Abstract
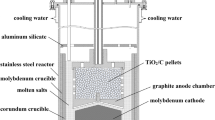
TiC0.5O0.5 was prepared through the carbothermal reduction of TiO2 under a flowing argon atmosphere, and then, it was used as an anode to prepare titanium metal through electrolysis in molten NaCl-KCl salt. The conductivities of the TiC0.5O0.5 samples at temperatures of 65.5–768.2 °C were investigated using the four-probe method, and the chemical valence of TiC0.5O0.5 was analyzed via X-ray photoelectron spectroscopy. Cyclic voltammetry and square-wave voltammetry were used to analyze the molten salt after electrolysis. The results show that the electrical conductivities of TiC0.5O0.5 are as high as 1.94 × 105 S·m−1 at 65.5 °C and 1.06 × 105 S·m−1 at 768.2 °C, suggesting that TiC0.5O0.5 can maintain good conductivity under high temperatures. There are Ti (II)–C, C–Ti (III)–O, and Ti (IV)–O bonds in the TiC0.5O0.5 structure. High-purity titanium powder was fabricated successfully via electrolysis at a voltage of 3.0 V for 4 h at 750 °C. The number of electrons transferred to the cathode during the reduction process was calculated to be 2.01 and 3.43, which corresponds, respectively, to the reduction reactions from Ti2+ and Ti3+/Ti4+ to titanium metal based on the results of square-wave voltammetry.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
G. Lütjering, J.C. Willliams, Titanium (Springer, Berlin, 2003), pp. 15–16
T. Mu, F. Zhu, B. Deng, Mater. Trans. 58, 535–538 (2017)
W. Kroll, Trans. Electrochem. Soc. 78, 35 (1940)
G.Z. Chen, D.J. Fray, T.W. Farthing, Nature 407, 361–364 (2000)
S.I. Pyosuke, O. Suzuki, Metall. Mater. Trans. B 34B, 277–285 (2003)
O.L.J. Wither, US Patent 20050166706. (2004)
S. Takeuchi, O. Watanabe, J. Jpn. Inst. Met. 28, 627–632 (1964)
Y. Hashimoto, J. Jpn. Inst. Met. 32, 1327–1334 (1968)
S. Jiao, H. Zhu, J. Mater. Res. 21, 2172–2175 (2006)
S. Jiao, H. Zhu, J. Alloys Compd. 438, 243–246 (2007)
C. Wu, M. Tan, G. Ye, D.J. Fray, X. Jin, A.C.S. Sustain, Chem. Eng. 7, 8340–8346 (2019)
B. Jiang, J. Xiao, K. Huang, J. Hou, S. Jiao, H. Zhu, J. Am. Ceram. Soc. 100, 2253–2265 (2017)
A. Afir, M. Achour, N. Saoula, J. Alloys Compd. 288, 124–140 (1999)
A. Maitre, D. Tetard, P. Lefort, J. Eur. Ceram. Soc. 20, 15–22 (2000)
K. Fu, F. Zhou, B. Liao, X. Wu, J. Yu, M. Ying, H. Zhou, X. Zhang, Ceram. Int. 41, 1701–1709 (2015)
J. Xiong, S. Xiong, Z. Guo, M. Yang, J. Chen, H. Fan, Ceram. Int. 38, 1815–1821 (2012)
O. Akhavan, E. Ghaderi, J. Phys. Chem. C. 113, 20214–20220 (2009)
L. Huang, L. Ai, M. Wang, J. Jiang, S. Wang, Int. J. Hydrog. Energy 44, 965–976 (2019)
C. Lu, L. Yang, B. Yan, L. Sun, P. Zhang, W. Zhang, Z. Sun, Adv. Funct. Mater. 30, 2000852 (2020)
D.-Y. Kim, B.N. Joshi, J.-J. Park, J.-G. Lee, Y.-H. Cha, T.-Y. Seong, S. In Noh, H.-J. Ahn, S.S. Al-Deyabe, S.S. Yoon, Ceram. Int. 40, 11089–11097 (2014)
Q. Qin, Y. Zhao, M. Schmallegger, T. Heil, J. Schmidt, R. Walczak, G. Gescheidt-Demner, H. Jiao, M. Oschatz, Angew. Chem. Int. Ed. 58, 13101–13106 (2019)
A.M. Jastrzębska, A. Szuplewska, A. Rozmysłowska-Wojciechowska, M. Chudy, A. Olszyna, M. Birowska, M. Popielski, J.A. Majewski, B. Scheibe, V. Natu, M.W. Barsoum, 2D Mater. 7, 025018 (2020)
L. Calvillo, G. Garcia, A. Paduano, O. Guillen-Villafuerte, C. Valero-Vidal, A. Vittadini, M. Bellini, A. Lavacchi, S. Agnoli, A. Martucci, J. Kunze-Liebhauser, E. Pastor, G. Granozzi, A.C.S. Appl, Mater. Interfaces 8, 716–725 (2016)
X. Ning, H. Åsheim, H. Ren, S. Jiao, H. Zhu, Metall. Mater. Trans. B 42, 1181–1187 (2011)
L.P. Polyakova, P. Taxil, E.G. Polyakov, J. Alloys Compd. 359, 244–255 (2003)
J.H. Christie, J.A. Turner, R.A. Osteryoung, Anal. Chem. 49, 1899–1903 (1977)
Acknowledgements
The authors thank financial support from the Programs for Natural Science Foundation of Guangxi (2019GXNSFAA185013), Science and Technology Major Project of Guangxi Province (AA18118030), and Guangxi Key Laboratory Development Foundation (GXYSSF1802).
Author information
Authors and Affiliations
Contributions
WL: performing the experiments, data collection and analysis, writing initial draft. BZ: data collection. HL: performing the experiments, TF: guidance. WL: guidance. FG: conceptualization, methodology, resources, supervision, validation, review and revised manuscript, financial support for the project.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Long, W., Zou, B., Li, H. et al. Fabrication of a TiC0.5O0.5 anode using the carbothermal method under a non-vacuum atmosphere and its application in metal titanium electrolysis. J Mater Sci: Mater Electron 33, 3045–3052 (2022). https://doi.org/10.1007/s10854-021-07506-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07506-w