Abstract
Crystallography reveals the rotomer in the salt isolated from EtOH solution has an anti disposition of the OH and imine-NH hydrogen atoms while that from EtOCH2CH2OH solution has a syn disposition. Both molecules have an E configuration about the C=N bond and are essentially planar (dihedral angles between aromatic groups = 2.4 (3)° and 6.42 (9)°, respectively). The anti-rotomer crystallizes in the monoclinic space group P21/n with a = 10.8795 (10) Å, b = 10.7730 (11) Å, c = 13.1257 (7) Å, λ = 104.806 (5)°, and Z = 4. The syn-rotomer crystallizes in the monoclinic space group C2/c with a = 14.4610 (5) Å, b = 11.3073 (5) Å, c = 17.8849 (7) Å, β = 91.904 (2)°, and Z = 8.
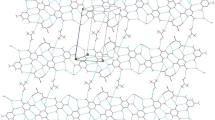
Graphical Abstract
Planar anti- and syn-rotomers of the title compound feature intramolecular O–H···N or intermolecular O–H···Cl hydrogen bonds, respectively, and two- and three-dimensional supramolecular architectures, respectively.





Similar content being viewed by others
References
Font M, Monge A, Ruiz I, Heras B (1997) Drug Des Discov 14:59
Nakamura T, Oka M, Aizawa K, Soda H, Fukuda M, Terashi K, Ikeda K, Mizuta Y, Noguchi Y, Kimura Y, Tsuruo T, Kohno S (1999) Biochem Biophys Res Commun 255:618
Montenegro RC, Lotufo LV, de Moraes MO, Pessoa CO, Rodriques FAR, Bispo MLF, Cardoso LNF, Kaiser CR, de Souza MVN (2011) Med Chem 7:599
Kaminsky D, Meltzer RI (1968) J Med Chem 11:160
de Souza MVN, Pais KC, Kaiser CR, Peralta MA, Ferreira ML, Lourenco MCS (2007) Bioorg Med Chem 17:1474
Candea ALP, Ferreira ML, Pais KC, Cardoso LNF, Kaiser CR, Henriques MGMO, Lourenco MCS, Bezerra FAFM, de Souza MVN (2009) Bioorg Med Chem Lett 19:6272
de Souza MVN, Vasconcelos TA, Cardoso SH, Almeida MV (2006) Curr Med Chem 13:455
Rustomjee R, Diacon AH, Allen J, Venter A, Reddy C, Patientia RF, Mthiyane TCP, de Marez T, Van Heeswijk R, Kerstens R, Koul A, de Beule K, Donald PR, McNeeley DF (2008) Antimicrob Agents Chemother 52:2831
Ferreira ML, Gonçalves RSB, Cardoso LNF, Kaiser CR, Candéa ALP, Henriques MGMO, Lourenço MCS, Bezerra FAFM, de Souza MVN (2010) ScientificWorldJournal 10:1347–1355
Musiol R, Jampilek J, Buchta V, Silva L, Halina H, Podeszwa B, Palka A, Majerz-Maniecka K, Oleksyn B, Polanski J (2006) Bioorg Med Chem 14:359
Warshakoon NC, Sheville J, Bhatt RT, Ji W, Mendez-Andino JL, Meyers KM, Kim N, Wos JA, Mitchell C, Paris JL, Pinney BB, Reizes O, Hu XE (2006) Bioorg Med Chem Lett 16:5207
Sloboda AE, Powell D, Poletto JF, Pickett WC, Gibbons JJ, Bell DH, Oronsky AL, Kerwar SJ (1991) J Rheumatol 18:855
Kaiser CR, Pais KC, de Souza MVN, Wardell JL, Wardell SMSV, Tiekink ERT (2009) CrystEngComm 11:1133
Goncalves RSB, Kaiser CR, de Souza MVN, Wardell JL, Wardell SMSV, Howie RA (2011) Z Kristallogr 226:793
Wardell JL, de Souza MVN, Wardell SMSV, Lourenço MCS (2011) J Mol Struct 990:67
Wardell SMSV, Wardell JL, Skakle JMS, Tiekink ERT (2011) Z Kristallogr 226:68
Goncalves RSB, Kaiser CR, Lourenco MCS, Bezerra FAFM, de Souza MVN, Wardell JL, Wardell SMSV, Henriques MGMO, Costa T (2012) Bioorg Med Chem 20:243
Kaiser CR, Pais KC, de Souza MVN, Wardell JL, Wardell SMSV, Tiekink ERT (2009) CrystEngComm 11:1133
Tiekink ET, Wardell SMSV, Wardell JL, Ferreira ML, de Souza MVN, Kaiser CR (2012) Acta Crystallogr E68:o1850
Wardell SMSV, Tiekink ERT, Wardell JL, Ferreira ML, de Souza MVN (2012) Acta Crystallogr E68:o1232
Sheldrick GM (2007) SADABS. University of Göttingen, Göttingen
Hooft RWW (1998) COLLECT data collection software. Nonius B.V, Delft
Otwinowski Z, Minor W (1997) In: Carter CW Jr, Sweet RM (eds) Methods in enzymology, vol 276: macromolecular crystallography, part A. Academic Press, New York, pp 307–326
Sheldrick GM (2008) Acta Crystallogr A64:211
Farrugia LJ (2012) J Appl Crystallogr 45:849
Gans J, Shalloway D (2001) J Mol Graph Model 19:557
Brandenburg K (2006) DIAMOND. Version 3.1c. Crystal Impact GbR, Postfach 1251, D-53002 Bonn, Germany
Spek AL (2003) J Appl Crystallogr 36:7
Ferreira ML, de Souza MVN, Wardell SMSV, Tiekink ERT, Wardell JL (2012) Acta Crystallogr E68:o1214
de Souza MVN, Ferreira ML, Wardell SMSV, Tiekink ERT, Wardell JL (2012) Acta Crystallogr E68:o1244
Ferreira ML, de Souza MVN, Howie RA, Tiekink ERT, Wardell JL, Wardell SMSV (2010) Acta Crystallogr E66:o696
de Souza MVN, Howie RA, Tiekink ERT, Wardell JL, Wardell SMSV (2010) Acta Crystallogr E66:o152
de Souza MVN, Howie RA, Tiekink ERT, Wardel JL, Wardell SMSV, Kaiser CR (2010) Acta Crystallogr E66:o698
Ferreira ML, de Souza MVN, Howie RA, Tiekink ERT, Wardell JL, Wardell SMSV (2009) Acta Crystallogr E65:o3239
Allen FH (2002) Acta Crystallogr B58:380
Yathirajan HS, Sarojini BK, Narayana B, Sunil K, Bolte M (2007) Acta Crystallogr E63:o2720
Jasinski JP, Butcher RJ, Narayana B, Sunil K, Yathirajan HS (2008) Acta Crystallogr E64:o481
de Souza MVN, Howie RA, Tiekink ERT, Wardell JL, Wardell SMSV (2009) Acta Crystallogr E65:o3204
Acknowledgments
Support from the Ministry of Higher Education, Malaysia, High-Impact Research scheme (UM.C/HIR-MOHE/SC/12) and the University of Malaya (UMRG125) for ERTT, and CNPq, Brazil for JLW is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Lima Ferreira, M., Milne, B.F., de Souza, M.V.N. et al. Structures of Two Rotamers of the HCl Salt of 4-Methyl-2-[(1E)-[2-(quinolin-4-yl)hydrazin-1-ylidene]methyl]phenol: Different Supramolecular Arrays. J Chem Crystallogr 44, 36–41 (2014). https://doi.org/10.1007/s10870-013-0481-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-013-0481-3