Abstract
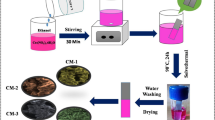
Spherical manganese carbonate (MnCO3) nanoparticles were prepared from manganese acetate by means of supercritical carbon dioxide (scCO2). The variation of morphology according to temperature, pressure, and solvent composition was studied. Quite uniformly sized MnCO3 nanoparticles were effectively produced in a short period of time with the aid of supercritical fluid. Furthermore, porous manganese oxide (MnOx) nanoparticles with high surface area were obtained via calcination process of MnCO3 nanoparticles, and its performance as a supercapacitor was investigated. The porous manganese oxide nanoparticles showed a high specific capacitance of 322 Fg−1 at 1 Ag−1 with excellent rate capability and cyclic stability. This study provides a new approach for the facile and eco-friendly fabrication of MnCO3 nanoparticles using scCO2 and their conversion to porous MnOx for supercapacitor application.







Similar content being viewed by others
References
Duan X, Ma J, Lian J, Kim T, Zheng W (2010) A novel surfactant-free route to MnCO3 steep rhombohedra crystals and their large-scale assembly into regular elongated patterns in a mixed solvent. Chem Commun 46:7133–7135
Pagnanelli F, Granata G, Moscardini E, Toro L (2013) Synthesis of MnCO3 nanoparticles by microemulsions: statistical evaluation of the effects of operating conditions on particle size distribution. J Nanopart Res 15:1887
Wang L, Tang F, Ozawa K, Chen ZG, Mukheri A, Zhu Y, Zou J, Cheng HM, Lu GQ (2009) A general single-source route for the preparation of hollow nanoporous metal oxide structures. Angew Chem 121:7182–7185
Yu SH, Colfen H, Antonietti M (2003) Polymer-controlled morphosynthesis and mineralization of metal carbonate superstructures. J Phys Chem B 107:7396–7405
Pal P, Pahari SK, Giri AK, Pal S, Bajaj HC, Panda AB (2013) Hierarchically order porous lotus shaped nano-structured MnO2 through MnCO3: chelate mediated growth and shape dependent improved catalytic activity. J Mater Chem A 1:10251–10258
Dang F, Hoshino T, Oaki Y, Hosono E, Zhou H, Imai H (2013) Synthesis of Li–Mn–O mesocrystals with controlled crystal phases through topotactic transformation of MnCO3. Nanoscale 5:2352–2357
Domingo C, Loste E, Gomez-Morales J, Garcia-carmona J, Fraile J (2006) Calcite precipitation by a high-pressure CO2 carbonation route. J Supercrit Fluid 36:202–215
Domingo C, Garcia-Carmona J, Loste E, Fanovich A, Fraile J, Gomez-Morales J (2004) Control of calcium carbonate morphology by precipitation in compressed and supercritical carbon dioxide media. J Cryst Growth 271:268–273
Wakayama H, Hall SR, Fukushima Y, Mann S (2006) CaCO3/biopolymer composite films prepared using supercritical CO2. Ind Eng Chem Res 45:3332–3334
Zhang C, Zhang J, Feng X, Li W, Zhao Y, Han B (2008) Influence of surfactants on the morphologies of CaCO3 by carbonation route with compressed CO2. Colloid Surf A 324:167–170
Xu S, Wu P (2014) A rapid, green and versatile route to synthesize metal carbonate superstructures via the combination of regenerated silk fibroin and compressed CO2. Cryst Eng Commun 16:1311–1321
Pudukudy M, Yaakob Z, Rajendran R (2014) Facile synthesis of mesoporous α-Mn2O3 microspheres via morphology conserved thermal decomposition of MnCO3 microspheres. Mater Lett 136:85–89
Ragupathy P, Park DH, Compet G, Vasan HN, Hwang SJ, Choy JH, Munichandraiah N (2009) Remarkable capacity retention of nanostructured manganese oxide upon cycling as an electrode material for supercapacitor. J Phys Chem C 113:6303–6309
Luo HM, Sun YX, Zhang FB, Zhang JQ, Feng HX (2014) Preparation and electrochemical performance of spherical MnOx with high capacitance. Ionics 20:1535–1546
Lee H, Kang J, Cho MS, Choi JB, Lee Y (2011) MnO2/graphene composite electrodes for supercapacitors: the effect of graphene intercalation on capacitance. J Mater Chem 21:18215–18219
Yang P, Li Y, Lin Z, Ding Y, Yue S, Wong CP, Cai X, Tan S, Mai W (2014) Worm-like amorphous MnO2 nanowires grown on textiles for high-performance flexible supercapacitors. J Mater Chem A 2:595–599
Lee SH, Yang J, Han YJ, Cho M, Lee Y (2015) Rapid and highly sensitive MnOx nanorods array platform for a glucose analysis. Sens Actuator B 218:137–144
Hu CC, Wang CC (2003) Nanostructures and capacitive characteristics of hydrous manganese oxide prepared by electrochemical deposition. J Electrochem Soc 150:A1079–A1084
Li W, Shao J, Liu Q, Liu X, Zhou X, Hu J (2015) Facile synthesis of porous Mn2O3 nanocubics for high-rate supercapacitors. Electrochim Acta 157:108–114
Zhang X, Yue L, Jin D, Zheng Y (2010) A facile template-free preparation of porous manganese oxides by thermal decomposition method. Inorg Mater 46:51–54
Wang L, Zhuo L, Zhang C, Zhao F (2015) Carbon dioxide-induced homogeneous deposition of nanometer-sized cobalt ferrite (CoFe2O4) on graphene as high-rate and cycle-stable anode materials for lithium-ion batteries. J Power Sources 275:650–659
Snow A, Ghaly AE, Cote R (2008) Treatment of stormwater runoff and landfill leachates using a surface flow constructed wetland. Am J Environ Sci 4:164–172
Cho MY, Roh KC, Park SM, Choi HJ, Lee JW (2010) Control of particle size and shape of precursors for ceria using ammonium carbonate as a precipitant. Mater Lett 64:323–326
Lee SH, Lee H, Cho MS, Nam JD, Lee Y (2013) Morphology and composition control of manganese oxide by the pulse reverse electrodeposition technique for high performance supercapacitors. J Mater Chem A 1:14606–14611
Gnanam S, Rajedran V (2011) Synthesis of CeO2 or α–Mn2O3 nanoparticles via sol–gel process and their optical properties. J Sol–Gel Sci Technol 58:62–69
Cheng F, Zhao J, Song W, Li C, Ma H, Chen J, Shen P (2006) Facile controlled synthesis of MnO2 nanostructures of novel shapes and their application in batteries. Inorg Chem 45:2038–2044
Song MK, Cheng S, Chen H, Qin W, Nam KW, Xu S, Yang XQ, Bongiorno A, Lee J, Bai J, Tyson TA, Cho J, Liu M (2012) Anomalous pseudocapacitive behavior of a nanostructured, mixed-valent manganese oxide. Nano Lett 12:3483–3490
Acknowledgements
This study was supported by the Energy Efficiency & Resources of the Korea Institute of Energy Technology Evaluation and Planning (KETEP) grant funded by the Korea government Ministry of Knowledge Economy (No. 20142010102690), and by the Basic Science Research Program through the National Research Foundation of Korea Grant funded by the Ministry of Science, ICT & Future Planning (2009-0083540). This study was also supported by the Industrial Strategic Technology Development program (10047780, Development of high power VSS for extreme temperature) funded by the Ministry of Trade, Industry and Energy of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Sang Ha Lee and Youngtak Kwon have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, S.H., Kwon, Y., Park, S. et al. Facile synthesis of MnCO3 nanoparticles by supercritical CO2 and their conversion to manganese oxide for supercapacitor electrode materials. J Mater Sci 50, 5952–5959 (2015). https://doi.org/10.1007/s10853-015-9133-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-015-9133-7