Abstract
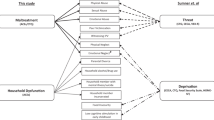
Childhood adversity is linked to psychological, behavioral, and physical health problems, including obesity and cardiometabolic disease. Epigenetic alterations are one pathway through which the effects of early life stress and adversity might persist into adulthood. Epigenetic mechanisms have also been proposed to explain why cardiometabolic health can vary greatly between individuals with similar Body Mass Index (BMIs). We evaluated two independent cross-sectional cohorts of adults without known medical illness, one of which explicitly recruited individuals with early life stress (ELS) and control participants (n = 195), and the other a general community sample (n = 477). In these cohorts, we examine associations between childhood adversity, epigenetic aging, and metabolic health. Childhood adversity was associated with increased GrimAge Acceleration (GAA) in both cohorts, both utilizing a dichotomous yes/no classification (both p < 0.01) as well as a continuous measure using the Childhood Trauma Questionnaire (CTQ) (both p < 0.05). Further investigation demonstrated that CTQ subscales for physical and sexual abuse (both p < 0.05) were associated with increased GAA in both cohorts, whereas physical and emotional neglect were not. In both cohorts, higher CTQ was also associated with higher BMI and increased insulin resistance (both p < 0.05). Finally, we demonstrate a moderating effect of BMI on the relationship between GAA and insulin resistance where GAA correlated with insulin resistance specifically at higher BMIs. These results, which were largely replicated between two independent cohorts, suggest that interactions between epigenetics, obesity, and metabolic health may be important mechanisms through which childhood adversity contributes to long-term physical and metabolic health effects.


Similar content being viewed by others
References
Adam, T. C., & Epel, E. S. (2007). Stress, eating and the reward system. Physiology and Behavior, 91, 449–458.
American Psychiatric, A.,& American Psychiatric, A., (2000) Task Force on D-I. Diagnostic and statistical manual of mental disorders : DSM-IV-TR. Washington, DC: American Psychiatric Association.
Andrade, S., Morais, T., Sandovici, I., Seabra, A. L., Constância, M., & Monteiro, M. P. (2021). Adipose tissue epigenetic profile in obesity-related dysglycemia - A systematic review. Front Endocrinol (lausanne), 12, 681649.
Baker, M. (2016). 1,500 scientists lift the lid on reproducibility. Nature, 533, 452–454.
Bell, C. G., Lowe, R., Adams, P. D., et al. (2019). DNA methylation aging clocks: Challenges and recommendations. Genome Biology, 20, 249.
Bellis, M. A., Hughes, K., Ford, K., Ramos Rodriguez, G., Sethi, D., & Passmore, J. (2019). Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: A systematic review and meta-analysis. Lancet Public Health, 4, e517–e528.
Bernstein, D. P., Stein, J. A., Newcomb, M. D., et al. (2003). Development and validation of a brief screening version of the childhood trauma questionnaire. Child Abuse and Neglect, 27, 169–190.
Bifulco, A., Brown, G. W., & Harris, T. O. (1994). Childhood Experience of care and abuse (CECA): A retrospective interview measure. Journal of Child Psychology and Psychiatry., 35, 1419–1435.
Bifulco, A., Brown, G. W., Lillie, A., & Jarvis, J. (1997). Memories of childhood neglect and abuse: Corroboration in a series of sisters. Journal of Child Psychology and Psychiatry., 38, 365–374.
Block, J. P., He, Y., Zaslavsky, A. M., Ding, L., & Ayanian, J. Z. (2009). Psychosocial stress and change in weight among US adults. American Journal of Epidemiology, 170, 181–192.
Boison, D. (2017). New insights into the mechanisms of the ketogenic diet. Current Opinion in Neurology, 30, 187–192.
Bray, G. A., Heisel, W. E., Afshin, A., et al. (2018). The science of obesity management: An endocrine society scientific statement. Endocrine Reviews, 39, 79–132.
Bremne, J. D., & Vermetten, E. (2001). Stress and development: Behavioral and biological consequences. Development and Psychopathology, 13, 473–489.
Camhi, S. M., Whitney Evans, E., Hayman, L. L., Lichtenstein, A. H., & Must, A. (2015). Healthy eating index and metabolically healthy obesity in U.S. adolescents and adults. Preventive Medicine, 77, 23–27.
Cao-Lei, L., Dancause, K. N., Elgbeili, G., et al. (2015). DNA methylation mediates the impact of exposure to prenatal maternal stress on BMI and central adiposity in children at age 13½ years: Project Ice Storm. Epigenetics, 10, 749–761.
Chandraratne, N. K., Fernando, A. D., & Gunawardena, N. (2018). Physical, sexual and emotional abuse during childhood: Experiences of a sample of Sri Lankan young adults. Child Abuse and Neglect, 81, 214–224.
Chao, A. M., Jastreboff, A. M., White, M. A., Grilo, C. M., & Sinha, R. (2017). Stress, cortisol, and other appetite-related hormones: Prospective prediction of 6-months changes in food cravings and weight. Obesity (silver Spring), 25, 713–720.
Chen, M. A., LeRoy, A. S., Majd, M., et al. (2021). Immune and epigenetic pathways linking childhood adversity and health across the lifespan. Frontiers in Psychology, 12, 788351.
Chen, X. Y., Lo, C. K. M., Chan, K. L., Leung, W. C., & Ip, P. (2022). Association between childhood exposure to family violence and telomere length: A meta-analysis. International Journal of Environmental Research and Public Health, 19, 12151.
Colich, N. L., Rosen, M. L., Williams, E. S., & McLaughlin, K. A. (2020). Biological aging in childhood and adolescence following experiences of threat and deprivation: A systematic review and meta-analysis. Psychological Bulletin, 146, 721–764.
Copeland, W. E., Shanahan, L., McGinnis, E. W., Aberg, K. A., & van den Oord, E. (2022). Early adversities accelerate epigenetic aging into adulthood: A 10-year, within-subject analysis. Journal of Child Psychology and Psychiatry, 63, 1308–1315.
Cribb, L., Hodge, A. M., Yu, C., et al. (2022). Inflammation and epigenetic aging are largely independent markers of biological aging and mortality. Journals of Gerontology. Series a, Biological Sciences and Medical Sciences, 77, 2378–2386.
Daigre, C., Rodríguez-Cintas, L., Tarifa, N., et al. (2015). History of sexual, emotional or physical abuse and psychiatric comorbidity in substance-dependent patients. Psychiatry Research, 229, 743–749.
Dallman, M. F., Pecoraro, N. C., & la Fleur, S. E. (2005). Chronic stress and comfort foods: Self-medication and abdominal obesity. Brain, Behavior, and Immunity, 19, 275–280.
Daniels, T. E., Mathis, K. J., Gobin, A. P., et al. (2023). Associations of early life stress with leptin and ghrelin in healthy young adults. Psychoneuroendocrinology, 149, 106007.
Dayeh, T., Tuomi, T., Almgren, P., et al. (2016). DNA methylation of loci within ABCG1 and PHOSPHO1 in blood DNA is associated with future type 2 diabetes risk. Epigenetics, 11, 482–488.
Duffy, K. A., McLaughlin, K. A., & Green, P. A. (2018). Early life adversity and health-risk behaviors: Proposed psychological and neural mechanisms. Annals of the New York Academy of Sciences, 1428, 151–169.
Essex, M. J., Boyce, W. T., Hertzman, C., et al. (2013). Epigenetic vestiges of early developmental adversity: Childhood stress exposure and DNA methylation in adolescence. Child Development, 84, 58–75.
Evans, G. W., Li, D., & Whipple, S. S. (2013). Cumulative risk and child development. Psychological Bulletin, 139, 1342–1396.
Fogelman, N., & Canli, T. (2019). Early life stress, physiology, and genetics: A review. Frontiers in Psychology, 10, 1668.
Föhr, T., Waller, K., Viljanen, A., et al. (2021). Does the epigenetic clock GrimAge predict mortality independent of genetic influences: An 18 year follow-up study in older female twin pairs. Clinical Epigenetics, 13, 128.
Fumagalli, F., Molteni, R., Racagni, G., & Riva, M. A. (2007). Stress during development: Impact on neuroplasticity and relevance to psychopathology. Progress in Neurobiology, 81, 97–217.
Gayer-Anderson, C., Reininghaus, U., Paetzold, I., et al. (2020). A comparison between self-report and interviewer-rated retrospective reports of childhood abuse among individuals with first-episode psychosis and population-based controls. Journal of Psychiatric Research, 123, 145–150.
Gutiérrez-Repiso, C., Linares-Pineda, T. M., Gonzalez-Jimenez, A., et al. (2021). Epigenetic biomarkers of transition from metabolically healthy obesity to metabolically unhealthy obesity phenotype: A prospective study. International Journal of Molecular Sciences, 22, 10417.
Hamlat, E. J., Prather, A. A., Horvath, S., Belsky, J., & Epel, E. S. (2021). Early life adversity, pubertal timing, and epigenetic age acceleration in adulthood. Developmental Psychobiology, 63, 890–902.
Hamlat, E. J., Neilands, T. B., Laraia, B., et al. (2023). Early life adversity predicts an accelerated cellular aging phenotype through early timing of puberty. Psychological Medicine, 53, 7720–7728.
Han, L. K. M., Aghajani, M., Clark, S. L., et al. (2018). Epigenetic aging in major depressive disorder. American Journal of Psychiatry, 175, 774–782.
Harvanek, Z. M., Fogelman, N., Xu, K., & Sinha, R. (2021). Psychological and biological resilience modulates the effects of stress on epigenetic aging. Translational Psychiatry, 11, 601.
Harvanek, Z. M., Boks, M. P., Vinkers, C. H., & Higgins-Chen, A. T. (2023). The cutting edge of epigenetic clocks: in search of mechanisms linking aging and mental health. Biological Psychiatry, 94, 694–705.
Heim, C., Newport, D. J., Heit, S., et al. (2000). Pituitary-adrenal and autonomic responses to stress in women after sexual and physical abuse in childhood. JAMA, 284, 592–597.
Higgins-Chen, A. T., Thrush, K. L., Wang, Y., et al. (2022). A computational solution for bolstering reliability of epigenetic clocks: Implications for clinical trials and longitudinal tracking. Nature Aging, 2, 644–661.
Horvath, S. (2013). DNA methylation age of human tissues and cell types. Genome Biology, 14, R115.
Horvath, S., & Raj, K. (2018). DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nature Reviews Genetics, 19, 371–384.
Hostinar, C. E., Nusslock, R., & Miller, G. E. (2018). Future directions in the study of early-life stress and physical and emotional health: Implications of the neuroimmune network hypothesis. Journal of Clinical Child and Adolescent Psychology, 47, 142–156.
Houseman, E. A., Accomando, W. P., Koestler, D. C., et al. (2012). DNA methylation arrays as surrogate measures of cell mixture distribution. BMC Bioinformatics, 13, 86.
Iacobini, C., Pugliese, G., Blasetti Fantauzzi, C., Federici, M., & Menini, S. (2019). Metabolically healthy versus metabolically unhealthy obesity. Metabolism, 92, 51–60.
Ikeda, Y., Suehiro, T., Nakamura, T., Kumon, Y., & Hashimoto, K. (2001). Clinical significance of the insulin resistance index as assessed by homeostasis model assessment. Endocrine Journal, 48, 81–86.
Joshi, D., Gonzalez, A., Lin, D., & Raina, P. (2023). The association between adverse childhood experiences and epigenetic age acceleration in the Canadian longitudinal study on aging (CLSA). Aging Cell, 22, e13779.
Jung, J., McCartney, D. L., Wagner, J., et al. (2023). Additive effects of stress and alcohol exposure on accelerated epigenetic aging in alcohol use disorder. Biological Psychiatry, 93, 331–341.
Jylhävä, J., Pedersen, N. L., & Hägg, S. (2017). Biological Age Predictors. eBioMedicine, 21, 29–36.
Kalinowski, J., Huang, Y., Rivas, M. A., et al. (2022). Stress overload and dna methylation in african american women in the intergenerational impact of genetic and psychological factors on blood pressure study. Epigenet Insights., 15, 25168657221126310.
Kho, M., Wang, Y. Z., Chaar, D., et al. (2021). Accelerated DNA methylation age and medication use among African Americans. Aging (albany NY)., 13, 14604–14629.
Kim, K., Joyce, B. T., Zheng, Y., et al. (2021). DNA methylation GrimAge and incident diabetes: the coronary artery risk development in young adults (CARDIA) study. Diabetes, 70, 1404–1413.
Klopack, E. T., Crimmins, E. M., Cole, S. W., Seeman, T. E., & Carroll, J. E. (2022). Accelerated epigenetic aging mediates link between adverse childhood experiences and depressive symptoms in older adults: Results from the health and retirement study. SSM Popul Health., 17, 101071.
Leachman, J. R., Rea, M. D., Cohn, D. M., Xu, X., Fondufe-Mittendorf, Y. N., & Loria, A. S. (2020). Exacerbated obesogenic response in female mice exposed to early life stress is linked to fat depot-specific upregulation of leptin protein expression. American Journal of Physiology Endocrinology and Metabolism, 319, E852–E862.
Li, Z., He, Y., Wang, D., Tang, J., & Chen, X. (2017). Association between childhood trauma and accelerated telomere erosion in adulthood: A meta-analytic study. Journal of Psychiatric Research, 93, 64–71.
Li, M., Bao, L., Zhu, P., & Wang, S. (2022). Effect of metformin on the epigenetic age of peripheral blood in patients with diabetes mellitus. Frontiers in Genetics, 13, 955835.
Liang, X., Sinha, R., Justice, A. C., Cohen, M. H., Aouizerat, B. E., & Xu, K. (2022). A new monocyte epigenetic clock reveals nonlinear effects of alcohol consumption on biological aging in three independent cohorts (N = 2242). Alcoholism, Clinical and Experimental Research, 46, 736–748.
Ling, C., & Rönn, T. (2019). Epigenetics in human obesity and type 2 diabetes. Cell Metabolism, 29, 1028–1044.
Lu, A. T., Quach, A., Wilson, J. G., et al. (2019). DNA methylation GrimAge strongly predicts lifespan and healthspan. Aging (albany NY), 11, 303–327.
Luo, A., Jung, J., Longley, M., et al. (2020). Epigenetic aging is accelerated in alcohol use disorder and regulated by genetic variation in APOL2. Neuropsychopharmacology, 45, 327–336.
MacDonald, K., Thomas, M. L., Sciolla, A. F., et al. (2016). Minimization of childhood maltreatment is common and consequential: results from a large, multinational sample using the childhood trauma questionnaire. PLoS ONE, 11, e0146058.
Marini, S., Davis, K. A., Soare, T. W., et al. (2020). Adversity exposure during sensitive periods predicts accelerated epigenetic aging in children. Psychoneuroendocrinology, 113, 104484.
Marquez, F. D., Risica, P. M., Mathis, K. J., Sullivan, A., Gobin, A. P., & Tyrka, A. R. (2021). Do measures of healthy eating differ in survivors of early adversity? Appetite, 162, 105180.
Matthews, D. R., Hosker, J. P., Rudenski, A. S., Naylor, B. A., Treacher, D. F., & Turner, R. C. (1985). Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia, 28, 412–419.
McCrory, C., Fiorito, G., Hernandez, B., et al. (2021). GrimAge outperforms other epigenetic clocks in the prediction of age-related clinical phenotypes and all-cause mortality. Journals of Gerontology. Series a, Biological Sciences and Medical Sciences, 76, 741–749.
McCrory, C., Fiorito, G., O’Halloran, A. M., Polidoro, S., Vineis, P., & Kenny, R. A. (2022). Early life adversity and age acceleration at mid-life and older ages indexed using the next-generation GrimAge and pace of aging epigenetic clocks. Psychoneuroendocrinology, 137, 105643.
McGee, S. L., & Hargreaves, M. (2020). Exercise adaptations: Molecular mechanisms and potential targets for therapeutic benefit. Nature Reviews. Endocrinology, 16, 495–505.
Merz, M. P., & Turner, J. D. (2021). Is early life adversity a trigger towards inflammageing? Experimental Gerontology., 150, 111377.
Moore, L. D., Le, T., & Fan, G. (2013). DNA methylation and its basic function. Neuropsychopharmacology, 38, 23–38.
Moser, S., Martins, J., Czamara, D., Lange, J., Müller-Myhsok, B., & Erhardt, A. (2022). DNA-methylation dynamics across short-term, exposure-containing CBT in patients with panic disorder. Translational Psychiatry, 12, 46.
Murlasits, Z., Kupai, K., & Kneffel, Z. (2022). Role of physical activity and cardiorespiratory fitness in metabolically healthy obesity: A narrative review. BMJ Open Sport and Exercise Medicine, 8, e001458.
Nilsson, P. M., Korduner, J., & Magnusson, M. (2020). Metabolically healthy obesity (MHO)-new research directions for personalised medicine in cardiovascular prevention. Current Hypertension Reports, 22, 18.
Oblak, L., van der Zaag, J., Higgins-Chen, A. T., Levine, M. E., & Boks, M. P. (2021). A systematic review of biological, social and environmental factors associated with epigenetic clock acceleration. Ageing Research Reviews, 69, 101348.
Pedroso, J. A. B., Ramos-Lobo, A. M., & Donato, J., Jr. (2019). SOCS3 as a future target to treat metabolic disorders. Hormones (athens, Greece), 18, 127–136.
Pidsley, R., Zotenko, E., Peters, T. J., et al. (2016). Critical evaluation of the Illumina MethylationEPIC BeadChip microarray for whole-genome DNA methylation profiling. Genome Biology, 17, 208.
Puterman, E., Lin, J., Blackburn, E., O’Donovan, A., Adler, N., & Epel, E. (2010). The power of exercise: Buffering the effect of chronic stress on telomere length. PLoS ONE, 5, e10837.
Quach, A., Levine, M. E., Tanaka, T., et al. (2017). Epigenetic clock analysis of diet, exercise, education, and lifestyle factors. Aging (albany NY), 9, 419–446.
R (2020): A Language and Environment for Statistical Computing [computer program]. R Foundation for Statistical Computing.
Rampersaud, R., Protsenko, E., Yang, R., et al. (2022). Dimensions of childhood adversity differentially affect biological aging in major depression. Translational Psychiatry, 12, 431.
Ridout, K. K., Levandowski, M., Ridout, S. J., et al. (2018). Early life adversity and telomere length: A meta-analysis. Molecular Psychiatry, 23, 858–871.
Rohde, K., Keller, M., la Cour, P. L., Blüher, M., Kovacs, P., & Böttcher, Y. (2019). Genetics and epigenetics in obesity. Metabolism, 92, 37–50.
Samblas, M., Milagro, F. I., & Martínez, A. (2019). DNA methylation markers in obesity, metabolic syndrome, and weight loss. Epigenetics, 14, 421–444.
Schmitz, L. L., Duffie, E., Zhao, W., et al. (2023). Associations of early-life adversity with later-life epigenetic aging profiles in the multi-ethnic study of atherosclerosis. American Journal of Epidemiology, 192, 1991–2005.
Sheehan, D. V., Lecrubier, Y., Sheehan, K. H., et al. (1998). The Mini-International Neuropsychiatric Interview (MINI): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. Journal of Clinical Psychiatry., 59, 22–33.
Shenk, C. E., Felt, J. M., Ram, N., et al. (2022). Cortisol trajectories measured prospectively across thirty years of female development following exposure to childhood sexual abuse: Moderation by epigenetic age acceleration at midlife. Psychoneuroendocrinology, 136, 105606.
Sinha, R., & Jastreboff, A. M. (2013). Stress as a common risk factor for obesity and addiction. Biological Psychiatry, 73, 827–835.
Stefan, N., Häring, H. U., Hu, F. B., & Schulze, M. B. (2013). Metabolically healthy obesity: Epidemiology, mechanisms, and clinical implications. The Lancet Diabetes and Endocrinology, 1, 152–162.
Stevens, A. J., Rucklidge, J. J., & Kennedy, M. A. (2018). Epigenetics, nutrition and mental health. Is there a relationship? Nutritional Neuroscience, 21(9), 602–613.
Suderman, M., Borghol, N., Pappas, J. J., et al. (2014). Childhood abuse is associated with methylation of multiple loci in adult DNA. BMC Medical Genomics, 7, 13.
Torres, S. J., & Nowson, C. A. (2007). Relationship between stress, eating behavior, and obesity. Nutrition, 23, 887–894.
Tracy, E. L., Tracy, C. T., Kim, J. J., Yang, R., & Kim, E. (2020). Cascading effects of childhood abuse on physical health issues in later adulthood through trait anxiety and poor daily sleep quality. Journal of Health Psychology, 26, 2342–2348.
Tsatsoulis, A., & Paschou, S. A. (2020). Metabolically healthy obesity: criteria, epidemiology, controversies, and consequences. Current Obesity Reports, 9, 109–120.
Turner, R. J., Wheaton, B., & Lloyd, D. A. (1995). The epidemiology of social stress. American Sociological Review, 60(1), 104–125.
Tyrka, A. R., Ridout, K. K., & Parade, S. H. (2016). Childhood adversity and epigenetic regulation of glucocorticoid signaling genes: Associations in children and adults. Development and Psychopathology, 28, 1319–1331.
van Dijk, S. J., Tellam, R. L., Morrison, J. L., Muhlhausler, B. S., & Molloy, P. L. (2015). Recent developments on the role of epigenetics in obesity and metabolic disease. Clinical Epigenetics, 7, 66.
Viola, T. W., Salum, G. A., Kluwe-Schiavon, B., Sanvicente-Vieira, B., Levandowski, M. L., & Grassi-Oliveira, R. (2016). The influence of geographical and economic factors in estimates of childhood abuse and neglect using the childhood trauma questionnaire: A worldwide meta-regression analysis. Child Abuse and Neglect, 51, 1–11.
Walaszczyk, E., Luijten, M., Spijkerman, A. M. W., et al. (2018). DNA methylation markers associated with type 2 diabetes, fasting glucose and HbA(1c) levels: A systematic review and replication in a case-control sample of the Lifelines study. Diabetologia, 61, 354–368.
Wang, S. H., Chung, P. S., Lin, Y. P., et al. (2021). Metabolically healthy obesity and physical fitness in military males in the CHIEF study. Science and Reports, 11, 9088.
Wegman, H. L., & Stetler, C. (2009). A meta-analytic review of the effects of childhood abuse on medical outcomes in adulthood. Psychosomatic Medicine, 71, 805–812.
Wiss, D. A., & Brewerton, T. D. (2020). Adverse childhood experiences and adult obesity: A systematic review of plausible mechanisms and meta-analysis of cross-sectional studies. Physiology and Behavior, 223, 112964.
Wolfe, D. A., & McGee, R. (1994). Dimensions of child maltreatment and their relationship to adolescent adjustment. Development and Psychopathology, 6, 165–181.
Womersley, J. S., Nothling, J., Toikumo, S., et al. (2022). Childhood trauma, the stress response and metabolic syndrome: A focus on DNA methylation. European Journal of Neuroscience, 55, 2253–2296.
Xu, K., Zhang, X., Wang, Z., Hu, Y., & Sinha, R. (2018). Epigenome-wide association analysis revealed that SOCS3 methylation influences the effect of cumulative stress on obesity. Biological Psychology, 131, 63–71.
Zannas, A. S., Arloth, J., Carrillo-Roa, T., et al. (2015). Lifetime stress accelerates epigenetic aging in an urban, African American cohort: Relevance of glucocorticoid signaling. Genome Biology, 16, 266.
Acknowledgements
We would like to acknowledge the study participants, the many research staff who helped to collect the data, Asi Polly Gobin for data management, and the Yale Center for Genome Analysis for DNA methylation profiling.
Funding
This research was supported by National Institutes of Health grants MH101107 (ART), UL1-DE019586 (RS), PL1-DA24859 (RS), R01-AA013892 (RS), NIH R01DA047063 (KX), NIH T32MH019961 (ZMH), and NIH R25MH071584 (ZMH). Z.M.H. is also supported by the Yale Physician Scientist Development Award and CTSA (NIH UL1 TR001863). Dr. Tyrka’s time was additionally supported by P20GM139767 and R01HD086487 (ART). Dr. Daniels received support from MH101076 (ART) and HD101392 (ART). Dr. Kudinova’s time was supported by NIMH (K23MH122587) and NIGMS Bradley Hospital COBRE Center (P20GM139743). Dr. Brick received support from NIDA (R01DA054116).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest relevant to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Harvanek, Z.M., Kudinova, A.Y., Wong, S.A. et al. Childhood adversity, accelerated GrimAge, and associated health consequences. J Behav Med (2024). https://doi.org/10.1007/s10865-024-00496-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10865-024-00496-0