Abstract
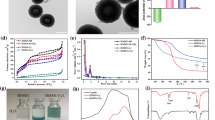
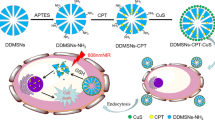
The cancer cell membrane (CCM) is widely used for nanomedicine encapsulation to improve their tumor-targeting ability. However, few researches are focused on drug release process and mechanism of these composite. Cell membrane fusion (two separate lipid membranes merge into a single continuous bilayer), one of the most fundamental processes in biology could be promoted by heating. Thus, in this manuscript, CCM was exploited as the gate keeper and tumor-targeting moiety of anticancer drug-loaded mesoporous silica nanoparticles (MSN). In addition, the drug release process and mechanism based on cell membrane fusion were studied. The loaded drugs included indocyanine green (ICG), a FDA-approved near-infrared photothermal agent for clinical applications, and doxorubicin (DOX), a chemotherapy drug. By coating CCM on drug-loaded MSN, the keeper could prevent unexpected drug leaking during the circulation and provide tumor targeting based on its specific recognition function. After arriving tumor tissue post-tail vein injection, the tumor position is irradiated by 808 nm light to trigger light–thermal conversion process of ICG to heat the composite (DOX-ICG@MSN@CCM) and promote cell membrane fusion between the encapsulated CCM and targeted tumor cell membrane. Such process can enhance the uptake efficacy of DOX-ICG@MSN@CCM to tumor cells. Furthermore, ICG could provide a synergistic photothermal therapy activity with DOX.








Similar content being viewed by others
References
Zhang Q, Wang XL, Li PZ et al (2014) Biocompatible, uniform, and redispersible mesoporous silica nanoparticles for cancer- targeted drug delivery in vivo. Adv Funct Mater 24:2450–2461
Zhang K, Xu LL, Jiang JG et al (2013) Facile large-scale synthesis of monodisperse mesoporous silica nanospheres with tunable pore structure. J Am Chem 135:2427–2430
Slowing II, Vivero-Escoto JL, Wu CW, Lin VSY (2008) Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv Drug Deliv Rev 60:1278–1288
Xiao D, Jia HZ, Zhang J, Liu CW, Zhuo RX, Zhang XZ (2014) A dual-responsive mesoporous silica nanoparticle for tumor-triggered targeting drug delivery. Small 10:591–598
Tarn D, Ferris DP, Barnes JC, Ambrogio MW, Stoddart JF, Zink JI (2014) A reversible light-operated nanovalve on mesoporous silica nanoparticles. Nanoscale 6:3335–3343
Chen CE, Pu F, Huang ZZ, Liu Z, Ren JS, Qu XG (2011) Stimuli-responsive controlled-release system using quadruplex DNA-capped silica nanocontainers. Nucleic Acids Res 39:1638–1644
Chen Z, Li Z, Lin Y, Yin M, Ren J, Qu X (2013) Bioresponsive hyaluronic acid-capped mesoporous silica nanoparticles for targeted drug delivery. Chem Eur J 19:1778–1783
Sun H, Su J, Meng Q et al (2017) Cancer cell membrane-coated gold nanocages with hyperthermia-triggered drug release and homotypic target inhibit growth and metastasis of breast cancer. Adv Funct Mater 27(3):1604300
Zhu J-Y, Zheng D-W, Zhang M-K et al (2016) Preferential cancer cell self-recognition and tumor self-targeting by coating nanoparticles with homotypic cancer cell membranes. NanoLett 16:5895–5901
Balasubramanian V, Correia A, Zhang HB et al (2017) Biomimetic engineering using cancer cell membranes for designing compartmentalized nanoreactors with organelle-like functions. Adv Mater 29:1605375
Chen WS, Zeng K, Liu H et al (2017) Cell membrane camouflaged hollow prussian blue nanoparticles for synergistic photothermal-/chemotherapy of cancer. Adv Funct Mater 27:1605795
Liu T, Shi C, Duan L et al (2018) A highly hemocompatible erythrocyte membrane-coated ultrasmall selenium nanosystem for simultaneous cancer radiosensitization and precise antiangiogenesis. J Mater Chem B 6:4756–4764
Chen Z, Zhao PF, Luo ZY et al (2016) Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy. ACS Nano 10:10049–10057
Guo X, Zhang Y, Liu J et al (2016) Red blood cell membrane-mediated fusion of hydrophobic quantum dots with living cell membranes for cell imaging. J Mater Chem B 4:4191–4197
Jahn R, Lang T, Südhof TC (2003) Membrane fusion. Cell 112:519–533
Wilschut J, Duzgunes N, Hoekstra D, Papahadjopoulos D (1985) Modulation of membrane fusion by membrane fluidity: temperature dependence of divalent cation induced fusion of phosphatidylserine vesicles. Biochem 24:8–14
Wang T, Li S, Zou Z et al (2018) A zeolitic imidazolate framework-8-based indocyanine green theranostic agent for infrared fluorescence imaging and photothermal therapy. J Mater Chem B 6:3914–3921
Shemesh CS, Hardy CW, Yu DS, Fernandez B, Zhang H (2014) Indocyanine green loaded liposome nanocarriers for photodynamic therapy using human triple negative breast cancer cells. Photodiagn Photodyn 11:193–203
Ran M, Tianyue J, Zhen G (2014) Enhanced anticancer efficacy by ATP-mediated liposomal drug delivery. Angew Chem 126:5925–5930
Mizutani H (2007) Mechanism of DNA damage and apoptosis induced by anticancer drugs through generation of reactive oxygen species. Yakugaku Zasshi 127:1837–1842
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (NO. 21571105 and 21671105), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD) and the Foundation of Jiangsu Collaborative Innovation Centre of Biomedical Functional Materials.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ding, Y., Zhu, Y., Wei, S. et al. Cancer cell membrane as gate keeper of mesoporous silica nanoparticles and photothermal-triggered membrane fusion to release the encapsulated anticancer drug. J Mater Sci 54, 12794–12805 (2019). https://doi.org/10.1007/s10853-019-03788-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-019-03788-y