Abstract
A thin endocrown restoration was often applied in endodontically treated teeth with vertical bite height loss or inadequate clinical crown length. A model of mandibular molars made by endocrown restoration with 1 mm thickness and 2 mm depth of pulp chamber was constructed and imported into FEA ANSYS v18.0 software. The three CAD/CAM materials, feldspathic (Mark2), lithium disilicate (EMAX), and lava ultimate (LU), were assigned, and the five load indenters were loaded on the full occlusal (FO), occlusal center (OC), central fossa (CF), buccal groove (BG), and mesiobuccal cusp (MC) of restoration in the model. The MinPS and MaxPS of the thin endocrown were significantly higher than those of tooth tissue in five types of loads except for the LU endocrown loaded in the FO group. The smaller the contact surface of the load was, the higher MaxPS and MinPS were. MaxPS and MinPS of the MC were the highest, followed by the BG and CF in the restoration. In the stress distribution of tooth tissue, MaxPS in the LU endocrown accumulated at the external edge of enamel and was significantly higher than MaxPS in Mark2 and EMAX endocrown concentrated on the chamber wall of dentin under OC, CF and BG loads. Within the limitations of this FEA study, the LU endocrown transferred more stress to tooth tissue than Mark2 and EMAX, and the maximum principal stress on endocrown restoration and tooth tissue at the mesiobuccal cusp load was higher than that at the central fossa and buccal groove load.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The restoration of endodontically treated teeth with extensive damage remains a clinical challenge [1]. Despite the clinical success obtained by the use of intraradicular posts and full-coverage crowns, one disadvantage of this method is the extra removal of sound tissue needed for fitting the post-retained foundation restoration [2]. Endocrown, a monoblock restoration, assembles the intraradicular post, the core and the crown in one component and was first introduced as an alternative treatment modality in 1995 by Pissis [3]. Endocrown is a reliable alternative to post-retained restorations and has often been used to restore endodontically treated teeth with significant loss of coronal tooth structure [4, 5].
A conservative coronal restoration endocrown utilizes the pulp chamber and adhesive cementation to increase the macromechanical retention of the restoration and reduces the removal of sound tooth tissue [6, 7]. The preservation of more sound tooth tissue signifies a thinner coronal restoration. As with any other restoration, an overall reduction of at least 2 mm in height is required in tooth preparation for an endocrown [8]. Along with the evolution of high-strength and tough restorative materials, a lower occlusal reduction was recommended to adopt a minimally invasive approach in the posterior region [9]. Etchable glass ceramic and nanocomposite resin materials are often used in chairside CAD/CAM fabrication of restorations due to their esthetic appearance, excellent biocompatibility, and mechanical properties [10,11,12]. Among the available chairside CAD/CAM materials, lithium disilicate glass–ceramic and nanofilled composite resin stand out [13]. Resin nanoceramics (Lava Ultimate, LU) display high-impact fracture resistance, low modulus and high flexural strength, and lithium disilicate (IPS e.max CAD, Ivoclar Vivadent, Schaan, Liechtenstein) possess excellent biomechanical characteristics with a superior flexural strength beyond 400 MPa [11, 12]. Therefore, LU and EMAX are often studied as thinner occlusal veneers to restore the occlusal appearance of defective teeth [14, 15].
Restoration thickness determines the fracture resistance of full-coverage occlusal veneers [14, 16]. Endodontically treated teeth with vertical bite height loss or inadequate clinical crown length are not uncommon and are difficult to rehabilitate using a complete crown; thus, an endocrown may be an optional selection. Under such circumstances, the restoration thickness of the endocrown did not reach at least a 2-mm requirement. Furthermore, from minimally invasive dentistry, a thinner occlusal thickness of endocrown was recommended due to the lower amount of sound tooth tissue removed. Therefore, we need to investigate the stress distribution of the endocrown with a thinner coronal to expect clinical application. In this study, finite element analysis was used to evaluate the stress distribution where the three types of CAD/CAM dental material of endocrown restoration had 1 mm of coronal thickness by the five types of load.
2 Methods and materials
2.1 The finite element analysis model
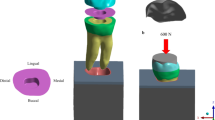
A 3D geometric model of an intact mandibular molar was obtained from microcomputed tomography (uCT50, Switzerland) with a voxel dimension of 9 μm and was reconstructed using a CAD software program and a reverse engineering program (Mimics Medical 20.0; Materialize NV and Geomagic Studio 12.0; Geomagic Inc). The constructed model simulates the endocrown restoration of a mandibular molar after root canal treatment. The external and internal contours of the tooth, alveolar bone (cortical and spongious bone), 0.3-mm-thick periodontal ligament, and dentin and pulp contours were outlined and assembled. A 3-D numerical model of the intact mandibular molar was constructed by assembling all the individual elements. In the model of the tooth with an endocrown restoration, the pulp in the root canal was replaced by gutta-percha, and the traditional endodontic cavity was filled with smart dentin resin (SDR) and the endocrown restoration. The endocrown restoration was designed with 1 mm of occlusal clearance and 2 mm of depth in the pulp chamber, and the axial walls presented an internal taper of 6°. The mechanical properties of the materials, tooth tissue, and bone (elastic modulus and Poisson ratio) were determined from published values, and three CAD/CAM restorative materials were simulated: feldspathic (Mark2), lithium disilicate (EMAX), and lava ultimate (LU) (Table 1). The luting cement between the endocrown restoration and the tooth was limited to 100-μm thickness. The Mark2 and EMAX restorations were adhered using Multilink N, and the LU restoration was adhered with RelyX™ Ultimate. The model was imported into finite element analysis software (FEA, ANSYS, v18.0; Swanson Analysis Inc.). All structures were assumed to be linearly elastic, isotropic, and homogeneously distributed. Nodal displacements on the surfaces of the model were constrained in all directions.
2.2 Stress analysis of the different loads
In the model of the endocrown restoration, physiological masticatory loads were simulated as an occlusal static load of 600 N on the Z axis, similar to the study by Ausiello et al. [17]. The methodology considered the contact between a food bolus (apple pulp) and the restoration surface during the closing phase of the chewing cycle. A cylinder indenter was made by subtracting the occlusal morphology of the endocrown restoration in ANSYS v18.0 software and almost covered the full occlusal (FO) surface of the endocrown restoration except for the lingual edge (108.57 mm2) to simulate central occlusion (Fig. 1A). A large sphere indenter (diameter: 8.57 mm) was generated by revolving in ANSYS v18.0 software and contacting the occlusal center (OC) area of the endocrown restoration (11.425 mm2) (Fig. 2A). Furthermore, three small sphere indenters (diameter: 3 mm) were assigned with Young’s modulus and Poisson’s ratio of bone and loaded at the central fossa (CF) (1.44 mm2), buccal groove (BG) (1.43 mm2) and mesiobuccal cusp (MC) (1.43 mm2) of the endocrown restoration and simulated suddenly biting bone fragments (Figs. 3A, 4A, 5A). The results in the restoration and tooth tissue were obtained using maximum principal stress (MaxPS) and minimum principal stress (MinPS) for quantitative analysis. Stress values differing by less than 5% were considered to be similar.
Stress distribution of a mandibular molar covered by an endocrown restoration with a 1-mm thickness and a 2-mm depth of pulp chamber. A cylinder indenter loaded the full occlusal (FO) surface of the restoration except for the lingual edge (contact area: 108.57 mm2) to simulate central occlusion (A). The restorations were made with lava ultimate (LU), feldspathic (Mark2) and lithium disilicate (EMAX). The MaxPS and MinPS values of the restoration (B) and tooth tissue (C) are displayed
Stress distribution of the mandibular molar covered by an endocrown restoration with a 1-mm thickness and a 2-mm depth of pulp chamber. A large spherical indenter (diameter: 8.57 mm) was generated by revolving in ANSYS v18.0 software and contacting the occlusal center (OC) area of the endocrown restoration (11.425 mm2) (A). The restorations were made with lava ultimate (LU), feldspathic (Mark2) and lithium disilicate (EMAX). The MaxPS and MinPS values of the restoration (B) and tooth tissue (C) are displayed
Stress distribution of the mandibular molar covered by an endocrown restoration with 1-mm thickness and a 2-mm depth of pulp chamber. A small spherical indenter (diameter: 3 mm) was assigned with Young’s modulus and Poisson’s ratio of bone and loaded at the central fossa (CF) (1.44 mm2) of the endocrown restorations to simulate suddenly biting a bone fragment (A). The restorations were made with lava ultimate (LU), feldspathic (Mark2) and lithium disilicate (EMAX). The MaxPS and MinPS values of the restoration (B) and tooth tissue (C) are displayed
Stress distribution of the mandibular molar covered by an endocrown restoration with a 1-mm thickness and a 2-mm depth of pulp chamber. A small spherical indenter (diameter: 3 mm) was assigned with Young’s modulus and Poisson’s ratio of bone and loaded at the buccal groove (BG) (1.43 mm2) of the endocrown restorations to simulate suddenly biting a bone fragment (A). The restorations were made with lava ultimate (LU), feldspathic (Mark2) and lithium disilicate (EMAX). The MaxPS and MinPS values of the restoration (B) and tooth tissue (C) are displayed
Stress distribution of the mandibular molar covered by an endocrown restoration with a 1-mm thickness and a 2-mm depth of pulp chamber. A small spherical indenter (diameter: 3 mm) was assigned with Young’s modulus and Poisson’s ratio of bone and loaded at the mesiobuccal cusp (MC) (1.43 mm2) of the endocrown restorations to simulate suddenly biting a bone fragment (A). The restorations were made with lava ultimate (LU), feldspathic (Mark2) and lithium disilicate (EMAX). The MaxPS and MinPS values of the restoration (B) and tooth tissue (C) are displayed
3 Results
3.1 Stress distribution of the endocrown restoration
In the stress analysis of the model, MaxPS denoted tensile stress (positive value), and MinPS signified compressive stress (negative value). The different loads showed different stress distributions on the endocrown restoration. The smaller the contact surface of the load, the higher MaxPS and MinPS in the five different loads. The MaxPS and MinPS units were megapascals (MPa) in the following results. In the FO load, the MaxPS (3.21) of the LU restoration was significantly lower than the MaxPS values of the Mark2 (19.5) and EMAX restorations (23.7), and the MinPS (11.9) of the LU restoration was higher than the MinPS values of the Mark2 (3.26) and EMAX (3.64) restorations (Fig. 1B). In the OC load by a large sphere, the MaxPS values of the LU (58.9), Mark2 (61.4) and EMAX (59.9) restorations were not significantly different, and the MinPS (51.2) of the LU restoration was slightly higher than the MinPS values of the Mark2 (40.3) and EMAX (41.2) restorations (Fig. 2B). In the three small sphere loads, the MaxPS and MinPS of the MC were the highest, followed by the BG and CF in the LU, Mark2, and EMAX restorations. The intaglio surface of the LU restoration showed compressive stress, whereas the intaglio surfaces of the Mark2 and EMAX restorations showed tensile stress at the site of a small sphere load (Figs. 3B, 4B, and 5B).
3.2 Stress distribution of the tooth tissue
The FEA of tooth tissue comprised the stress analysis of enamel and dentin. Overall, the MinPS and MaxPS of the thin endocrown were significantly higher than those of tooth tissue in the five types of loads except for the LU endocrown loaded in the FO group (Table 2). In the stress distribution of tooth tissue by the FO load, the MaxPS (25.8) of tooth tissue in the LU restoration was significantly higher than those in the Mark2 (16.0) and EMAX (15.5) restorations, and the MinPS values by the three materials were not obviously different (Fig. 1C). In OC load, the stress distribution of tooth tissue showed that the MaxPS (21.7) in the LU endocrown accumulated in the external edge of enamel and was significantly higher than the MaxPS values in Mark2 (8.39) and EMAX (9.86) endocrowns, which were concentrated on the chamber wall of dentin (Fig. 2C). Similarly, stress analysis of the tooth tissue in the CF load revealed that the MaxPS (18.0) was concentrated at the external edge of enamel in the LU restoration and was significantly higher than the MaxPS values on the chamber wall of dentin in the Mark2 (12.9) and EMAX (14.3) restorations (Fig. 3C). Stress distribution by the BG load showed that the MaxPS (33.6) at the external edge of the enamel in the LU restoration was almost double those of the MaxPS values on the chamber wall of dentin in the Mark2 (16.5) and EMAX (18.7) restorations (Fig. 4C). The MaxPS values of tooth tissue in all three material restorations were at the internal edge of the enamel when the MC was loaded by the small sphere, and the MaxPS (56.0) in the LU restoration was higher than the MaxPS values in the Mark2 (35.4) and EMAX (32.9) restorations (Fig. 5C). In sum, tooth tissue received higher stress distribution in the LU endocrown restoration than in the Mark2 and EMAX restorations. The enamel supported more tensile stress, and the dentin mostly undertook the compressive stress.
4 Discussion
Endocrown has been considered a reliable alternative in the restoration of teeth with bad damage after root canal treatment [4, 13]. From minimally invasive dentistry, conservative treatment in tooth preparation for an endocrown is inevitable to decrease the thickness of the restoration. Furthermore, when the height of the crown is limited, whether a thin endocrown restoration meets the clinical requirement still needs further investigation. Finite element analysis has often been used in dental biomechanical research to estimate the stress distribution in a dental field and to predict the practicability of the restoration. FEA is a reproducible and noninvasive technique that simulates the oral environment and obtains stress values at any point [18,19,20,21]. In this study, we constructed a model of endocrown restoration with a pulp chamber thickness of 1 mm and a depth of 2 mm by FEA and evaluated the stress distribution of the restoration made by LU, Mark2, and EMAX materials. Thin restorations were considered to accumulate more stress in the structures than thick restorations and may induce the formation of cracks and increase the failure risk in critical thickness restorations [14, 22, 23]. Therefore, 1 mm thickness of endocrown was selected as a critical thickness restoration to analyze the stress distribution. In addition, a 2-mm depth of the pulp chamber was considered optimal in endocrown restoration [24, 25].
In this study, three endocrown restorations under five different loads were analyzed. The indenter contacting the almost FO surface and the OC of the restoration, respectively, denotes a situation of central occlusive and biting food. The three sites of restoration (CF, BG, and MC) were loaded by a small spherical indenter intended to simulate bone fragments. In stress analysis, MaxPS is a tensile stress measurement used to judge the material failure that is assumed to be due to brittleness and implies a fracture tendency of the components [26]. In the larger contact area of load (FO), the LU restoration showed a lower MaxPS than the Mark2 and EMAX restorations, whereas the smaller contact area of load (CF, BG, and MC) revealed a higher stress concentration in the LU restoration than in the Mark2 and EMAX restorations. This phenomenon indicated that the LU material dispersed the stress at the evenly occluded contact, but high tensile stress occurred in the highlighted site of the LU restoration when encountering an unexpected bite. Furthermore, regardless of whether there was a large or small contact area of restoration in the test loads, the tooth tissue sustained significantly higher tensile stress with the LU restoration than with the Mark2 and EMAX restorations, and the enamel received more tensile stress than the dentin. The elastic modulus of the LU restoration was significantly lower than those of the Mark2 and EMAX restorations [11], and the low elastic modulus of the materials transferred more stress to dental tissue, which is consistent with the Yamanel et al.’s study [27]. Thus, the high elastic modulus of the materials may absorb more stress and reduce the stress distribution of tooth tissue. The low elastic modulus of the LU restoration mainly showed compressive stress in the whole restoration in the five test loads and transferred stress to the tooth tissue. The high elastic modulus of the Mark2 and EMAX restorations displayed little tensile stress on the intaglio surface of the restoration, and less stress was passed to tooth tissue. The data suggested that a thin Mark2 or EMAX restoration may tend to develop radial cracks and protect the tooth tissue.
The effect of material thickness on restoration fractures has been studied by some authors [14, 16, 22, 28, 29]. Thinner restorations accumulate more stress in the structures than thicker restorations and may cause the formation of cracks and increase the fracture risk in critical thickness. In our study, almost the same contact area of the indenter was loaded on the CF, BG, and MC of the restoration. We found that the tensile stress levels of the intaglio surfaces of the Mark2 and EMAX restorations in the BG and MC groups were significantly higher than those in the CF group. The thickness of the CF was obviously greater than those of the BG and the MC in endocrown restorations due to the 2-mm depth of the intracoronal extension. Furthermore, despite the lack of a significant difference between the tensile stress of the intaglio surfaces of the Mark2 or EMAX restorations in the BG and MC groups, the MaxPS was concentrated on the chamber wall of the tooth tissue in the BG group, and MaxPS occurred on the enamel in the MC group. Meanwhile, the Mark2 and EMAX restorations in the CF group transferred the MaxPS to the chamber wall of the tooth tissue. The results indicated that the different sites of load on the occlusal surface of the endocrown may result in the different stress distributions of tooth tissue. A lower MaxPS tended to concentrate on the dentin, and a higher MaxPS appeared on the enamel. The thin endocrown restoration sustained primary stress, whereas the tooth tissue received relatively low stress when encountering an unexpected bite, such as a bone fragment or small stone.
In the full-coverage occlusal of load, tooth tissue sustained the higher tensile stress than the LU endocrown restoration, and inverse results in Mark2 and EMAX restoration, which indicated that the low elastic modulus of materials may take little tensile stress, and the primary stress was transferred to the tooth tissue in the central occlusion. A high elastic modulus material will sustain a larger amount of stress and reduce the tensile stress on the tooth tissue. When 600 N was vertically loaded on the OC area of the thin endocrown, the MaxPS on the tooth tissue became lower than that in the LU restoration, and the MaxPS was transferred on the chamber wall in the Mark2 and EMAX restorations. This outcome might be related to the thickness of the restoration. The central thickness of the endocrown was three times that of the surroundings. Therefore, during rehabilitation of the thin endocrown, intracoronal extension is helpful to disperse the stress of the tooth tissue. In our FEA study, only a 2-mm depth was employed. We still need to explore the effects of other depths of extension on the stress distribution of tooth tissue with thin endocrown.
5 Conclusion
Within the limitations of this FEA study of thin endocrown restoration of an endodontically treated molar, the following conclusions were drawn:
-
1.
The LU endocrown transferred more stress to tooth tissue than the Mark2 and EMAX restorations, irrespective of the loads.
-
2.
The smaller the contact surface when the restoration was loaded, the higher MaxPS and MinPS were in the three material restorations.
-
3.
At the same load force and area, the MaxPS on the endocrown restoration and tooth tissue at the MC load was higher than that at the CF and the BG, regardless of the restorative materials.
References
Zhu Z, Dong XY, He S, Pan X, Tang L. Effect of post placement on the restoration of endodontically treated teeth: a systematic review. Int J Prosthodont. 2015;28:475–83. https://doi.org/10.11607/ijp.4120
Lazari PC, Oliveira RC, Anchieta RB, Almeida EO, Freitas Junior AC, Kina S, et al. Stress distribution on dentin-cement-post interface varying root canal and glass fiber post diameters. A three-dimensional finite element analysis based on micro-CT data. J Appl Oral Sci. 2013;21:511–7. https://doi.org/10.1590/1679-775720130203
Pissis P. Fabrication of a metal-free ceramic restoration utilizing the monobloc technique. Pract Periodontics Aesthet Dent. 1995;7:83–94.
Al-Dabbagh RA. Survival and success of endocrowns: a systematic review and meta-analysis. J Prosthet Dent. 2021;125:415 e411–15.e9. https://doi.org/10.1016/j.prosdent.2020.01.011
Sedrez-Porto JA, Rosa WL, da Silva AF, Munchow EA, Pereira-Cenci T. Endocrown restorations: a systematic review and meta-analysis. J Dent. 2016;52:8–14. https://doi.org/10.1016/j.jdent.2016.07.005
El-Damanhoury HM, Haj-Ali RN, Platt JA. Fracture resistance and microleakage of endocrowns utilizing three CAD-CAM blocks. Oper Dent. 2015;40:201–10. https://doi.org/10.2341/13-143-L
Sofan E, Sofan A, Palaia G, Tenore G, Romeo U, Migliau G. Classification review of dental adhesive systems: from the IV generation to the universal type. Ann Stomatol. 2017;8:1–17. https://doi.org/10.11138/ads/2017.8.1.001
Fages M, Bennasar B. The endocrown: a different type of all-ceramic reconstruction for molars. J Can Dent Assoc. 2013;79:d140.
Magne P, Schlichting LH, Maia HP, Baratieri LN. In vitro fatigue resistance of CAD/CAM composite resin and ceramic posterior occlusal veneers. J Prosthet Dent. 2010;104:149–57. https://doi.org/10.1016/S0022-3913(10)60111-4
Grzebieluch W, Mikulewicz M, Kaczmarek U. Resin composite materials for chairside CAD/CAM restorations: a comparison of selected mechanical properties. J Healthc Eng. 2021;2021:8828954 https://doi.org/10.1155/2021/8828954
Belli R, Wendler M, de Ligny D, Cicconi MR, Petschelt A, Peterlik H, et al. Chairside CAD/CAM materials. Part 1: measurement of elastic constants and microstructural characterization. Dent Mater. 2017;33:84–98. https://doi.org/10.1016/j.dental.2016.10.009
Wendler M, Belli R, Petschelt A, Mevec D, Harrer W, Lube T, et al. Chairside CAD/CAM materials. Part 2: Flexural strength testing. Dent Mater. 2017;33:99–109. https://doi.org/10.1016/j.dental.2016.10.008
Govare N, Contrepois M. Endocrowns: a systematic review. J Prosthet Dent. 2020;123:411–418.e9. https://doi.org/10.1016/j.prosdent.2019.04.009
Albelasy E, Hamama HH, Tsoi JKH, Mahmoud SH. Influence of material type, thickness and storage on fracture resistance of CAD/CAM occlusal veneers. J Mech Behav Biomed Mater. 2021;119:104485 https://doi.org/10.1016/j.jmbbm.2021.104485
Heck K, Paterno H, Lederer A, Litzenburger F, Hickel R, Kunzelmann KH. Fatigue resistance of ultrathin CAD/CAM ceramic and nanoceramic composite occlusal veneers. Dent Mater. 2019;35:1370–7. https://doi.org/10.1016/j.dental.2019.07.006
Sasse M, Krummel A, Klosa K, Kern M. Influence of restoration thickness and dental bonding surface on the fracture resistance of full-coverage occlusal veneers made from lithium disilicate ceramic. Dent Mater. 2015;31:907–15. https://doi.org/10.1016/j.dental.2015.04.017
Ausiello P, Ciaramella S, Martorelli M, Lanzotti A, Gloria A, Watts DC. CAD-FE modeling and analysis of class II restorations incorporating resin-composite, glass ionomer and glass ceramic materials. Dent Mater. 2017;33:1456–65. https://doi.org/10.1016/j.dental.2017.10.010
Dal Piva AMO, Tribst JPM, Borges ALS, Souza R, Bottino MA. CAD-FEA modeling and analysis of different full crown monolithic restorations. Dent Mater. 2018;34:1342–50. https://doi.org/10.1016/j.dental.2018.06.024
Zhu J, Rong Q, Wang X, Gao X. Influence of remaining tooth structure and restorative material type on stress distribution in endodontically treated maxillary premolars: a finite element analysis. J Prosthet Dent. 2017;117:646–55. https://doi.org/10.1016/j.prosdent.2016.08.023
Lin CL, Chang YH, Chang CY, Pai CA, Huang SF. Finite element and Weibull analyses to estimate failure risks in the ceramic endocrown and classical crown for endodontically treated maxillary premolar. Eur J Oral Sci. 2010;118:87–93. https://doi.org/10.1111/j.1600-0722.2009.00704.x
Ulusoy N, Gulec Alagoz L. Evaluation of two CAD/CAM materials for Nayyar core and post-retained restorations: three dimensional stress analysis. J Mech Behav Biomed Mater. 2021;117:104381 https://doi.org/10.1016/j.jmbbm.2021.104381
Abu-Izze FO, Ramos GF, Borges ALS, Anami LC, Bottino MA. Fatigue behavior of ultrafine tabletop ceramic restorations. Dent Mater. 2018;34:1401–9. https://doi.org/10.1016/j.dental.2018.06.017
de Abreu RA, Pereira MD, Furtado F, Prado GP, Mestriner W Jr., Ferreira LM. Masticatory efficiency and bite force in individuals with normal occlusion. Arch Oral Biol. 2014;59:1065–74. https://doi.org/10.1016/j.archoralbio.2014.05.005
Hayes A, Duvall N, Wajdowicz M, Roberts H. Effect of endocrown pulp chamber extension depth on molar fracture resistance. Oper Dent. 2017;42:327–34. https://doi.org/10.2341/16-097-L
Rocca GT, Daher R, Saratti CM, Sedlacek R, Suchy T, Feilzer AJ, et al. Restoration of severely damaged endodontically treated premolars: the influence of the endo-core length on marginal integrity and fatigue resistance of lithium disilicate CAD-CAM ceramic endocrowns. J Dent. 2018;68:41–50. https://doi.org/10.1016/j.jdent.2017.10.011
Asmussen E, Peutzfeldt A, Sahafi A. Finite element analysis of stresses in endodontically treated, dowel-restored teeth. J Prosthet Dent. 2005;94:321–9. https://doi.org/10.1016/j.prosdent.2005.07.003
Yamanel K, Caglar A, Gulsahi K, Ozden UA. Effects of different ceramic and composite materials on stress distribution in inlay and onlay cavities: 3-D finite element analysis. Dent Mater J. 2009;28:661–70. https://doi.org/10.4012/dmj.28.661
Ruggiero MM, Soares Gomes R, Pedroso Bergamo ET, Freitas MIM, Bonfante EA, Del Bel Cury AA. Resin-matrix ceramics for occlusal veneers: effect of thickness on reliability and stress distribution. Dent Mater. 2021;37:e131–9. https://doi.org/10.1016/j.dental.2020.11.002.
Monteiro JB, Riquieri H, Prochnow C, Guilardi LF, Pereira GKR, Borges ALS, et al. Fatigue failure load of two resin-bonded zirconia-reinforced lithium silicate glass-ceramics: effect of ceramic thickness. Dent Mater. 2018;34:891–900. https://doi.org/10.1016/j.dental.2018.03.004
Omran TA, Garoushi S, Lassila L, Shinya A, Vallittu PK. Bonding interface affects the load-bearing capacity of bilayered composites. Dent Mater J. 2019;38:1002–11. https://doi.org/10.4012/dmj.2018-304
Gulec L, Ulusoy N. Effect of endocrown restorations with different CAD/CAM materials: 3D finite element and weibull analyses. Biomed Res Int. 2017;2017:5638683 https://doi.org/10.1155/2017/5638683
Zheng Z, He Y, Ruan W, Ling Z, Zheng C, Gai Y, et al. Biomechanical behavior of endocrown restorations with different CAD-CAM materials: a 3D finite element and in vitro analysis. J Prosthet Dent. 2020;125:890–9. https://doi.org/10.1016/j.prosdent.2020.03.009
Braganca GF, Mazao JD, Versluis A, Soares CJ. Effect of luting materials, presence of tooth preparation, and functional loading on stress distribution on ceramic laminate veneers: a finite element analysis. J Prosthet Dent. 2021;125:778–87. https://doi.org/10.1016/j.prosdent.2020.02.005
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. 81870750) and the Guangdong Financial Fund for High-Caliber Hospital Construction (174-2018-XMZC-0001-03-0125/A-01). We are grateful to the Elsevier Language Editing Service for professionally editing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Meng, Q., Zhang, Y., Chi, D. et al. Resistance fracture of minimally prepared endocrowns made by three types of restorative materials: a 3D finite element analysis. J Mater Sci: Mater Med 32, 137 (2021). https://doi.org/10.1007/s10856-021-06610-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-021-06610-x