Abstract
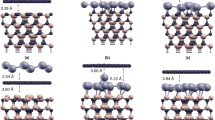
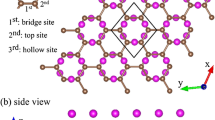
Herein, we studied the structure and hydrogenation of graphene supported on hydrogen-terminated SiC, with intercalated Li atoms. Strong nonbonded interactions occur between graphene and the Si–H groups. The latter were found to be significantly augmented by the intercalation of Li atoms, which enhanced the SiH::pi interactions. Although the electronic structure of graphene did not experience significant changes when supported on hydrogen-terminated SiC, a small gap of 0.04 eV was computed at the HSEH1PBE/6-31G* level. The clustering of Li atoms over graphene was prevented by the SiC support. A significant enhancement of reactivity could be corroborated due to the presence of intercalated Li atoms, given that the C–H binding energy was increased by almost 1 eV with respect to pristine graphene. We expect that hydrogen-terminated SiC with intercalated Li atoms can be used to enhance the chemical reactivity of graphene, given that it strongly interacts with graphene but does not compete for the electrons donated by Li.




Similar content being viewed by others
References
Denis PA, Iribarne F (2011) Comparative study of defect reactivity in graphene. J Phys Chem C 117:19048–19055
Denis PA (2013) Organic chemistry of graphene: the Diels–Alder reaction. Chem Eur J 19:15719–15725
Cao Y, Osuna S, Liang Y, Haddon RC, Houk KN (2013) Diels-Alder reactions of graphene: computational predictions of products and sites of reaction. J Am Chem Soc 135:17643–17649
Fan X, Nouchi R, Tanigaki K (2011) Effect of charge puddles and ripples on the chemical reactivity of single layer graphene supported by SiO2/Si substrate. J Phys Chem C 115:12960–12964
Gao X, Wang Y, Liu X, Chan TL, Irle S, Zhao Y, Zhang SB (2011) Regioselectivity control of graphene functionalization by ripples. Phys Chem Chem Phys 13:19449–19453
Denis PA, Pereyra C (2015) Structural characterization and chemical reactivity of dual doped graphene. Carbon 87:106–115
Denis PA, Pereyra C, Iribarne F (2014) Theoretical characterization of sulfur and nitrogen dual-doped graphene. Comp Theor Chem 1049:13–19
Denis PA (2014) Chemical reactivity and band-gap opening of graphene doped with gallium, germanium, arsenic, and selenium atoms. ChemPhysChem 15:3994–4000
Ferrighi L, Datteo M, Di Valentin C (2014) Boosting graphene reactivity with oxygen by boron doping: density functional theory modeling of the reaction path. J Phys Chem C 118:223–230
Chua CK, Sofer Z, Luxa J, Pumera M (2015) Selective nitrogen functionalization of graphene by Bucherer-type reaction. Chem Eur J 21:8090–8095
Denis PA (2011) Chemical reactivity of lithium doped monolayer and bilayer graphene. J Phys Chem C 115:13392–13398
Tapia A, Acosta C, Medina-Esquivel RA, Canto G (2011) Potassium influence in the adsorption of hydrogen on graphene: a density functional theory study. Comput Mater Sci 50:2427–2432
Logsdail AJ, Johnston RL, Akola J (2013) Improving the adsorption of Au Atoms and nanoparticles on graphite via Li intercalation. J Phys Chem C 117:22683–22695
Huang LF, Ni MY, Zhang GR, Zhou WH, Li YG, Zheng XH, Zeng Z (2011) Modulation of the thermodynamic, kinetic, and magnetic properties of the hydrogen monomer on graphene by charge doping. J Chem Phys 135:064705
Huang LF, Cao TF, Gong PL, Zeng A, Zhang C (2012) Tuning the adatom-surface and interadatom interactions in hydrogenated graphene by charge doping. Phys Rev B 86:125433
Denis PA (2013) Chemical reactivity of electron-doped and hole-doped graphene. J Phys Chem C 11:3895–3902
Denis PA, Iribarne F (2015) Strong N-doped graphene: the case of 4-(1, 3-Dimethyl-2, 3-dihydro-1 H-benzoimidazol-2-yl) phenyl) dimethylamine (N-DMBI). J Phys Chem C 119:5770–5777
Ambrosetti A, Silvestry PL (2016) Enhanced chemical reactivity of graphene on a Ni(111) substrate. J Chem Phys 144:111101
Zhao W, Gebhardt J, Spath F, Gotterbarm K, Gleichweit C, Steiruck HP, Gorling A, Papp C (2015) Reversible hydrogenation of graphene on Ni(111)—synthesis of “graphone”. Chem Eur J 21:3347
Wang QH, Jin Z, Kim K, Hilmer AJ, Paulus GLC, Shih CJ, Ham MH, Sanchez-Yamagishi JD, Watanabe K, Taniguchi T, Kong J, Jarillo-Hererro P, Strano MS (2012) Understanding and controlling the substrate effect on graphene electron-transfer chemistry via reactivity imprint lithography. Nat Chem 4:724–732
Ding Y, Peng Q, Gan L, Wu R, Ou X, Zhang Q, Luo Z (2016) Stacking modes induced reactivity enhancement for twisted bilayer graphene. Chem Mat 28:1034
Dion M, Rydberg H, Schroder E, Langreth DC, Lundqvist BI (2004) Van der Waals density functional for general geometries. Phys Rev Lett 92:246401
Soler JM, Artacho E, Gale JD, Garcia A, Junquera J, Ordejon P, Sanchez-Portal D (2002) The SIESTA method for Ab initio order-N materials simulation. J Phys 14:2745–2779
Ordejon P, Artacho E, Soler JM (1996) Self-consistent order-N density-functional Calculations for very large systems. Phys Rev B 53:R10441–R10444
Boys FS, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Mol Phys 19:553–566
Denis PA (2009) Denisty functional investigation of thiolated and thioepoxidated graphene. J Phys Chem C 113:5612–5619
Troullier N, Martins JL (1991) efficient pseudopotentials for plane-wave calculations. Phys Rev B 43:1993–2006
Denis PA, Iribarne F (2012) Cooperative behavior in functionalized graphene: explaining the occurrence of 1,3 cycloaddition of azomethine ylides onto graphene. Chem Phys Lett 550:111–117
Zhao Y, Truhlar DG (2006) A new local density functional for main-group thermochemistry, transition metal bonding, thermochemical kinetics, and noncovalent interactions. J Chem Phys 125:194101
Zhao Y, Truhlar DG (2008) Density functionals with broad applicability in chemistry. Theor Chem Account 120:215–241
Heyd J, Scuseria GE (2004) Assessment and validation of a screened coulomb hybrid density functional. J Chem Phys 120:7274–7280
Barone V, Scuseria GE (2004) Theoretical Study of the electronic properties of narrow single-walled carbon nanotubes: beyond the local density approximation. J Chem Phys 121:10376–10379
Hehre W, Radom L, PvR Schleyer, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New Work
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA et al (2009) Gaussian 09, revision D. Gaussian, Inc, Wallingford
Sun GF, Xia JF, Xue QK, Li L (2009) Nanotechnology 20:335701
Halle J, Neel N, Kroger J (2016) Filling the gap: Li-intercalated graphene on Ir(111). J Phys Chem C 120:5067
Vezin B, Dugourd P, Rayane D, Labastie P, Broyer M (1993) First observation of an excited state of Li2H. Chem Phys Lett 202:209–215
Acknowledgements
The author thanks PEDECIBA Quimica, CSIC, and ANII for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Denis, P.A. Structure and chemical reactivity of lithium-doped graphene on hydrogen-saturated silicon carbide. J Mater Sci 52, 1348–1356 (2017). https://doi.org/10.1007/s10853-016-0429-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-016-0429-z