Abstract
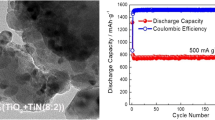
Tin-based materials have been considered as next-generation candidates to replace carbon as anode materials for lithium-ion batteries (LIBs) owing to their high theoretical capacities and electrical conductivity. However, the commercialization of tin-based materials is still challenging owing to volumetric expansion due to continuous cycling, which further degrades the lithium storage capacity. One strategy for mitigating this issue is the incorporation of intermetallic compounds into the electrode material, which buffers the mechanical stability of the electrode. Herein, we report the successful synthesis of tin-based intermetallic anode compounds (Co3Sn2, Mn2Sn, and Ni3Sn2) using a facile flux method. The prepared materials belonging to the P63/mmc space group were further implemented as the anode in LIBs, and a comparative analysis was conducted. Interestingly, among the prepared samples, the Mn2Sn electrode was found to offer the lowest sheet resistance (36.4 Ω sq−1) and charge transfer resistance (53.3 Ω), which were beneficial for enhancing the specific capacity up to approximately 379.2 mA h g−1 with extended stability up to 50 cycles at a current density of 0.05 A g−1.





Similar content being viewed by others
Data availability
The data that underlie the findings of this study are available from the corresponding authors upon reasonable request.
References
M. Armand, J. Tarascon, Issues and challenges facing rechargeable lithium batteries. Nature 414(6861), 359–367 (2001)
Y. Liang et al., Coupling of triporosity and strong Au–Li interaction to enable dendrite-free lithium plating/stripping for long-life lithium metal anodes. J. Mater. Chem. A 8(35), 18094–18105 (2020)
S. Chu, Y. Cui, N. Liu, The path towards sustainable energy. Nat. Mater. 16(1), 16–22 (2017)
Y. Chen et al., Ultrafine Mo-doped SnO2 nanostructure and derivative Mo-doped Sn/C nanofibers for high-performance lithium-ion batteries. Nanoscale 10(36), 17378–17387 (2018)
B. Li et al., Extraordinary lithium ion storage capability achieved by SnO2 nanocrystals with exposed 221 facets. Nanoscale 10(34), 16217–16230 (2018)
G.-A. Nazri, G. Pistoia, Lithium Batteries: Science and Technology (Springer, London, 2008)
Y. Chen et al., LiFePO4/C ultra-thin nano-flakes with ultra-high rate capability and ultra-long cycling life for lithium ion batteries. J. Alloys Compd. 749, 1063–1070 (2018)
L. Chen et al., Integrated structure of tin-based anodes enhancing high power density and long cycle life for lithium ion batteries. ACS Appl. Energy Mater. 3(9), 9337–9347 (2020)
X. Tang et al., Recent development of ionic liquid-based electrolytes in lithium-ion batteries. J. Power Sources 542, 231792 (2022)
T. Osaka et al., Surface characterization of electrodeposited lithium anode with enhanced cycleability obtained by CO2 addition. J. Electrochem. Soc. 144(5), 1709 (1997)
P. Arora, R.E. White, M. Doyle, Capacity fade mechanisms and side reactions in lithium-ion batteries. J. Electrochem. Soc. 145(10), 3647 (1998)
M. Obrovac, V. Chevrier, Alloy negative electrodes for Li-ion batteries. Chem. Rev. 114(23), 11444–11502 (2014)
M. Xia et al., Improving the electrochemical properties of a SiO@C/graphite composite anode for high-energy lithium-ion batteries by adding lithium fluoride. Appl. Surf. Sci. 480, 410–418 (2019)
Y. Mu et al., Growing vertical graphene sheets on natural graphite for fast charging lithium-ion batteries. Carbon 173, 477–484 (2021)
X.-L. Wang et al., Amorphous hierarchical porous GeOx as high-capacity anodes for Li ion batteries with very long cycling life. J. Am. Chem. Soc. 133(51), 20692–20695 (2011)
Y. Hwa et al., High capacity and rate capability of core–shell structured nano-Si/C anode for Li-ion batteries. Electrochim. Acta 71, 201–205 (2012)
P. Kim et al., High-capacity CVD-grown Ge nanowire anodes for lithium-ion batteries: simple chemical etching approach for oxide removal. J. Mater. Sci.: Mater. Electron. 32(2), 2103–2112 (2021)
M.I. Bodnarchuk et al., Colloidal tin–germanium nanorods and their Li-ion storage properties. ACS Nano 8(3), 2360–2368 (2014)
Y. Zhao et al., Significant impact of 2D graphene nanosheets on large volume change tin-based anodes in lithium-ion batteries: a review. J. Power Sources 274, 869–884 (2015)
P. Meduri et al., Hybrid tin oxide nanowires as stable and high capacity anodes for Li-ion batteries. Nano Lett. 9(2), 612–616 (2009)
R. Kim, D. Nam, H. Kwon, Electrochemical performance of a tin electrodeposit with a multi-layered structure for Li-ion batteries. J. Power Sources 195(15), 5067–5070 (2010)
T. Zhang et al., Yolk–shell-structured Si@TiN nanoparticles for high-performance lithium-ion batteries. RSC Adv. 12(30), 19678–19685 (2022)
Y. Xu, J. Guo, C. Wang, Sponge-like porous carbon/tin composite anode materials for lithium ion batteries. J. Mater. Chem. 22(19), 9562–9567 (2012)
Y. Xu et al., Uniform nano-Sn/C composite anodes for lithium ion batteries. Nano Lett. 13(2), 470–474 (2013)
G. Zhang et al., Tin quantum dots embedded in nitrogen-doped carbon nanofibers as excellent anode for lithium-ion batteries. Nano Energy 9, 61–70 (2014)
Y. Yu, C.H. Chen, Y. Shi, A tin-based amorphous oxide composite with a porous, spherical, multideck-cage morphology as a highly reversible anode material for lithium-ion batteries. Adv. Mater. 19(7), 993–997 (2007)
C. Gu et al., Non-aqueous electrodeposition of porous tin-based film as an anode for lithium-ion battery. J. Power Sources 214, 200–207 (2012)
W. Choi et al., Microstructure and electrochemical properties of a nanometer-scale tin anode for lithium secondary batteries. J. Power Sources 136(1), 154–159 (2004)
G. Derrien et al., Nanostructured Sn–C composite as an advanced anode material in high-performance lithium-ion batteries. Adv. Mater. 19(17), 2336–2340 (2007)
B. Ma et al., Hollow silicon–tin nanospheres encapsulated by N-doped carbon as anode materials for lithium-ion batteries. ACS Appl. Nano Mater. 1(12), 6989–6999 (2018)
Y.-C. Chen et al., Size effect of tin oxide nanoparticles on high capacity lithium battery anode materials. Surf. Coat. Technol. 202(4–7), 1313–1318 (2007)
W.M. Zhang et al., Tin-nanoparticles encapsulated in elastic hollow carbon spheres for high-performance anode material in lithium-ion batteries. Adv. Mater. 20(6), 1160–1165 (2008)
H. Sakaguchi et al., Ce–Sn intermetallic compounds as new anode materials for rechargeable lithium batteries. J. Power Sources 119, 50–55 (2003)
U. Nwokeke et al., The electrochemical behavior of low-temperature synthesized FeSn2 nanoparticles as anode materials for Li-ion batteries. J. Power Sources 196(16), 6768–6771 (2011)
W. Cui et al., Nanostructural CoSnC anode prepared by CoSnO3 with improved cyclability for high-performance Li-ion batteries. Electrochim. Acta 56(13), 4812–4818 (2011)
L. Chang et al., RE–Sn (RE = Y, Ce and Gd) alloys as anode materials for lithium-ion batteries. New J. Chem. 42(14), 11525–11529 (2018)
W. Dong et al., A robust and conductive black tin oxide nanostructure makes efficient lithium-ion batteries possible. Adv. Mater. 29(24), 1700136 (2017)
W. Ai et al., High-rate, long cycle-life Li-ion battery anodes enabled by ultrasmall tin-based nanoparticles encapsulation. Energy Storage Mater. 14, 169–178 (2018)
M.-G. Park et al., Sn-based nanocomposite for Li-ion battery anode with high energy density, rate capability, and reversibility. ACS Nano 12(3), 2955–2967 (2018)
F.-S. Ke et al., Fabrication and properties of three-dimensional macroporous Sn–Ni alloy electrodes of high preferential (1 1 0) orientation for lithium ion batteries. Electrochem. Commun. 9(2), 228–232 (2007)
C. Zhang et al., Preparation and electrochemical performances of nanoscale FeSn2 as anode material for lithium ion batteries. J. Alloys Compd. 457(1–2), 81–85 (2008)
H.-R. Jung, W.-J. Lee, Preparation and characterization of Ni–Sn/carbon nanofibers composite anode for lithium ion battery. J. Electrochem. Soc. 158(6), A644 (2011)
G. Wang et al., Stabilization of Sn anode through structural reconstruction of a Cu–Sn intermetallic coating layer. Adv. Mater. 32(42), 2003684 (2020)
G. Liu et al., Fully active bimetallic phosphide Zn0.5Ge0.5P: a novel high-performance anode for Na-ion batteries coupled with diglyme-based electrolyte. ACS Appl. Mater. Interfaces 14(28), 31803–31813 (2022)
Y.-S. Lee, K.-S. Ryu, Study of the lithium diffusion properties and high rate performance of TiNb6O17 as an anode in lithium secondary battery. Sci. Rep. 7(1), 1–13 (2017)
X. Lou et al., Crystal structure modification enhanced FeNb11O29 anodes for lithium-ion batteries. ChemElectroChem 4(12), 3171–3180 (2017)
Z. Li et al., Three-dimensionally ordered macroporous SnO2 as anode materials for lithium ion batteries. Ceram. Int. 42(16), 18887–18893 (2016)
F. Oliveira et al., Simple analytical method for determining electrical resistivity and sheet resistance using the van der Pauw procedure. Sci. Rep. 10(1), 1–8 (2020)
L.-F. Cui et al., Light-weight free-standing carbon nanotube-silicon films for anodes of lithium ion batteries. ACS Nano 4(7), 3671–3678 (2010)
Y. Ji et al., Polyoxometalate-based bottom-up fabrication of graphene quantum dot/manganese vanadate composites as lithium ion battery anodes. Chem. A Eur. J. 23(65), 16637–16643 (2017)
W. Zhang et al., A cyclized polyacrylonitrile anode for alkali metal ion batteries. Angew. Chem. 133(3), 1375–1383 (2021)
L. Zhong et al., Tin nanoparticles as an effective conductive additive in silicon anodes. Sci. Rep. 6(1), 1–8 (2016)
D. Guan et al., A comparative study of enhanced electrochemical stability of tin–nickel alloy anode for high-performance lithium ion battery. J. Alloys Compd. 617, 464–471 (2014)
M.O. Guler et al., Intermetallic Ni3Sn4-based graphene@carbon hybrid composites for lithium-ion batteries. Int. J. Energy Res. 42(9), 2961–2970 (2018)
C. Botas et al., Sn- and SnO2-graphene flexible foams suitable as binder-free anodes for lithium ion batteries. J. Mater. Chem. A 3(25), 13402–13410 (2015)
D. Yin et al., CuO nanorod arrays formed directly on Cu foil from MOFs as superior binder-free anode material for lithium-ion batteries. ACS Energy Lett. 2(7), 1564–1570 (2017)
G. Liu et al., Novel Ni–Ge–P anodes for lithium-ion batteries with enhanced reversibility and reduced redox potential. Inorgan. Chem. Front. 10, 699–711 (2023)
Funding
This work was supported by the Institute of Information & communications Technology Planning and Evaluation (IITP) Grant funded by the Korea government (MSIT) (No. 2021-0-00259, Development of a Fast Wireless Charging System for Portable Terminals with improved heat dissipation and shielding performance). This work was supported by the National Research Foundation of Korea (NRF-2021R1A2C1008272, FY2021). This work was supported by the DGIST institution specific program (21-BRP-07) and the faculty startup funds from DGIST.
Author information
Authors and Affiliations
Contributions
JK and BC contributed equally to this study. JK participated in writing the manuscript and performing the electrochemical experiments. BC synthesized the tin-based intermetallic materials (Co3Sn2, Mn2Sn, and Ni3Sn2) and collected the material characterization data. SV was involved in writing and editing the manuscript. KP and JB are the corresponding authors who contributed equally to this study.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, J., Cho, B., Vallem, S. et al. Comparative study of the (Co/Mn/Ni)xSny intermetallic compounds as anode active materials for lithium-ion batteries. J Mater Sci: Mater Electron 34, 1722 (2023). https://doi.org/10.1007/s10854-023-11093-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10854-023-11093-3