Abstract
Optical coherence tomography enables quick scans of translucent objects in a simple environment. Here, we apply this technique to wood-based biofoam. We measure the geometrical properties of the foam, such as bubble eccentricity and density fluctuations, in addition to characterising the possible orientation of fibres. We find that the wood-based foams are extremely suitable for optical coherence tomography due to their translucent nature and large changes of optical density between air-filled bubbles and solid films. Measurement of bubble eccentricity revealed a reasonably high aspect ratio of 1:2, enabling the orientation of long cellulose fibres if added to the mixture. The results demonstrate an effective method to characterise foamlike metamaterials. Furthermore, focusing on eccentricity enables the adjustment of the foam’s manufacturing method and, in turn, helps to produce anisotropic structures.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Liquid foams consist of gas bubbles that are separated from each other by thin liquid films. The unique properties of foams find a wide range of purposes in nature and industry [1, 2]. Foam forming technology enables manufacturing of new products to replace plastics with biodegradable materials [3,4,5,6], the delivery of drugs [7] and thermal insulators [8] among others.
The ultimate question for the designer is how to select the best bubble distribution for a specific structure [9]. Therefore, in order to understand the internal foam structure, different techniques have been used to characterise them such as scanning electron microscopy (SEM), light microscopy (LM) and three-dimensional reconstruction approach using X-ray microcomputed tomography [10, 11]. In this article, we use optical coherence tomography (OCT), a non-invasive optical imaging technique that can provide an internal description of the material.
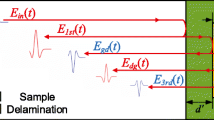
Optical coherence tomography (OCT) is a method based on low-coherence interferometry for depth-resolved imaging within turbid media. It has rapidly evolved in recent years, making it applicable to a large number of fields [12]. OCT is well known for its high depth and transversal resolution (decoupled from each other) and high probing depth in scattering media, along with its contact-free and non-invasive operation. It uses scattered light, from which it derives high resolution, cross-sectional imaging for non-invasive investigation using the Fourier diffraction projection theorem [12].
From the material science perspective, optimising and verifying the unit cell properties on a microscale create the desired properties on a macroscale [13]. Creating orientated rod-shaped structures, for example by elongating bubbles, increases the compression strength in one direction while reducing it in the cross-direction. This can be achieved in the laboratory scale by freeze casting increasing compression strength in the direction of the bubbles’ major axes by nearly fourfold compared to the bubbles’ minor axes [14, 15]. Following this idea, we have made a bio-based porous materials (Fig. 1) by introducing anisotropy by weighting its internal structure in a particular direction [3].
The internal structure of similar porous materials has been explored using different techniques [11, 16]. In general, the 2D methods such as SEM, light microscopy or direct CCD imaging yield similar results as X-ray microtomography, but the former is limited compared to 3D methods [17]. We will show that OCT is a suitable technique for our material due to its translucent properties.
Novel metamaterials are obtained by controlling the local bending and compression via local anisotropy. The simplest example of this is auxetic foams [18] with a negative poisson ratio. A more complicated example of how local anisotropy can be used is compliant mechanisms. These foamlike structures can have functionalities such as bistable switches [19] and, for example, hollow tubular structures, such as foams with elongated bubbles, which can bear enormous stresses [20].
Foam-based materials are the future solution for energy efficient materials due to their lightweight and insulation properties, similar to aerogels [15]. In addition, the reduction of materials in flow state [21] and solid state [22, 23] reduces waste, addressing the demand for eco-friendly materials.
The resolution of 1 \(\mu \mathrm {m}\) and imaging volume of \(1\, \hbox {mm}^3\) hits the sweet spot of current mass production of modern insulation and packaging materials. OCT is suitable for investigating the characteristic length, bubble size, grain size and fibre size. In this article, we use OCT to extract the 3D location and shape of the bubbles of our in-house made bio-based solid foam material. Using this technique, we characterise the anisotropic structures found in foam created by a simple industrially scalable process, using a proprietary cellulose-based FoamWood solution.
Materials and methods
A foam forming method was utilised to create the rods [3]. The material used is an aqueous solution of a proprietary FoamWood mixture containing fibres, cellulose and its derivatives, with a solids content of 15%.
a The schematic view of the set-up shows how the white foam is driven onto the hot plate from the foamer device via controlled air pressure. The linear stage moves as the foam is extruded, creating a solid, rod-shaped object that is dried by the radiant heater. b A picture of the measurement device showing the rod creation process
Figure 2 shows the set-up used to create the foam rods. The foam is deposited onto a solid surface from a nozzle. The final product was pushed from the foaming device, which consists of a sealed cup and a spinning blade, onto the solid plate using a pressure controller (pressure \(\Delta P = 0.1\) Pa). As the foam is deposited, the solid plate moves linearly, creating a rod of \(l = 150\, \hbox {mm}\) in length and \(w = 6\, \hbox {mm}\) in width. The hot plate moves at a speed of \(v= 1\, \hbox {mm}\,\hbox {s}^{-1}\). The process is repeated until 5 rods are deposited onto the plate in succession. The rods are dried using a radiant heater.
a In OCT systems, light is split into a reference arm that consists of mirrors and a sample arm. The light reflected from the sample and the mirror system recombine into the camera. The time dependent interference signal between the two beams determines the optical density of the sample in 3D. b Side view of a single rod. The scanned volume is marked with a green box. c Rendering figure of a 3D block of foam. The walls of the bubbles are shown as dark blue
The rods were then scanned using an optical coherence tomography system, GAN620 from Thorlabs®, which uses a laser with a wavelength of 900 nm. Figure 3a shows the device together with a sample of foam. The OCT system consists of one fixed and one moving mirror. By adjusting the position of the latter, an interference pattern is observed when the difference in optical path length between the two mirrors equals the difference in path length from the individual parts of the sample. The OCT used here is capable of measuring a depth of 1.4 mm in water, with a horizontal (x, y) resolution of \(\Delta x = 2.2 \, \mu \mathrm {m}\) and \(\Delta z = 1.64 \, \mu \mathrm {m}\) in depth direction. Consequently, this allows us to scan the internal structure of the samples with high resolution.
Multiple rods were scanned along a square cuboid of maximum size \(x \times y \times z = 10 \times 16 \times 2.01\,\hbox {mm}^3\). Figure 3b shows a single rod, where the green rectangle represents the area that was scanned. The reconstructed signal of that area is shown in Fig. 3c, where the major axis of the rod is along the y-axis and the z-axis is parallel to the direction of the laser beam. Once the sample is scanned, we proceed to detect and characterise the bubbles contained within the rod, which are seen as holes in Fig. 3c.
Results and discussion
Figure 4a shows an xz cross section of a rod, where the solid material appears in the white level, and the air around and inside the foam appears in the black level. The OCT is capable of scanning the entire thickness of the rod. Each surface point of the rod (x, y) provides a reflective signal from the material. As an example, Fig. 4b shows the signal captured along the blue line in Fig. 4a. Sharp refractive index variations between layers in the sample correspond to the intensity peaks in the interference pattern. The dashed black line indicates the mean air intensity, whose value is used as a threshold \(\mathcal {T}\). Above this value, the signal depicts the interaction with the films of the foam. Figure 4c shows the main steps for the image processing.
a 2D scan of the foam: solid material appears in the white level and the air in the black level. b Intensity of the backscattered light versus depth, an example of a signal captured by the OCT along the blue line in Fig. 4a. The mean value of the air is marked with a dashed black line, which corresponds to the threshold \(\mathcal {T}\). c A flow chart for image processing
The raw data were analysed using MATLAB and Python softwares. For each surface point (x, y), a Gaussian filter of standard deviation \(\gamma \), with a value range of \(0.4< \gamma < 0.75\), is applied.
Once the noise is attenuated, the signal is re-scaled using the threshold value \(\mathcal {T}\) given by the intensity of the surrounding air (dashed black line in Fig. 4b) to separate the solid phase from the porous phase. Every intensity value below the threshold \(I < \mathcal {T}\) is converted to zero, while \(I > \mathcal {T}\) to ones. We then extract the 26-connected voxels (3D pixel) from these binarized data, and each collection of neighbouring voxels is labelled as one bubble. The 26-connected voxels are neighbours to every voxel that touches one of their faces, edges or corners. The entire image processing is depicted in (Fig. 4c). Figure 5 shows a rendering of the rod with films in blue and the biggest bubbles detected in orange, confirming that the detection algorithm does detect bubbles. Here, to properly visualise the bubbles, we only show the bottom part of the rod.
Each bubble is labelled and its three dimensions measured. In order to determine the dimensions, we calculate the first and second central moments: the first is the centroid of the bubble \(\vec {r}\), and the second is its variance \(\sigma _i^2\) for \(i = (x,y,z)\). We assign each length of the bubble as \(\sigma _i\), which represents the expanse of the voxel group. The bubbles’ orientations are determined by the angles \(\alpha _i\) for \(i=(x,y,z)\). To compute them, the major eigenvector of every bubble is projected to each ordered pair.
Table 1 shows all the scanned volumes and the number of detected bubbles. There is no relation between the number of detected bubbles and the size of the sample. This is mainly related to the threshold \(\mathcal {T}\), which certainly depends on the structure of each sample.
Bubble size is restricted to a range \(V_\mathrm{{min}}< V < V_\mathrm{{max}}\): while the lower limit neglects clusters of only a few points, the upper limit removes the surrounding air. Using these limits, the number of bubbles for each sample is listed in Table 1. The average bubble size confirms that the bubbles are indeed elongated in a particular dimension.
To evaluate the degree of elongation, we calculate the length of the bubbles along the x, y and z directions. Figure 6a shows the standard deviation of y-coordinates, \(\sigma _y\), of the bubbles as a function of the standard deviation of x-coordinates, \(\sigma _x\). As we can see, the bubbles are elongated along the main axis of the rod (the y-axis) given that most of the points are above the black line and \(\sigma _y\) deviates from the relation \(\sigma _x = \sigma _y\), which represents bubbles of circular cross section. Moreover, Fig. 6b reveals that the bubbles are mainly flat.
a Bubble dimensions \(\sigma _x\) versus \(\sigma _y\) for all the samples in Table 1. Black dashed line represents \(\sigma _x = \sigma _y\). The relation \(\sigma _y > \sigma _x\) implies that the bubbles are elongated along the y-axis. b Bubble dimensions \(\sigma _z\) vs. \(\sigma _y\) for all the samples in Table 1. Dashed black line represents \(\sigma _z = \sigma _y\) and solid black line is \(\sigma _y = 8.25\sigma _z\). The relation \(\sigma _y > \sigma _z\) implies that the bubbles are flat
The fibre networks and bubble size distributions in mixing are characterised by a log-normal distribution [24]. To improve their description, each distribution of bubble dimensions was fitted by a log-normal distribution with coefficient A:
\(\tilde{\mu }\) and \(\tilde{\rho }\) being fitting parameters that describe the mean and standard deviation, respectively. Figure 7 shows the distribution regarding each axis. The excellent fit supports the fact that the deposition of the material on the plate follows a random process. We note that the intensity of the peaks is always higher and closer to zero for \(\sigma _x\), which means it is more likely to obtain narrower bubbles.
Lastly, to confirm that the major axes of the bubbles are in the main direction of the rod, we calculate the eigenvectors and -values of each bubble. Using the major eigenvectors \(\vec {v}\), we are able to determine the principal axes of the bubbles. The angle \(\alpha \), the major eigenvector formed with each ordered pair, represents the orientation of the bubble. It is defined as
where \(\hat{n}_i\) is the normal vector of each ordered plane i. Figure 8 shows the distribution of the angles for each scanned sample. The angles, \(\alpha _y\), between the vectors and the yz-plane centre around \(0^{\circ }\). Therefore, the major axes of the bubbles run mostly parallel to the y-axis, which is also the major axis of the rod, as previously mentioned.
Distribution of the bubble orientation for each sample in Table 1. The bubbles run along the major axis of the rod which is represented by the peak around \(0^{\circ }\)
OCT proofs to be an suitable technique for explore the internal structure for bio-based solid foams. In comparison with X-ray tomography, OCT is able to detect bubble (pore) size for similar materials [25]. The bubble orientation caused by foamer device can be also detected under the OCT [26]. Consequently, the internal structure studied here lead to significantly excellent macroscale properties of the bio-based foam [3].
Conclusions
The foam forming method described was used to create long rods made of fibres, cellulose and its derivatives. The resulting material contains a high number of bubbles, whose shape and orientation was studied using optical coherence tomography (OCT).
The optical properties of our bio-based foam allows us to explore its internal structure. By filtering the OCT signal, we were able to detect multiple spots (air pockets) inside the sample. We have shown that the bubbles are mainly longer in the main axis of the rod, which is attributed to the shear introduced during the manufacturing process as well as the foam forming and drying method. In addition, the short height of the bubbles allows them to be represented as flat ellipsoids, as shown in Fig. 9a. Finally, we have measured the orientation of each bubble, showing that the bubbles are in fact orientated along the y-axis. To visualise the bubble distribution inside the sample, Fig. 9b shows a top view of the rod, where each bubble is represented by an ellipse projected on the xy-plane.
As a consequence of the elongations of the bubbles, the surrounding mixture is also along the major axis of the rod. Therefore, the material properties described here prompts us to hypothesise that the fibres are distributed along the y-axis (see Appendix). The method used to make the rods creates an anisotropic material that enhances its mechanical properties [27]. Future work will focus on the mechanical properties of our bio-based rods [3].
From these results, we conclude that optical coherence tomography is a perfect technique to explore FoamWood solutions without any modification of the material.
References
Hill C, Eastoe J (2017) Foams: from nature to industry. Adv Colloid Interface Sci 247:4963. https://doi.org/10.1016/j.cis.2017.05.013
Li T, Chen C, Brozena AH, Zhu J, Xu L, Dreimeier C, Dai J, Rojas OJ, Isogai A, Wågberg L, Hu L (2021) Developing fibrillated cellulose as a sustainable technological material. Nature 590:47. https://doi.org/10.1038/s41586-020-03167-7
Reichler M, Rabensteiner S, Törnblom L, Coffeng S, Viitanen L, Jannuzzi L, Mäkinen T, Mac Intyre J, Koivisto J, Puisto A, Alava M (2021) Scalable method for bio-based solid foams that mimic wood. Sci Rep 24:306. https://doi.org/10.1038/s41598-021-03764-0
Jiang T, Duan Q, Zhu J, Liu H, Yu L (2020) Starch-based biodegradable materials: challenges and opportunities. Adv Ind Eng Polym Res 3:8. https://doi.org/10.1016/j.aiepr.2019.11.003
Kontturi E, Laaksonen P, Linder M.B, Gröschel A.H, Rojas O.J, Ikkala O (2018) Advanced materials through assembly of nanocelluloses. Adv Mater 1703:779. https://doi.org/10.1002/adma.201703779
Li J, Wei L, Leng W, Hunt JF, Cai Z (2018) Fabrication and characterization of cellulose nanofibrils/epoxy nanocomposite foam. J Mater Sci 53:4949. https://doi.org/10.1007/s10853-017-1652-y
Löbmann K, Wohlert J, Müllertz A, Wågberg L, Svagan A (2017) Cellulose nanopaper and nanofoam for patient-tailored drug delivery. Adv Mater Interfaces 4:1600655. https://doi.org/10.1002/admi.201600655
Pöhler T, Jetsu P, Fougerón A, Barraud V (2017) Use of papermaking pulps in foam-formed thermal insulation materials. Nord Pulp Paper Res J 32:367. https://doi.org/10.3183/npprj-2017-32-03-p367-374
Bhate D (2019) Four questions in cellular material design. Materials 12:1060. https://doi.org/10.3390/ma12071060
Niskanen, K. J. Papermaking science and technology: Book 16, Paper physics. (Finnish Paper Engineers’ Association/Paperi ja Puu Oy, Finland, 1998)
Marulier C, Dumont P, Orgéas L, Rolland du Roscoat S, Caillerie D (2015) 3D analysis of paper microstructures at the scale of fibres and bonds. Cellulose 22:1517. https://doi.org/10.1007/s10570-015-0610-6
Fercher A, Drexler W, Hitzenberger C, Lasser T (2003) Optical coherence tomography - principles and applications. Rep Prog Phys 66:239. https://doi.org/10.1088/0034-4885/66/2/204
Yu X, Zhou J, Liang H, Jiang Z, Wu L (2018) Mechanical metamaterials associated with stiffness, rigidity and compressibility: a brief review. Prog Mater Sci 94:114. https://doi.org/10.1016/j.pmatsci.2017.12.003
Scotti K, Dunand D (2018) Freeze casting - a review of processing, microstructure and properties via the open data repository, freezecasting.net. Prog Mater Sci 94:243. https://doi.org/10.1016/j.pmatsci.2018.01.001
Ghanadpour M, Wicklein B, Carosio F, Wågberg L (2018) All-natural and highly flame-resistant freeze-cast foams based on phosphorylated cellulose nanofibrils. Nanoscale 10:4085. https://doi.org/10.1039/C7NR09243A
Hjelt T, Ketoja JA, Kiiskinen H, Koponen AI, Pääkkönen E (2021) Foam forming of fiber products: a review. J Dispers Sci Technol 1:37. https://doi.org/10.1080/01932691.2020.1869035
Al-Qararah AM, Ekman A, Hjelt T, Kiiskinen H, Timonen J, Ketoja JA (2016) Porous structure of fibre networks formed by a foaming process: a comparative study of different characterization techniques. J Microsc 264(1):88–101. https://doi.org/10.1111/jmi.12420
Bhullar S (2015) Three decades of auxetic polymers: a review. e-Polymers 15:205. https://doi.org/10.1515/epoly-2014-0193
Gou Y, Chen G, Howell LL (2020) A design approach to fully compliant multistable mechanisms employing a single bistable mechanism. Mech Based Des Struct Mach 1d:24. https://doi.org/10.1080/15397734.2019.1707685
Rayneau-Kirkhope D, Mao Y, Farr R (2012) Ultralight fractal structures from hollow tubes. Phys Rev Lett 109:204301. https://doi.org/10.1103/PhysRevLett.109.204301
Lai CY, Rallabandi B, Perazzo A, Zheng Z, Smiddy S, Stone H (2018) Foam-driven fracture. Proc Natl Acad Sci 115:8082. https://doi.org/10.1073/pnas.1808068115
Al-Qararah A, Hjelt T, Kinnunen K, Beletski N, Ketoja J (2012) Exceptional pore size distribution in foam-formed fibre networks. Nord Pulp Paper Res J 27:226. https://doi.org/10.3183/npprj-2012-27-02-p226-230
Al-Qararah A, Hjelt T, Koponen A, Harlin A, Ketoja J (2013) Bubble size and air content of wet fibre foams in axial mixing with macro-instabilities. Colloids Surf A Physicochem Eng Asp 436:1130. https://doi.org/10.1016/j.colsurfa.2013.08.051
Alava M, Niskanen K (2006) The physics of paper. Rep Prog Phys 69:669. https://doi.org/10.1088/0034-4885/69/3/r03
Al-Qararah AM, Ekman A, Hjelt T, Ketoja JA, Kiiskinen H, Koponen A, Timonen J (2015) A unique microstructure of the fiber networks deposited from foam-fiber suspensions. Colloids Surf A Physicochem Eng Asp 482:544–553. https://doi.org/10.1016/j.colsurfa.2015.07.010
Paunonen S, Timofeev O, Torvinen K, Turpeinen T, Ketoja J (2018) Improving compression recovery of foam-formed fiber materials. BioResources 13:2
Alimadadi M, Uesaka T (2016) 3D-oriented fiber networks made by foam forming. Cellulose 23:661. https://doi.org/10.1007/s10570-015-0811-z
Acknowledgements
The authors acknowledge support from the Academy of Finland Center of Excellence programme, 278367. JK acknowledge the funding from Academy of Finland (308235) and FinnCERES flagship programme (151830423), Business Finland (211715) and Aalto University (974109903) as well as Aalto Science IT project for computational resources. LV acknowledges the funding from the Vilho, Yrjö and Kalle Väisälä Foundation via personal grant.
Funding
Open Access funding provided by Aalto University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The exact composition of bubble matrix is a proprietary biodgradeable foam manufactured in Aalto University in the project FoamWood.
Additional information
Handling Editor: Maude Jimenez.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A Scanning Electron Microscopy
Appendix A Scanning Electron Microscopy
A Zeiss EVO HD 15 scanning electron microscopy (SEM) was used to image the foam rods. Samples were coated with 4 nm gold–palladium using a Leica EM ACE 600 vacuum sputter coater to improve the conductivity of the samples and thus the quality of the SEM images. Figure 10 shows our best results. Both figures shows the fibres align in a particular direction, while Fig. 10a shows clusters of fibres within the foam, Fig. 10b shown few fibres coming out of the foam. The ridges continue as “whiskers” further confirming that we can create oriented structures not only in bubble, but also in the (nano)fibre level.
Foam rod under Scanning Electron Microscopy (same bar scale). a Clusters of fibres going in the same direction. b Multiple fibres coming out of the foam (white part). Figure is reproduced from [3]
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mac Intyre, J.R., Raka, D., Aydin, M. et al. Measuring biofoam anisotropy using optical coherence tomography. J Mater Sci 57, 11663–11672 (2022). https://doi.org/10.1007/s10853-022-07297-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-022-07297-3