Abstract
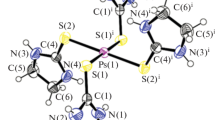
Equimolar amounts of the proton acceptor 2-aminopyridine 1 and proton donor picolinic acid 2 were solvent-free grinded to give complex 5. X-ray diffraction was used to determine the crystal structure of the new complex 5 that found to be ionic, with proton transfer occurring to the aromatic nitrogen of the 2-aminopyridine moiety. Nonclassical hydrogen bonding exist in the solid state structure of 5, such as, 2-aminopyridine-carboxylic acid supramolecular heterosynthon was carefully investigated and compared to those exist in cocrystals in terms of supramolecular chemistry and crystal engineering. Complex 5 crystallizes in the monoclinic P2(1)/n space group with unit cell parameters of: a = 8.7714(12) Å, b = 12.1579(17) Å, c = 10.8546(15) Å, with α = 90°, β = 110.100(4)°, γ = 90°, and Z = 4.
Graphical Abstract
Equimolar amounts of the proton acceptor 2-aminopyridine 1 and proton donor picolinic acid 2 were solvent-free grinded to give complex 5. X-ray diffraction was used to determine the crystal structure of the new complex 5 that found to be ionic, with proton transfer occurring to the aromatic nitrogen of the 2-aminopyridine moiety. Nonclassical hydrogen bonding exist in the solid state structure of 5, such as, 2-aminopyridine-carboxylic acid supramolecular heterosynthon was carefully investigated and compared to those exist in cocrystals in terms of supramolecular chemistry and crystal engineering.











Similar content being viewed by others
References
Mac Donald JC, Dorrestein PC, Pilley MM (2001) Crystallogr Growth Des 1(1):29
Glaser R, Lewis M (1999) Org Lett 1(2):273
Semo N, Osheroff MA, Koskl WSJ (1983) Phys Chem 87:2302
Ramirez FHeJ, Lebrilla CB (1999) J Am Chem Sos 121:4726
Tarkka RM, Zhang X, Jenekhe SA (1996) J Am Chem Soc 118:9438
Nogami M, Diako Y, Akai T, Kasuga T (2001) J Phys Chem B105:4653
Ramezanipour F, Aghabozorg H, Shokrollahi A, Shamsipur M, Stoeckli-Evans H, Soleimannejad J, Sheshmani SJ (2005) Mol Struct 779:77
Smith G, Wermuth UD, Healy PC (2006) J Chem Crystallogr 36(12):841
Nishimura K, Khasanov SS, Saito G (2002) J Mater Chem 12:1693
Aghabozorg H, Ramezanipour F, Nakhjavan B, Soleimannejad J, Gharamaleki JA, Sharif MA (2007) Cryst Res Technol 42(11):1137
Moghimi A, Aghabozorg H, Sheshmani S, Kickelbick G, Soleimannejad (2005) J Anal Sci 21:141
Aghabozorg H, Saei AA, Ramezanipour F (2005) Acta Cryst E 61:o3242
Moghami A, Shokrollahi M, Shamsipur M, Aghabozorg H, Ranjbar M (2004) J Mol Struct 701:49
Alshahateet SF (2010) J Chem Crystallogr 40:191
Alshahateet SF (2010) J Chem Crystallogr. doi:10.1007/s10870-010-9872-x
Alshahateet SF (2010) Mol Cryst Liquid Cryst 533:1
Zaghal MH, Qaseer HA, El-Qisairi AK, Alshahateet SF, Shatnawi MY, Dawe LN (2009) J Chem Crystallogr 39:564
Alshahateet SF, Bishop R, Craig DC, Scudder ML (2010) Cryst Growth Des 10:1842
Alshahateet SF, Bishop R, Craig DC, Kooli F, Scudder ML (2008) CrystEngComm 10:297
Alshahateet SF (2010) JJC 5(4):317
Alghezawi NM, Alshahateet SF, Bishop R (2010) JJC 5(3):211
Sekhon BS (2009) Ars Pharm 50(3):99
Reddy LS, Basavoju S, Vangala VR, Nangia A (2006) Cryst Growth Des 6(1):161
Bis JA, Zaworotko MJ (2005) Cryst Growth Des 5(3):1169
Stahly GP (2007) Cryst Growth Des 7:1007
Tan T-F, Han J, Pang M-L, Song H-B, Ma Y-X, Meng J-B (2006) Cryst Growth Des 6(5):1186
Siemens Energy and Automation, Inc (1996) SMART and SAINT software reference manuals, version 4.0. Analytical Instrumentation, Madison, WI
Sheldrick M (1996) SADABS software for empirical absorption correction. University of Göttingen, Gottingen
Siemens Energy and Automation, Inc (1996) SHELXTL reference manuals, version 5.03. Analytical Instrumentation, Madison, WI
Boenigk D, Mootz D (1988) J Am Chem Soc 110:2135
Cowan JA, Howard JAK, Mclntyre GJ, Lo SM-F, Williams ID (2003) Acta Crystallogr B59:794
Bernstein J, Dunitz JD, Gavezzotti A (2008) Cryst Growth Des 8(6):2011
Borthwick PW (1980) Acta Crystallogr B36:628
Bis JA, McLaughlin OL, Vishweshwar P, Zaworotko MJ (2006) Cryst Growth Des 6(12):2648
Etter MC (1982) J Am Chem Soc 104:1095
Etter MC (1991) J Phys Chem 95:4601
Etter MC (1990) Acc Chem Res 23:120
Acknowledgments
The authors thank Mutah University for support needed to finish this work, Prof. Koh Lip Lin and Geok Kheng Tan from the National University of Singapore for their technical support and assistance. Many thanks are also due to Prof. M. J. Zaworotko (University of South Florida) for his great help and unlimited support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alshahateet, S.F., Alghezawi, N.M. Crystallographic Analyses of High-Z Value Structure of a Pyridinium–Carboxylate Complex. J Chem Crystallogr 41, 708–714 (2011). https://doi.org/10.1007/s10870-010-9958-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-010-9958-5