Abstract
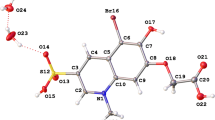
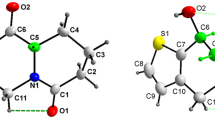
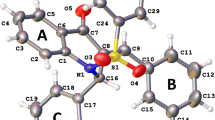
The structures of sulphur atom tethered quinone containing flexible carboxylic acid (3-methyl-1,4-dioxo-1,4-dihydronaphthalen-2-ylsulfanyl)acetic acid (1) and its molecular complex with 4,4′-bipyridine (3) are determined. The compound 1 crystallizes in P-1 (triclinic, a = 7.5378(6) Å, b = 7.6413(7) Å, c = 10.3101(9) Å; α = 89.779 (7)°, β = 81.042 (5)°, γ = 89.101(7)°) and the molecular complex 3 crystallises in P2(1)/n (monoclinic, a = 9.3383(7) Å, b = 3.970(3) Å, c = 42.130(3) Å, β = 91.056(5)°) space groups, respectively. The R 22 (8) type hydrogen bonding between dicarboxylic acid groups present in the parent compound 1 is lost on interaction with 4, 4′-bipyridine; in the molecular complex 3 R 22 (7) type of O···H–C and O–H···N interactions are present between the pyridine rings and carboxylic acid groups. The molecular complex (4) derived from 3-carboxymethylsulfanyl-1,4-dihydroxynaphthalen-2-yl-sulfanyl) acetic acid (2) with triphenylphosphine oxide in 1:2 ratio, crystallises in C2/c space group have monoclinic, a = 26.0494(13) Å, b = 10.5402(5) Å, c = 17.1023(8) Å, β = 108.719 (5)°). The triphenylphosphine oxide molecules are preferentially held by O–H···O interactions between carboxylic acid and P=O bond.
Graphical Abstract
The structures of (3-methyl-1,4-dioxo-1,4-dihydronaphthalen-2-ylsulfanyl) acetic acid , its molecular complex with 4,4′-bipyridine and molecular complex of 3-carboxymethylsulfanyl-1,4-dihydroxy naphthalen-2-yl-sulfanyl)acetic acid with triphenylphosphine oxide are presented






Similar content being viewed by others
References
Du M, Zhang Z-H, Zhao X-J, Cai H (2006) Cryst Growth Des 6:114
Beatty AM, Schneider CM, Simpson AE, Zaher JL (2002) Cryst Eng Comm 4:282
Du M, Zhang Z-H, Zhao X-J (2006) Cryst Growth Des 6:335
Pedireddi VR, Chaterjee S, Ranganthan A, Rao CNR (1998) Tetrahedron 54:9457
Xiao J, Yang M, Lauher JW, Fowler FW (2000) Angew Chem Int Ed 39:2132
Aakeröy CB, Beatty AM, Helfrich BA, Niuwenhuyzen M (2003) Cryst Growth Des 3:159
Vishweshwar P, Nangia A, Lynch VM (2003) Cryst Growth Des 3:783
Braga D, Maini L, Sanctis G, Rubini K, Grepioni F, Chierotti MR, Gobetto R (2003) Chem Eur J 9:5538
Singh D, Bhattacharyya P, Baruah JB (2010) Crystal Growth Des 10:348
Barooah N, Sarma RJ, Baruah JB (2006) Cryst Eng Comm 8:608
Erxleben A (2003) Coord Chem Rev 246:203
Fletchera AJ, Thomasa KM, Rosseinsky MJ (2005) J Solid State Chem 178:2491
Uemura K, Matsuda R, Kitagawa S (2005) J Solid State Chem 178:2420
Delgado FS, Sanchiz J, Ruiz-Perez C, Lloret F, Julve M (2004) Crys Eng Comm 6:73
Delgado FS, Hernandez-Molina M, Sanchiz J, Ruiz-Perez C, Rodriguez-Martin Y, Lopez T, Lloret F, Julve M (2004) Cryst Eng Comm 6:929
GildeMuro I, Lezama L, Insausti M, Rojo T (2004) Polyhedron 23:859
Delgado FS, Ruiz-Perez C, Sanchiz J, Lloret F, Julve M (2006) Cryst Eng Comm 8:530
Pasan J, Sanchiz J, Ruiz-Perez C, Lloret F, Julve M (2005) Inorg Chem 44:7794
Lightfoot P, Snedden A (1999) JCS Dalton Trans 3549
Rodriquez-Martin Y, Sanchiz J, Ruiz-Perez C, Lloret F, Julve M (2002) Cryst Eng Comm 4:631
Mondal P, Karmakar A, Singh WM, Baruah JB (2008) Cryst Eng Comm 10:1550
Karmakar A, Baruah JB, Shankar RB (2009) Cryst Eng Comm 11:832
Yaghi OM, O’keefe M, Ockwig NW, Chae HK, Eddaoudi M, Kim J (2003) Nature 423:705
Heller A, Feldman B (2008) Chem Rev 108:2482
Aubart KM, Heathcock CH (1999) J Org Chem 64:16
Huang Y, Zhang J, Pettus TRR (2005) Org Lett 7:26
Liu JK (2006) Chem Rev 106:2209
Tandon VK, Maurya HK, Yadav DB, Tripathi A, Kumar M, Shukla PK (2006) Bioorg Med Chem Lett 16:5883
Benites J, Rojo L, Valderrama JA, Taper H, Calderon PB (2008) Eur J Med Chem 43:1813
Eyong KO, Kumar PS, Kuete V, Folefoc GN, Nkengfack EA, Baskaran S (2008) Bioorg Med Chem Lett 18:5387
O’Brien PJ (1991) Chem Biol Interact 80:1
Tucker JHR, Collinson SR (2002) Chem Soc Rev 31:147
Lehn JM (1995) Supramolecular chemistry, concepts and perspectives. VCH, Weinheim
Katz E, Baron R, Willner I (2005) J Am Chem Soc 127:4060
Hogan CF, Hutchison JA, Ghiggino KP, Langford SJ, Latter JM, Lau VL, Murali V (2005) Supramol Chem 17:513
Lee J-Y, Park S-M (1998) J Phys Chem B 102:9940
Cooke G, Sindelar V, Rotello VM (2003) Chem Commun 752
Singh WM, Baruah JB (2009) J Mol Struct 931:82
Bernstein J, Davis RE, Shimoni L, Chang NL (1995) Angew Chem Int Ed 34:1555
Sheldrick GM (2008) Acta Cryst A64:112
Bauscher M, Maentele W (1992) J Phys Chem 96:11101
Miller GY, Ouimet L, Smith TDK (2000) J Org Chem 65:8831
Amatore C, Capobianco G, Farnia G, Sandona G, Saveant J-M (1985) J Am Chem Soc 107:1815
Acknowledgments
The authors thank Council of Scientific and Industrial Research, New-Delhi, India for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marjit Singh, W., Baruah, J.B. Characterization of Molecular Complexes of 1,4-Naphthoquinone Derivatives. J Chem Crystallogr 41, 952–958 (2011). https://doi.org/10.1007/s10870-011-0024-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-011-0024-8