Abstract
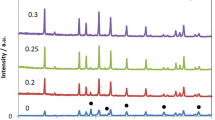
The series Li7−4xGexLa3Zr2O12 has been synthesized using conventional solid-state reaction method by substituting Germanium (Ge) at the Li site with the varying content of Ge (x) from 0.05 to 0.20. The conducting cubic phase is confirmed using XRD analysis. The surface morphology and elemental distribution have been studied with the help of SEM characterization. The densities of the samples were calculated. For the confirmation of functional groups present, IR spectroscopy has been studied. The modulus and ac conductivity studies have also been examined. A complex impedance study has been carried out in the frequency range 20 Hz to 20 MHz .The highest ionic conductivity has been observed for 0.10 Ge. The minimum activation energy of 0.56 eV is associated with the highest conductivity value of 7.23 × 10−6 S/cm at room temperature. The increment in ionic conductivity by one order at room temperature makes 0.10 Ge containing ceramic sample a promising candidate as a solid electrolyte.










Similar content being viewed by others
Data availability
Manuscript has no associated data.
References
E.J. Cussen, J. Mater. Chem. 20, 5167 (2010)
C. Sun, J. Liu, Y. Gong, D.P. Wilkinson, J. Zhang, Nano Energy. 33, 363 (2017)
J.M. Tarascon, M. Armand, Nature. 414, 359 (2001)
V. Thangadurai, S. Narayanan, D. Pinzaru, Chem. Soc. Rev. 43, 4714 (2014)
V. Thangadurai, S. Adams, W. Weppner, Chem. Mater. 12, 2998 (2004)
L. Van Wüllen, T. Echelmeyer, H.W. Meyer, D. Wilmer, Phys. Chem. Chem. Phys. 9, 3298 (2007)
R.H. Shin, S.I. Son, Y.S. Han, Y. Do Kim, H.T. Kim, S.S. Ryu, W. Pan, Solid State Ionics. 301, 10 (2017)
W. Zhang, J. Nie, F. Li, Z.L. Wang, C. Sun, Nano Energy. 45, 413 (2018)
Y. Kato, S. Hori, T. Saito, K. Suzuki, M. Hirayama, A. Mitsui, M. Yonemura, H. Iba, R. Kanno, Nat. Energy. 1, 1 (2016)
W. Greatbatch, J.H. Lee, W. Mathias, M. Eldridge, J.R. Moser, A.A. Schneider, IEEE Trans. Biomed. Eng. BME. –18, 317 (1971)
B.B. Owens, J. Power Sources. 90, 2 (2000)
A.V. Deshpande, V.K. Deshpande, Solid State Ionics. 154–155, 433 (2002)
J.T.S. Irvine, A.R. West, J. Solid State Chem. 69, 126 (1987)
E.I. Burmakin, B.D. Antonov, G.S. Shekhtman, Inorg. Mater. 46, 540 (2010)
J. Wang, C.W. Sun, Y.D. Gong, H.R. Zhang, J.A. Alonso, M.T. Fernández-Diaz, Z.L. Wang, J.B. Goodenough, Chin. Phys. B 27, 0 (2018)
M. Aote, A.V. Deshpande, Ceram. Int. 49, 40011 (2023)
R. Murugan, V. Thangadurai, W. Weppner, Angew. Chem. Int. Ed. 46, 7778 (2007)
Y.T. Chen, A. Jena, W.K. Pang, V.K. Peterson, H.S. Sheu, H. Chang, R.S. Liu, J. Phys. Chem. C 121, 15565 (2017)
Y. Tian, Y. Zhou, W. Wang, Y. Zhou, Ceram. Int. 48, 963 (2022)
D. Rettenwander, C.A. Geiger, G. Amthauer, ChemInform 44, no (2013) Inorg. Chem. 2014, 53(12) 6264–6269 https://doi.org/10.1021/ic500803h
S. Aktaş, O.M. Özkendir, Y.R. Eker, Ş. Ateş, Ü. Atav, G. Çelik, W. Klysubun, J. Alloys Compd. 792, 279 (2019)
Y. Zhang, J. Deng, D. Hu, F. Chen, Q. Shen, L. Zhang, S. Dong, Electrochim. Acta. 296, 823 (2019)
D.K. Schwanz, A. Villa, M. Balasubramanian, B. Helfrecht, E.E. Marinero, AIP Adv. (2020). https://doi.org/10.1063/1.5141764
M. Aote, A.V. Deshpande, J. Phys. Chem. Solids. 190, 111980 (2024)
S. Hu, Y.F. Li, R. Yang, Z. Yang, L. Wang, Ceram. Int. 44, 6614 (2018)
M. Huang, W. Xu, Y. Shen, Y.H. Lin, C.W. Nan, Electrochim. Acta. 115, 581 (2014)
H. Geng, K. Chen, D. Yi, A. Mei, M. Huang, Y. Lin, C. Nan, Xiyou Jinshu Cailiao Yu Gongcheng/Rare. Met. Mater. Eng. 45, 612 (2016)
R.H. Brugge, J.A. Kilner, A. Aguadero, Solid State Ionics. 337, 154 (2019)
X. Zhan, S. Lai, M.P. Gobet, S.G. Greenbaum, M. Shirpour, Phys. Chem. Chem. Phys. 20, 1447 (2018)
P. Scherrer, Nachr. Ges. Wiss. Göttingen. 2698 (1918)
J.I. Langford, A.J.C. Wilson, J. Appl. Cryst. 11102 (1978)
V. Uvarov, I. Popov, Mater. Charact. 85, 111 (2013)
M.M. Islam, T. Bredow, J. Phys. Chem. Lett. 6, 4622 (2015)
E. Rangasamy, J. Wolfenstine, J. Sakamoto, Solid State Ionics. 206, 28 (2012)
C. Liu, K. Rui, C. Shen, M.E. Badding, G. Zhang, Z. Wen, J. Power Sources. 282, 286 (2015)
V. Gajraj, A. Kumar, S. Indris, H. Ehrenberg, N. Kumar, C.R. Mariappan, Ceram. Int. 48, 29238 (2022)
S. Dahiya, R. Punia, S. Murugavel, A.S. Maan, Solid State Sci. 55, 98 (2016)
P. Wakudkar, A.V. Deshpande, J. Phys. Chem. Solids. 155, 110092 (2021)
P. Wakudkar, A.V. Deshpande, Solid State Ionics. 345, 115185 (2020)
G.R. Gajula, L.R. Buddiga, N. Vattikunta, Mater. Chem. Phys. 230, 331 (2019)
Acknowledgements
One of the authors would like to express sincere appreciation to VNIT, Nagpur, for providing a Ph.D. fellowship. The author appreciate the support of DST FIST project number SR/FST/PSI/2017/5(C) for the XRD facility provided by the Department of Physics at VNIT, Nagpur.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
MA: Material preparation, data analysis, writing original draft, conceptualization, editing, proof reading. AD: Editing, supervising, conceptualization.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aote, M., Deshpande, A.V. Study of Ge-doped garnet type Li7La3Zr2O12 as solid electrolyte for Li-ion battery application. J Mater Sci: Mater Electron 35, 586 (2024). https://doi.org/10.1007/s10854-024-12338-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10854-024-12338-5