Abstract
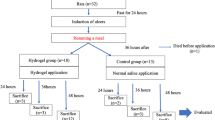
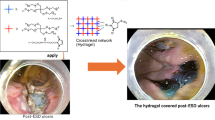
The gastrointestinal (GI) endoscopy has become a standard diagnostic tool for GI ulcers and cancer. In this study we studied endoscopic application of epidermal growth factor-containing chitosan hydrogel (EGF-CS gel) for treatment of GI ulcer. We hypothesized that directional ulcer-coating using EGF-CS gel via endoscope would precipitate ulcer-healing. EGF-CS gel was directly introduced to the ulcer-region after ulceration in acetic acid-induced gastric ulcer (AAU) and mucosal resection-induced gastric ulcer (MRU) rabbit and pig models. The ulcer dimensions and mucosal thicknesses were estimated and compared with those in the control group. Healing efficacy was more closely evaluated by microscopic observation of the ulcer after histological assays. In the AAU model, the normalized ulcer size of the gel-treated group was 2.3 times smaller than that in the non-treated control group on day 3 after ulceration (P < 0.01). In the MRU model, the normalized ulcer size of the gel-treated group was 5.4 times smaller compared to that in the non-treated control group on day 1 after ulceration (P < 0.05). Histological analysis supported the ability of EGF-CS gel to heal ulcers. The present study suggests that EGF-CS gel is a promising candidate for treating gastric bleeding and ulcers.








Similar content being viewed by others
References
Shaheen NJ, Hansen RA, Morgan DR, Gangarosa LM, Ringel Y, Thiny MT, et al. The burden of gastrointestinal and liver diseases. Am J Gastroenterol. 2006;101(9):2128–38.
Chai J, Baatar D, Tarnawski A. Serum response factor promotes re-epithelialization and muscular structure restoration during gastric ulcer healing. Gastroenterology. 2004;126(7):1809–18.
Lockrey G, Lim L. Peptic ulcer disease in older people. J Pharm Pract Res. 2011;41(1):58–61.
Boers M, Tangelder MJ, van Ingen H, Fort JG, Goldstein JL. The rate of NSAID-induced endoscopic ulcers increases linearly but not exponentially with age: a pooled analysis of 12 randomised trials. Ann Rheum Dis. 2007;66(3):417–8.
Holster IL, Kuipers EJ. Update on the endoscopic management of peptic ulcer bleeding. Curr Gastroenterol Rep. 2011;13(6):525–31.
Gotoda T. Endoscopic resection of early gastric cancer. Gastric Cancer. 2007;10(1):1–11.
Hayashi T, Matsuyama T, Hanada K, Nakanishi K, Uenoyama M, Fujita M, et al. Usefulness of photocrosslinkable chitosan for endoscopic cancer treatment in alimentary tract. J Biomed Mater Res B Appl Biomater. 2004;71(2):367–72.
Kumano I, Ishihara M, Nakamura S, Kishimoto S, Fujita M, Hattori H, et al. Endoscopic submucosal dissection for pig esophagus by using photocrosslinkable chitosan hydrogel as submucosal fluid cushion. Gastrointest Endosc. 2012;75(4):841–8.
Hattori H, Amano Y, Nogami Y, Takase B, Ishihara M. Hemostasis for severe hemorrhage with photocrosslinkable chitosan hydrogel and calcium alginate. Ann Biomed Eng. 2010;38(12):3724–32.
Ishizuka T, Ishihara M, Aiko S, Nogami Y, Nakamura S, Kanatani Y, et al. Experimental evaluation of photocrosslinkable chitosan hydrogel as injection solution for endoscopic resection. Endoscopy. 2009;41(1):25–8.
Saito Y, Uraoka T, Matsuda T, Emura F, Ikehara H, Mashimo Y, et al. Endoscopic treatment of large superficial colorectal tumors: a case series of 200 endoscopic submucosal dissections (with video). Gastrointest Endosc. 2007;66(5):966–73.
Tsuda Y, Ishihara M, Amako M, Arino H, Hattori H, Kanatani Y, et al. Photocrosslinkable chitosan hydrogel can prevent bone formation in both rat skull and fibula bone defects. Artif Organs. 2009;33(1):74–7.
Tsuda Y, Hattori H, Tanaka Y, Ishihara M, Kishimoto S, Amako M, et al. Ultraviolet light-irradiated photocrosslinkable chitosan hydrogel to prevent bone formation in both rat skull and fibula bone defects. J Tissue Eng Regen Med. 2013;7(9):720–8.
Ueno H, Yamada H, Tanaka I, Kaba N, Matsuura M, Okumura M, et al. Accelerating effects of chitosan for healing at early phase of experimental open wound in dogs. Biomaterials. 1999;20(15):1407–14.
Peluso G, Petillo O, Ranieri M, Santin M, Ambrosio L, Calabro D, et al. Chitosan-mediated stimulation of macrophage function. Biomaterials. 1994;15(15):1215–20.
Muzzarelli R, Tarsi R, Filippini O, Giovanetti E, Biagini G, Varaldo PE. Antimicrobial properties of N-carboxybutyl chitosan. Antimicrob Agents Chemother. 1990;34(10):2019–23.
Rask F, Dallabrida SM, Ismail NS, Amoozgar Z, Yeo Y, Rupnick MA, et al. Photocrosslinkable chitosan modified with angiopoietin-1 peptide, QHREDGS, promotes survival of neonatal rat heart cells. J Biomed Mater Res A. 2010;95(1):105–17.
Ono K, Saito Y, Yura H, Ishikawa K, Kurita A, Akaike T, et al. Photocrosslinkable chitosan as a biological adhesive. J Biomed Mater Res. 2000;49(2):289–95.
Ono K, Ishihara M, Ozeki Y, Deguchi H, Sato M, Saito Y, et al. Experimental evaluation of photocrosslinkable chitosan as a biologic adhesive with surgical applications. Surgery. 2001;130(5):844–50.
Ong SY, Wu J, Moochhala SM, Tan MH, Lu J. Development of a chitosan-based wound dressing with improved hemostatic and antimicrobial properties. Biomaterials. 2008;29(32):4323–32.
Carpenter G, Cohen S. Epidermal growth factor. J Biol Chem. 1990;265(14):7709–12.
Brown GL, Curtsinger LJ, White M, Mitchell RO, Pietsch J, Nordquist R, et al. Acceleration of tensile strength of incisions treated with EGF and TGF-beta. Ann Surg. 1988;208(6):788–94.
Celebi N, Erden N, Gonul B, Koz M. Effects of epidermal growth factor dosage forms on dermal wound strength in mice. J Pharm Pharmacol. 1994;46(5):386–7.
Herbst RS. Review of epidermal growth factor receptor biology. Int J Radiat Oncol Biol Phys. 2004;59(2 Suppl):21–6.
Celebi N, Turkyilmaz A, Gonul B, Ozogul C. Effects of epidermal growth factor microemulsion formulation on the healing of stress-induced gastric ulcers in rats. J Control Release. 2002;83(2):197–210.
Alemdaroglu C, Degim Z, Celebi N, Zor F, Ozturk S, Erdogan D. An investigation on burn wound healing in rats with chitosan gel formulation containing epidermal growth factor. Burns. 2006;32(3):319–27.
Takahashi M, Ota S, Shimada T, Hamada E, Kawabe T, Okudaira T, et al. Hepatocyte growth factor is the most potent endogenous stimulant of rabbit gastric epithelial cell proliferation and migration in primary culture. J Clin Invest. 1995;95(5):1994–2003.
de Carpani Kaski M, Rentsch R, Levi S, Hodgson HJ. Corticosteroids reduce regenerative repair of epithelium in experimental gastric ulcers. Gut. 1995;37(5):613–6.
Kitajima T, Okuhira M, Tani K, Nakano T, Hiramatsu A, Mizuno T, et al. Cell proliferation kinetics in acetic acid-induced gastric ulcer evaluated by immunohistochemical staining of proliferating cell nuclear antigen. J Clin Gastroenterol. 1993;17(Suppl 1):S116–20.
Sairam K, Priyambada S, Aryya NC, Goel RK. Gastroduodenal ulcer protective activity of Asparagus racemosus: an experimental, biochemical and histological study. J Ethnopharmacol. 2003;86(1):1–10.
Relan NK, Fligiel SE, Dutta S, Tureaud J, Chauhan DP, Majumdar AP. Induction of EGF-receptor tyrosine kinase during early reparative phase of gastric mucosa and effects of aging. Lab Invest. 1995;73(5):717–26.
Wong WM, Playford RJ, Wright NA. Peptide gene expression in gastrointestinal mucosal ulceration: ordered sequence or redundancy? Gut. 2000;46(2):286–92.
Acknowledgments
This study was supported by a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A111060). The large animal study was supported by the National Center of Efficacy Evaluation for the Development of Health Products Targeting Digestive Disorders (NCEED).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jin Hee Maeng and Byoung Wook Bang contributed equally to this study.
Rights and permissions
About this article
Cite this article
Maeng, J.H., Bang, B.W., Lee, E. et al. Endoscopic application of EGF-chitosan hydrogel for precipitated healing of GI peptic ulcers and mucosectomy-induced ulcers. J Mater Sci: Mater Med 25, 573–582 (2014). https://doi.org/10.1007/s10856-013-5088-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-013-5088-x