demonstrated distinctively lower temperatures and salinities than other stations in the subtropical and tropical areas (Fig. 2A and B, Fig. S1). NP6, on the outer edge of NPSG, also presented unique intermediate thermohaline conditions. As for the low-latitude area, stations in the South Pacific were characterized by a significantly lower average SST (20.6 ℃) than the eastern Indian (average of 27.8 ℃) and other NPSG stations (NP7–15; 27.8 ℃; Kruskal–Wallis; p < 0.05) and a relatively higher average SSS. The South Pacific water was also well-mixed, whereas a prominent temperature gradient in the upper 200 m was observed in NPSG and the eastern Indian Ocean (Fig. 2A). Moreover, the water mass of the eastern Indian Ocean displayed lower salinity in the upper 100 m (except at EI5), which was different from that of NPSG with higher salinity (Fig. 2B and Table S1). Another notable feature of the eastern Indian stations was a significantly lower average DO250 (2.4 mL O2 L−1) compared to the NPSG stations (average 4.5 mL O2 L−1) and the South Pacific stations (average 4.2 mL O2 L−1; Kruskal–Wallis; p < 0.05). Hypoxic conditions (< 1 mL O2 L−1) were observed in the shallow layers at the northern Indian stations and SP1 (49–145 m).
The surface Chl-a concentration was similar across the three ocean regions. However, the average integrated Chl-a from 0 to 250 m was significantly lower in the South Pacific (17.1 mg m−2) than in the NPSG area (29.7 mg m−2) and eastern Indian Ocean (27.9 mg m−2; Kruskal–Wallis; p < 0.05). Deeper subsurface chlorophyll maxima (below 100 m) were obtained at stations in the subtropical zone, particularly in western NPSG (NP11–15; average 128 m). Furthermore, a regional pattern of Chl-a was evident in each ocean (Fig. 3A). The average surface Chl-a levels of stations EI1–4 and SP1–2 were significantly higher than the southern EI7–10 and SP3–5 (One-way ANOVA, p < 0.05). The western NPSG stations displayed a relatively greater average integrated Chl-a concentration (33.5 mg m−2) than the eastern NPSG stations (25.9 mg m−2). Additionally, the average integrated primary production (PP) was significantly lower in the NPSG region (0.21 g C m−2 d−1) than in the eastern Indian (0.73 g C m−2 d−1) and South Pacific Oceans (0.70 g C m−2 d−1; One-way ANOVA, p < 0.05). Like the regional pattern of Chl-a, the northern stations in the eastern Indian and South Pacific Oceans had significantly higher average surface and integrated PP than the southern stations (one-way ANOVA, p < 0.05). However, the eastern NPSG displayed a higher average surface and integrated PP than the western NPSG, in contrast to the integrated Chl-a pattern in the NPSG region. The integrated nitrogen fixation of the NPSG region was significantly greater (average 91.1 μmol N m−2 d−1) than that of the low-latitude South Pacific (16.0 μmol N m−2 d−1) and the eastern Indian Oceans (45.7 μmol N m−2 d−1; Kruskal–Wallis; p < 0.05).
3.2 Abundance and diversity of euphausiids
A total of 41 euphausiid species (including nine rare species) from six genera were identified in the study area (Table S2): thirteen species of Euphausia, nine of Stylocheiron, eight of Thysanopoda, six of Hansarsia, three of Nematobrachion, and two of Thysanoessa. The average length (mean ± SD) of identified euphausiid species ranged from 4.9 ± 0.1 (S. suhmii) to 29.3 ± 2.6 mm (T. orientalis). The total abundance of adult euphausiids significantly varied among stations from 13.9 ind. 1000 m–3 at NP13 to 1893.3 ind. 1000 m–3 at EI1 (Fig. 4A). Moreover, euphausiid abundance was strongly and positively correlated with surface PP, integrated PP, and surface Chl-a and negatively correlated with DO250 (Table 2). Specifically, the eastern Indian Ocean with a higher integrated PP and lower DO level possessed a significantly greater euphausiid abundance (average 462.4 ind. 1000 m–3) than the North Pacific (71.6 ind. 1,000 m–3; SP:214.7 ind. 1000 m–3). A southward-decreasing gradient of euphausiid abundance was observed in the eastern Indian Ocean, consistent with the surface Chl-a tendency. Euphausiid abundance in the NPSG region followed the same pattern as the surface and integrated PP, showing a significantly higher abundance in eastern NPSG (NP6–10; peaked at NP10) than in western NPSG (Mann–Whitney U; p < 0.05).
A Euphausiid species composition and abundance (individuals/ 1,000 m3) at each sampling station. Different colors indicate different euphausiid species. B Euphausiid species diversity (H′ and D) and total species number (N) of each station. Subtropical stations with higher species diversity are shaded
In contrast to the euphausiid abundance pattern, the eastern Indian Ocean presented a relatively lower euphausiid species number and diversity (mean ± SD; N = 12.8 ± 6.3, H′ = 1.5 ± 0.7, D = 0.6 ± 0.2) than the North Pacific (N = 14.3 ± 3.2, H′ = 2.0 ± 0.5, D = 0.8 ± 0.2) and South Pacific Oceans (N = 16.6 ± 2.5, H′ = 2.1 ± 0.4, D = 0.8 ± 0.1). Correspondingly, euphausiid species diversity was negatively associated with surface PP, integrated PP, and surface Chl-a, and the species number was negatively related to surface Chl-a and PP. A positive correlation between species diversity and DO concentration was observed (Table 2). Moreover, within the lower latitudes, there was a general increasing trend in euphausiid species diversity from the equator to ~ 20°S both in the eastern Indian Ocean and the South Pacific. Further, stations settled in the subtropical gyres (latitude ~ 20–30°) showed significantly higher H’ and D than the tropical stations (One-way ANOVA, p < 0.05, Fig. 4B).
3.3 Euphausiid community structure related to environmental factors
Based on the abundance and composition of euphausiids at each station, cluster analysis helped delineate three major groups of euphausiid communities in the study area at a 22% similarity level, the CC, subtropical, and tropical groups (Fig. 5). The CC group containing only NP3 at a higher latitude, dominated by Euphausia pacifica (Fig. 4A), was considerably different from the other two groups (at a 10% similarity level); therefore, it was considered an outgroup. The subtropical group consisting of stations NP4–15, EI9–10, and SP3–5 displayed significantly lower euphausiid abundance and higher euphausiid species number and diversity than the tropical group comprising stations EI1–8 and SP1–2 (Fig. 4B; One way ANOVA, p < 0.05). These two groups were further divided into three subgroups at a 45% similarity level (subtropical: marginal NPSG, central NPSG, and austral eastern Indian Ocean [EI]-South Pacific [SP] subgroups; tropical: Bay of Bengal [BoB], equatorial EI-SP, and equatorial EI subgroups; Fig. 5). The equatorial EI subgroup only contained station EI5 at the equator whereas other stations located in the equatorial region of the South Pacific (SP1–2) and the eastern Indian Ocean (EI4 and EI7–8) were included in the equatorial EI-SP subgroup.
A Dendrogram derived by cluster analysis on the euphausiid community using log(x + 1)-transformed species abundance data and the Bray–Curtis similarity index. B Multidimensional scaling ordination plot overlapping clustered analysis suggesting similarities between euphausiid communities. C Geographic distributions of different clustered groups
DistLM analysis revealed that most of the assessed environmental variables significantly influenced the low-latitude euphausiid community structure individually (p < 0.05), especially DO250 (Pseudo F = 12.13; p < 0.01), surface PP (Pseudo F = 10.71; p < 0.01), and total PP level (Pseudo F = 10.02; p < 0.01) (Table S3). Moreover, the combination of SST, SSS, S500, DO250, surface Chl-a, surface PP, integrated PP, and integrated nitrogen fixation optimally accounted for euphausiid community structure patterns across the study area (R2 = 0.72). According to the RDA ordination, these environmental variables explained 40.7% and 13.4% of the total variance in euphausiid communities on Axis1 and Axis2, respectively (Fig. 6). The subtropical and tropical groups bore strong connections to integrated PP and DO250, which were highly correlated with Axis1. The secondary structure of the two euphausiid communities was mainly affected by surface PP, Chl-a, and thermohaline conditions. Within the subtropical community, the austral EI-SP subgroup correlated with higher salinity, distinct from the marginal and central NPSG subgroups associated with warmer and less saline water. For the tropical community, the equatorial EI and BoB subgroups with higher surface PP and SST were clearly separated from the equatorial EI-SP subgroup (Fig. 6 and Fig. 5C).
Distance-based redundancy analysis (dbRDA) triplot illustrating the fitted distance-based linear model for low-latitude euphausiid community groups (except for NP3) and representative euphausiid species in the study area. Percentages (%) suggest the proportion of the fitted variation and the total variation in the Bray–Curtis resemblance matrix explained by dbRDA axes. The environmental variables that significantly explain the variation in low-latitude euphausiid communities identified by the model are indicated as vectors in solid lines. Representative species were annotated by vectors as gray dashed lines. (Esib: E. sibogae; Hgra: H. gracilis; Edio: E. diomedeae; Eten: E. tenera; Hten: H. tenella; Egib: E. gibba; Hatl: H. atlantica; Scar: S. carinatum; Emut: E. mutica; Sabb: S. abbreviatum; Ebre: E. brevis; Erec: E. recurva; Ehem: E. hemigibba; Egibo: E. gibboides)
3.4 Representative euphausiid species in subtropical and tropical areas
SIMPER analysis revealed that E. brevis, E. mutica, and Stylocheiron abbreviatum were primary contributors to the internal similarity of the subtropical community (higher Av.sim values; Table 3). All three species occupied > 10% of the total subtropical community abundance (high Av.abund value), and they were evenly distributed (high Sim/SD ratio) at the subtropical stations (Fig. 4A and Table 3). However, S. abbreviatum was scarce (< 5 ind. 1000 m–3) at most stations, yet only overwhelmingly abundant at NP10, in contrast to E. brevis and E. mutica (Fig. 4A and Table S2). Furthermore, the heterogeneous distribution of specific euphausiid species shaped the secondary structure of the subtropical euphausiid community. The absence of E. hemigibba only in the austral EI-SP, relatively greater abundances of Hansarsia atlantica and S. carinatum in the south hemisphere, and higher density of E. brevis in central NPSG segregated the austral EI-SP subgroup and other two subgroups; the absence of T. astylata/aequalis together with the prevalence of E. hemigibba and E. mutica at the edge of NPSG, in addition to the dominance of E. gibboides and E. recurva in the subarctic-subtropical transition zone of NP, promoted discrepancy between the marginal and central NPSG euphausiid communities (Fig. 4A and Table S4).
Euphausia diomedeae and H. gracilis, which were always observed simultaneously, typified the tropical euphausiid population (Fig. 4A and Table 3). Both species were discovered at every station in the tropical group; however, E. diomedeae (accounting for 28.3% of the tropical euphausiid abundance) was more abundant than H. gracilis (10.4%; Fig. 4A). Within the tropical community, the equatorial EI assemblage that was significantly occupied by T. tricuspidata differed considerably from the BoB and equatorial EI-SP assemblages (Figs. 4A and 5A). Euphausia sibogae uniquely characterized the BoB population, and the higher abundance and frequent occurrence of E. tenera and H. tenella in the equatorial area also intensified the dissimilarities between the equatorial EI-SP and other subgroups (Fig. 4A and Table S5). The distribution characteristics of the representative euphausiid species that established the subtropical and tropical euphausiid communities are summarized in Table 4.
4 Discussion
The present study successfully provides a holistic account of euphausiid community structure at low latitudes covering the Pacific and Indian Oceans, complementing the three previous seminal studies regarding biogeography (Brinton 1962, Brinton and Gopalakrishnan 1975) and ecology of euphausiids (Lettesier et al. 2011a). As we posited, integrated primary production (PP) was the most critical environmental driver controlling the euphausiid community structure in low latitudes. PP significantly correlated with the euphausiid abundance and species diversity. Euphausiid species displayed distinct distribution patterns based on their association with PP, dividing the low-latitude population into subtropical and tropical groups.
4.1 Primary productivity affecting euphausiid abundance and species diversity in low latitudes
At the marine food web base, primary productivity is a critical determinant of the abundance of higher trophic levels, including zooplankton and fish (Matsuzaki et al. 2018; Capuzzo et al. 2022; Behrenfeld et al. 2019). The higher integrated PP rates triggered by mesoscale eddies in the Bay of Bengal, equatorial eastern Indian Ocean, and tropical South Pacific off Peru are known to sustain the large tropical assemblages of euphausiid abundance (Gordon et al. 2017; Stramma et al. 2013; Czeschel et al. 2018; Hu et al. 2022). However, low rates of PP limited by the short nutrient supply in the subtropical gyres significantly restrained the subtropical population (Begon et al. 2014; Steinberg et al. 2008).
The characteristic high euphausiid species diversity in the subtropics in the present study agreed with the admitted latitudinal gradient of the global biodiversity pattern (Gaston 2000) and the modeling results of Letessier et al. (2011a) in the Pacific Ocean, in which the highest euphausiid species richness was observed in the western NPSG and center of SPSG. The euphausiid species diversity in the present study was negatively correlated with the surface/total PP significantly, indicating the niche partitioning resulting from a stable vertical distribution structure or varied feeding preferences of coexisting euphausiid species was a vital driver of higher diversity in the oligotrophic regions (Hayward and McGowan 1979; Longhurst 1985). Since the species number and evenness were high at subtropical stations, finer-scale niche partitioning among euphausiid species, reflecting different responses to food resource availability, is expected in subtropics. Additionally, the higher stability of the subtropical gyre centers facilitated the increased zooplankton species diversity (Brinton 1962; Letessier et al. 2011a).
Our study detected no clear correlation between any diversity indices and seawater temperature, agreeing with the observations of McWilliam (1977) in the Indian Ocean. One explanation for the “divergence” between our results and the high temperature-high diversity link of Letessier et al. (2009, 2011a) was that the responses of zooplankton species diversity to environmental drivers displayed remarkable spatial scale dependencies. Studies above were performed in considerably broad latitudinal gradients with apparent temperature changes (up to 60° difference in latitude, and 20 ℃ difference in SST). Yet, most of our study sites were concentrated in the low latitudes (with < 10 ℃ differences in SST). Unsurprisingly, species diversity was positively correlated with SST concerning only the NPSG area, including station NP4 of low temperature (Spearman correlation = 0.673, p < 0.05; Fig. S2).
4.2 Ecology and biogeography of representative euphausiid species in low latitudes
The correlations between PP level and representative euphausiid species indicated their feeding strategies or functional responses to adapt to changing food and environmental conditions (Gentleman et al. 2003; Bachiller et al. 2016), further influencing the euphausiid community structure. For example, filter-feeding omnivorous Euphausia species would consume more heterotrophic prey than autotrophic one in a lower phytoplankton density environment (Perissinotto et al. 2000; Stuart and Pillar 1990; Henschke et al. 2016; Zhou et al. 2021). Hence, subtropical E. brevis correlating with integrated/surface PP negatively should be more carnivorous than tropical E. diomedeae (Fig. 6). Zhou et al. (2021) discovered that gut contents of E. brevis contained mainly copepods and mixotrophic dinoflagellates due to the lower PP contributed by picophytoplankton. By contrast, E. diomedeae was likely to utilize more eukaryotic phytoplankton such as diatoms, phototrophic dinoflagellates, and pelagophytes that contributed to the enhanced PP in the equatorial eastern Indian Ocean (Isaji et al. 2022) and the tropical South Pacific (Rousseaux and Gregg 2012; Bruland et al. 2005). Within the subtropical assemblage, E. hemigibba associated with surface PP positively displayed an herbivory tendency compared to E. brevis (Zhou et al. 2021). This led E. hemigibba and E. brevis to dominate the relatively productive marginal NPSG and the oligotrophic central NPSG, respectively.
Unlike Euphausia, the raptorial Stylocheiron and versatile Hansarsia (capable of raptorial feeding and weak filter feeding; Gopalakrishnan 1975) are not subject to the low encounter rate with food particles in oligotrophic environments (Prowe et al. 2019; Ge et al. 2022), thereby showing cosmopolitan distributions in the low latitudes. Of interest was the elevated abundance of the mesopelagic S. abbreviatum (Wiebe et al. 2016) at NP10 near the Hawaii islands. To some extent, increased new production and vertical carbon flux supported by nitrogen fixation favored them through the bottom-up control of the food web (Fig. 6, Nagatomo et al. 2023). Ongoing research regarding the feeding ecology of the representative species would offer more details for advancing the current knowledge of euphausiid distribution patterns in low-latitude ecosystems.
Dissolved oxygen (DO) affected the low-latitude euphausiid community structure significantly, in addition to PP. Most representative subtropical euphausiid species correlated positively with DO (Fig. 6), underlying their weak ability to adapt to a lower DO concentration than the tropical ones. Investigations on euphausiids’ hypoxia tolerance were mainly conducted in the world’s three major Oxygen Minimum Zones (OMZs), in which subtropical representatives of the present study were hardly detected. Only E. gibboides and E. recurva could use the OMZ during the day as a refuge from predators in the Gulf of California (Ambriz-Arreola et al. 2017) and the Southwest Atlantic Ocean (Werner and Bucholz 2013), respectively. This corresponded to their higher abundance and absence of other subtropical species in the eastern NPSG margin with a shallower upper boundary of the low-oxygen zone (Fig. 4A, Table S1). On the contrary, the superior hypoxia adaptation of tropical E. diomedeae, H. gracilis, and E. tenera allowed them to inhabit the oxygen-depleted northern Indian Ocean and the eastern tropical Pacific (Antezana 2009; Tremblay et al. 2020; Seibel et al. 2016).
4.3 Sampling limitations
Quantitative sampling of euphausiids is sometimes difficult because of their net avoidance behavior (Brinton 1967; Wiebe et al. 2013; Gislason et al. 2023). The Matsuda-Oozeki-Hu Trawl, with an ability of working at a relatively higher towing speed, showed a superiority in collecting macroplanktonic crustaceans compared to other gears (Yamamura and Yasuma 2010). The abundance and distribution of representative euphausiid species were comparable with previous reports (Brinton 1962, Brinton and Gopalakrishnan 1975; Brinton et al. 2000). Although the coarse mesh size tends to result in an underestimation of smaller species and larvae, our study mainly focused on the adult euphausiids, and the average total length of identified euphausiid species was consistent with Brinton et al. (2000) (Table S2, Fig. S3). This indicates that the bias from a larger mesh opening was minor. Future studies with multiple tows at each station and modified trawls with strobe light systems should be implemented to minimize the impact further. Additional sampling stations along the latitudinal gradient across the equatorial Pacific Ocean and the South Pacific subtropical gyre are expected to include more environmental gradients.
5 Conclusions
The present study revealed that the euphausiid community structure, numerical abundance, and species diversity pattern in the subtropical and tropical zones across the Indian and Pacific Oceans were most significantly influenced by primary productivity rates in the water column. Our results indicate that food resources appear more critical than seawater temperature in shaping euphausiids’ community structure in an oligotrophic ecosystem with subtle hydrography gradients. A closer examination of the feeding habits and conditions of euphausiids could offer more insights for assessing and predicting their responses to the changing environment.
References
Ambriz-Arreola I, Gómez-Gutiérrez J, del Carmen F-G, Lavaniegos BE, Godínez-Domínguez E (2012) Influence of coastal upwelling-downwelling variability on tropical euphausiid abundance and community structure in the inshore Mexican central Pacific. Mar Ecol Prog Ser 451:119–136. https://doi.org/10.3354/meps09607
Ambriz-Arreola I, Gómez-Gutiérrez J, del Carmen Franco-Gordo M et al (2017) Vertical pelagic habitat of euphausiid species assemblages in the Gulf of California. Deep Sea Res Part I Oceanogr Res 123:75–89. https://doi.org/10.1016/j.dsr.2017.03.008
Anderson MJ, Gorley RN, Clarke KR (2008) Permanova+ for primer: guide to software and statistical methods. PRIMER-E, Plymouth
Antezana T (2009) Species-specific patterns of diel migration into the Oxygen Minimum Zone by euphausiids in the Humboldt Current Ecosystem. Prog Oceanogr 83:228–236. https://doi.org/10.1016/j.pocean.2009.07.039
Bachiller E, Skaret G, Nøttestad L, Slotte A (2016) Feeding ecology of Northeast Atlantic mackerel, Norwegian spring-spawning herring and blue whiting in the Norwegian Sea. PLoS ONE 11:e0149238. https://doi.org/10.1371/journal.pone.0149238
Baker ADC, Boden BP, Brinton E (1990). Practical guide to the euphausiids of the world. British Museum (Natural History), London
Barkley RA (1968) Oceanographic atlas of the Pacific Ocean. University of Hawaii Press, In Oceanographic Atlas of the Pacific Ocean. https://doi.org/10.1515/9780824891541
Begon M, Howarth RW, Townsend CR (2014) Essentials of ecology. John Wiley & Sons Inc, New Jersey
Behrenfeld MJ, Gaube P, Della Penna A et al (2019) Global satellite-observed daily vertical migrations of ocean animals. Nature 576:257–261. https://doi.org/10.1038/s41586-019-1796-9
Bianchi D, Stock C, Galbraith ED, Sarmiento JL (2013) Diel vertical migration: Ecological controls and impacts on the biological pump in a one-dimensional ocean model. Glob Biogeochem 27:478–491. https://doi.org/10.1002/gbc.20031
Brinton E (1962) The distribution of Pacific euphausiids. Bull Scripps Inst Oceanogr 8:51–270
Brinton E (1967) Vertical migration and avoidance capability of euphausiids in the California current. Limnol Oceanogr 12:451–483. https://doi.org/10.4319/lo.1967.12.3.0451
Brinton E, Gopalakrishnan K (1973) The distribution of Indian Ocean euphausiids. The biology of the Indian Ocean. Springer, Berlin, pp 357–382
Brinton E, Townsend A (2003) Decadal variability in abundances of the dominant euphausiid species in southern sectors of the California Current. Deep-Sea Res II: Top Stud Oceanogr 50:2449–2472. https://doi.org/10.1016/S0967-0645(03)00126-7
Brinton E, Ohman MD, Townsend AW, Knight MD, Bridgeman AL (2000). Euphausiids of the world ocean. Series: World Biodiversity Database CD-ROM Series Window version, 1.
Bruland KW, Rue EL, Smith GJ, DiTullio GR (2005) Iron, macronutrients and diatom blooms in the Peru upwelling regime: brown and blue waters of Peru. Mar Chem 93:81–103. https://doi.org/10.1016/j.marchem.2004.06.011
Capuzzo E, Wright S, Bouch P et al (2022) Variability in structure and carbon content of plankton communities in autumn in the waters south-west of the UK. Prog Oceanogr 204:102805. https://doi.org/10.1016/j.pocean.2022.102805
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Austral Ecol 18:117–143. https://doi.org/10.1111/j.1442-9993.1993.tb00438.x
Czeschel R, Schütte F, Weller RA, Stramma L (2018) Transport, properties, and life cycles of mesoscale eddies in the eastern tropical South Pacific. Ocean Sci 14:731–750. https://doi.org/10.5194/os-14-731-2018
de Boyer MC, Madec G, Fischer AS, Lazar A, Iudicone D (2004) Mixed layer depth over the global ocean: an examination of profile data and a profile-based climatology. J Geophys Res Oceans. https://doi.org/10.1029/2004JC002378
Dilling L, Wilson J, Steinberg D, Alldredge A (1998) Feeding by the euphausiid Euphausia pacifica and the copepod Calanus pacificus on marine snow. Mar Ecol Prog Ser 170:189–201. https://doi.org/10.3354/meps170189
Everson I (2008) Krill: biology, ecology and fisheries. Wiley-Blackwell, Chichester
Gaston K (2000) Global patterns in biodiversity. Nature 405:220–227. https://doi.org/10.1038/35012228
Ge R, Li Y, Chen H, Lei F, Zhuang Y, Liu G (2022) Diel Vertical distribution of mesozooplankton functional groups in the north pacific subtropical gyre: a case study. Front Mar Sci 9:854642. https://doi.org/10.3389/fmars.2022.854642
Gentleman W, Leising A, Frost B, Strom S, Murray J (2003) Functional responses for zooplankton feeding on multiple resources: a review of assumptions and biological dynamics. Deep-Sea Res II: Top Stud Oceanogr 50:2847–2875. https://doi.org/10.1016/j.dsr2.2003.07.001
Gibbons MJ (1995) Observations on euphausiid assemblages of the south coast of South Africa. S Afr J Mar Sci 16:141–148. https://doi.org/10.2989/025776195784156674
Gislason A, Petursdottir H, Reynisson P (2023) Effect of strobe lights on catches and length distributions of euphausiids collected by Bongo nets. J Plankton Res 45:99–109. https://doi.org/10.1093/plankt/fbac063
Gopalakrishnan K (1975) Biology and taxonomy of the genus Nematoscelis (Crustacea, Euphausiacea). Fish Bull 73:797–814
Gordon AL, Shroyer E, Murty VSN (2017) An intrathermocline eddy and a tropical cyclone in the Bay of Bengal. Sci Rep 7:1–8. https://doi.org/10.1038/srep46218
Hama T, Miyazaki T, Ogawa Y, Iwakuma T, Takahashi M, Otsuki A, Ichimura S (1983) Measurement of photosynthetic production of a marine phytoplankton population using a stable 13C isotope. Mar Biol 73:31–36. https://doi.org/10.1007/BF00396282
Hayward TL, McGowan JA (1979) Pattern and structure in an oceanic zooplankton community. Am Zool 19:1045–1055
Henschke N, Everett JD, Richardson AJ, Suthers IM (2016) Rethinking the role of salps in the ocean. Trends Ecol Evol 31:720–733. https://doi.org/10.1016/j.tree.2016.06.007
Horii S, Takahashi K, Shiozaki T et al (2023) East-west variabilities of N2 fixation activity in the subtropical North Pacific Ocean in summer: potential field evidence of the phosphorus and iron co-limitation in the western area. J. Geophys Res Oceans 128:e2022JC019249. https://doi.org/10.1029/2022JC019249
Hu Z, Ma X, Peng Y et al (2022) A large subsurface anticyclonic eddy in the Eastern Equatorial Indian Ocean. J Geophys Res Oceans 127:e2021JC018130. https://doi.org/10.1029/2021JC018130
Isaji Y, Yoshikawa C, Ogawa NO et al (2022) Nitrogen sources for phytoplankton in the eastern Indian Ocean determined from δ15N of chlorophyll a and divinylchlorophyll a. Geochem Geophys Geosyst 23:e2021GC010057. https://doi.org/10.1029/2021GC010057
Letessier TB, Cox MJ, Brierley AS (2009) Drivers of euphausiid species abundance and numerical abundance in the Atlantic Ocean. Mar Biol 156:2539–2553. https://doi.org/10.1007/s00227-009-1278-y
Letessier TB, Cox MJ, Brierley AS (2011a) Drivers of variability in euphausiid species abundance throughout the Pacific Ocean. J Plankton Res 33:1342–1357. https://doi.org/10.1093/plankt/fbr033
Letessier TB, Falkenhaug T, Debes H, Bergstad OA, Brierley AS (2011b) Abundance patterns and species assemblages of euphausiids associated with the Mid-Atlantic Ridge, North Atlantic. J Plankton Res 33:1510–1525. https://doi.org/10.1093/plankt/fbr056
Lilly LE, Ohman MD (2021) Euphausiid spatial displacements and habitat shifts in the southern California Current System in response to El Niño variability. Prog Oceanogr 193:102544. https://doi.org/10.1016/j.pocean.2021.102544
Longhurst AR (1985) Relationship between diversity and the vertical structure of the upper ocean. Deep-Sea Res I: Oceanogr Res Pap 32:1535–1570. https://doi.org/10.1016/0198-0149(85)90102-5
Matsuzaki SS, Suzuki K, Kadoya T, Nakagawa M, Takamura N (2018) Bottom-up linkages between primary production, zooplankton, and fish in a shallow, hypereutrophic lake. Ecology 99:2025–2036. https://doi.org/10.1002/ecy.2414
McWilliam PS (1977) Further studies of plankton ecosystems in the eastern Indian Ocean. VI. Ecology of the Euphausiacea. Mar Freshw Res 28:627–644. https://doi.org/10.1071/MF9770627
Mohr W, Grosskopf T, Wallace DW, LaRoche J (2010) Methodological underestimation of oceanic nitrogen fixation rates. PLoS ONE 5:e12583. https://doi.org/10.1371/journal.pone.0012583
Nagatomo Y, Horii S, Hirai J et al (2023) Geographic distribution of micronektonic fish communities in the subtropical North Pacific: the effect of primary productivity and nitrogen fixation. Prog Oceanogr 217:103086. https://doi.org/10.1016/j.pocean.2023.103086
Oozeki Y, Takasuka A, Kubota H, Barange M (2007) Characterizing spawning habitats of Japanese sardine, Sardinops melanostictus, Japanese anchovy, Engraulis japonicus, and Pacific round herring, Etrumeus teres, in the Northwestern Pacific. CCOFI Rep 48:191–203
Park JI, Kang CK, Suh HL (2011) Ontogenetic diet shift in the euphausiid Euphausia pacifica quantified using stable isotope analysis. Mar Ecol Prog Ser 429:103–109. https://doi.org/10.3354/meps09091
Perissinotto R, Gurney L, Pakhomov EA (2000) Contribution of heterotrophic material to diet and energy budget of Antarctic krill, Euphausia superba. Mar Biol 136:129–135. https://doi.org/10.1007/s002270050015
Prowe AF, Visser AW, Andersen KH, Chiba S, Kiørboe T (2019) Biogeography of zooplankton feeding strategy. Limnol Oceanogr 64:661–678. https://doi.org/10.1002/lno.11067
Rivera-Gómez M, Giraldo A, Lavaniegos BE (2019) Structure of euphausiid assemblages in the Eastern Tropical Pacific off Colombia during el Niño, La Niña and neutral conditions. J Exp Mar Biol Ecol 516:1–15. https://doi.org/10.1016/j.jembe.2019.04.002
Roden GI (1971) Aspects of the transition zone in the northeastern Pacific. J Geophys Res 76:3462–3475
Roger C (1994) The plankton of the tropical western Indian ocean as a biomass indirectly supporting surface tunas (yellowfin, Thunnus albacares and skipjack, Katsuwonus pelamis). Environ Biol Fishes 39:161–172. https://doi.org/10.1007/BF00004934
Rohde K (1992) Latitudinal gradients in species diversity: the search for the primary cause. Oikos 65:514–527. https://doi.org/10.2307/3545569
Rombouts I, Beaugrand G, Ibaňez F, Gasparini S, Chiba S, Legendre L (2009) Global latitudinal variations in marine copepod diversity and environmental factors. Proc R Soc Lond B Biol Sci 276:3053–3062. https://doi.org/10.1098/rspb.2009.0742
Rousseaux CS, Gregg WW (2012) Climate variability and phytoplankton composition in the Pacific Ocean. J Geophys Res Oceans 117:C10006. https://doi.org/10.1029/2012JC008083
Rutherford S, D'Hondt S, Prell W (1999) Environmental controls on the geographic distribution of zooplankton diversity. Nature 400:749–753. https://doi.org/10.1038/23449
Sato T, Shiozaki T, Hashihama F et al (2022) Low nitrogen fixation related to shallow nitracline across the Eastern Indian Ocean. J Geophys Res Biogeosci 127:e2022JG007104. https://doi.org/10.1029/2022JG007104
Seibel BA, Schneider JL, Kaartvedt S, Wishner KF, Daly KL (2016) Hypoxia tolerance and metabolic suppression in oxygen minimum zone euphausiids: implications for ocean deoxygenation and biogeochemical cycles. Integr Comp Biol 56:510–523
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Shaw JJ (2023) Hansarsia nom. nov., a replacement name for Nematoscelis G.O. Sars, 1883 (Crustacea: Euphausiidae), a junior homonym of Nematoscelis Wollaston, 1867 (Coleoptera: Staphylinidae: Aleocharinae). Zootaxa 5256:84–86
Simpson EH (1949) Measurement of diversity. Nature 163:688
Steinberg DK, Cope JS, Wilson SE, Kobari T (2008) A comparison of mesopelagic mesozooplankton community structure in the subtropical and subarctic North Pacific Ocean. Deep-Sea Res II: Top Stud Oceanogr 55:1615–1635. https://doi.org/10.1016/j.dsr2.2008.04.025
Stramma L, Bange HW, Czeschel R, Lorenzo A, Frank M (2013) On the role of mesoscale eddies for the biological productivity and biogeochemistry in the eastern tropical Pacific Ocean off Peru. Biogeosciences 10:7293–7306. https://doi.org/10.5194/bg-10-7293-2013
Stuart V, Pillar SC (1990) Diel grazing patterns of all ontogenetic stages of Euphausia lucens and in situ predation rates on copepods in the southern Benguela upwelling region. Mar Ecol Prog Ser 64:227–241
Sutton AL, Beckley LE (2016) Influence of the Leeuwin Current on the epipelagic euphausiid assemblages of the south-east Indian Ocean. Hydrobiologia 779:193–207. https://doi.org/10.1007/s10750-016-2814-7
Sutton AL, Beckley LE (2017) Species richness, taxonomic distinctness and environmental influences on euphausiid zoogeography in the Indian Ocean. Diversity 9:23. https://doi.org/10.3390/d9020023
Tremblay N, Gómez-Gutiérrez J, Zenteno-Savín T, Robinson CJ, Sánchez-Velascoa L (2010) Role of oxidative stress in seasonal and daily vertical migration of three krill species in the Gulf of California. Limnol Oceanogr 55:2570–2584. https://doi.org/10.4319/lo.2010.55.6.2570
Tremblay N, Hünerlage K, Werner T (2020) Hypoxia tolerance of 10 Euphausiid species in relation to vertical temperature and oxygen gradients. Front Physiol 11:248. https://doi.org/10.3389/fphys.2020.00248
Werner T, Buchholz F (2013) Diel vertical migration behaviour in Euphausiids of the northern Benguela current: seasonal adaptations to food availability and strong gradients of temperature and oxygen. J Plankton Res 35:792–812. https://doi.org/10.1093/plankt/fbt030
Wiebe PH, Lawson GL, Lavery AC, Copley NJ, Horgan E, Bradley A (2013) Improved agreement of net and acoustical methods for surveying euphausiids by mitigating avoidance using a net-based LED strobe light system. ICES J Mar Sci 70:650–664. https://doi.org/10.1093/icesjms/fst005
Wiebe PH, Bucklin A, Kaartvedt S, Røstad A, Blanco-Bercial L (2016) Vertical distribution and migration of euphausiid species in the Red Sea. J Plankton Res 38:888–903. https://doi.org/10.1093/plankt/fbw038
Yamamura O, Yasuma H (2010). 3 Second Micronekton Inter-calibration Experiment, MIE-2. PICES Scientific Report No. 38 2010, 37.
Yoon WD, Yang JY, Lim D, Cho SH, Park GS (2006) Species composition and spatial distribution of euphausiids of the yellow sea and relationships with environmental factors. Ocean Sci J 41:11–29. https://doi.org/10.1007/BF03022402
Zar JH (1999) Biostatistical analysis. Pearson Education India, Noida
Zhou F, Hirai J, Hamasaki K, Horii S, Tsuda A (2021) Feeding ecology of three euphausiid species in the North Pacific ocean inferred from 18S V9 metabarcoding and stable isotope analysis. Front Mar Sci 8:756067. https://doi.org/10.3389/fmars.2021.756067
Acknowledgements
We thank the captains and all members of the cruises KH17-4, KH18-6, and KH19-6 on board the RV Hakuho-Maru for their assistance with sample collection. We also thank our two reviewers for their significant contribution to providing valuable comments in improving this paper. This study was supported by the Environment Research and Technology Development Fund (JPMEERF20224R03) of the Environmental Restoration and Conservation Agency provided by the Ministry of the Environment of Japan. This work was also supported by JST SPRING, Grant Number JPMJSP2108.
Funding
Open Access funding provided by The University of Tokyo.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhou, F., Hirai, J., Sato, T. et al. Primary productivity impacts community structure of euphausiids in the low-latitude Indian and Pacific Oceans. J Oceanogr (2024). https://doi.org/10.1007/s10872-024-00713-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10872-024-00713-z