Abstract
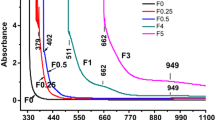
Glass samples of the system (100 − x) (0.5 Li2O–0.2Al2O3–0.3P2O5) + xSeO2 (x is ranging from 0 to 12 mol%, labeled as LAPS x ) were prepared using the melt quenching technique. FTIR spectral studies indicate that selenium ions mostly occupy network modifying sites due to isolated selenite (SeO3 2−) groups up to 6 mol% of SeO2 (LAPS6) in the LAPS glass network. This has a tremendous effect on the electrical properties. Glass forming ability parameter (K gl) and the glass transition temperature (T g) of LAPS samples were characterized by DTA traces. Electrical measurements were carried out as a function of frequency and temperature over the frequency range of 10 Hz to 106 Hz and a temperature range of 303–423 K. The electric modulus formalism was applied to study the relaxation behavior using the impedance data for all the samples at 303 K and also for analyzing the relaxation behavior of the highest conducting sample (6 mol% of SeO2) at different temperatures. An attempt has been made to relate the measured properties to the structural modifications due to the modifying effect of isolated selenite (SeO3 2−) groups in the glass network.











Similar content being viewed by others
References
Abrahams I, Hadzifejzovic E (2000) Solid State Ion 134:249
Jun Du, Beth J, Michael L (2005) Mater Lett 59:2821
Gao C, Drummond H (1999) J Am Ceram Soc 82(3):561
Scrosati B (2000) Electrochim Acta 45:2461
Ritchie AG (2004) J Power Sources 136:285
Baskaran GS, Fllower G, Veeraiah N (2007) J Alloys Compd 431:303
Kityk IV, Ebothe J, Qingsheng L, Shaoyong S, Fang J (2006) Nanotechnology 17:1871
Muller D, Ladwig G, Hallas E (1983) Phys Chem Glasses 24:37
Lee CH, Sohn HJ, Kim MG (2005) Solid State Ion 176:1237
Lee CH, Joo KH, Kim JH, Woo SG, Kang T, Park YJ, Oh Y (2002) Solid State Ion 149:59
Karabulut M, Metwalli E, Brow RK (2001) J Non Cryst Solids 283:211
Gedam RS, Deshpande VK (2006) Solid State Ion 177:2589
Balaji Rao R, Gerhardt RA (2008) Mater Chem Phys 112(1):186
Foltyh MF, Wasiucionek M, Nowinski JL (2005) Solid State Ion 176:2137
Varshneya AK (1994) Fundamentals of inorganic glasses. Academic Press, Inc., San Diego, p 96
Hruby A (1972) Czech J Phys 22:1187
Moreau F, Duran A, Munoz F (2009) J Eur Ceram Soc 29:1895
Elisa M, Cristina Vasiliu I, Grigoras CEA, Niciu H, Niciu D, Meghea A, Iftime N, Giurginca M, Trodahl HJ, Dalley M (2006) Opt Mater 28:621
Little Flower G, Srinivasa Reddy M, Baskaran GS, Veeraiah N (2007) Opt Mater 30:357
Yanko B, Dimitriev S, Yordanov I, Luben I (1995) J Non Cryst Solids 192:179
Maia LF, Rodrigues ACM (2004) Solid State Ion 168:87
Kluvanek P, Klement R, Karacona M (2007) J Non Cryst Solids 353:2004
Mogus-Milankovi A, Santi AS, Karabulut M, Day DE (2007) J Non Cryst Solids 330:128
Muthupari S, Raghavan SL, Rao KJ (1996) J Phys Chem 100:4243
Rolling B, Ingram MD (2002) J Non Cryst Solids 265:113
Alexander MN, Onarto PIK, Struck CW, Rozen JR, Tasker GW (1986) J Non Cryst Solids 79:137
Nobre MAL, Lafendri S (2001) J Phys Chem Solids 62:1999
Verhoef AH, Denhartog HW (1994) Solid State Ion 68:305
Elliot SR, Owens AP (1994) Solid State Ion 70:27
Almond DP, West AR (1983) Solid State Ion 11:57
Gerhardt RA (1994) J Phys Chem Solids 55:1491
Moynihan CT, Boesch LP, laberge NL (1973) Phys Chem Glasses 14:122
Ghosh A, Pan A (2000) Phys Rev Lett 84:2188
Schroder TB, Dyre JC (2000) Phys Rev Lett 84:310
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koti Reddy, C.V., Balaji Rao, R., Chandra Mouli, K. et al. Studies on lithium alumino phosphate glasses doped with selenium ions for hard electrolytes. J Mater Sci 47, 6254–6262 (2012). https://doi.org/10.1007/s10853-012-6545-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-012-6545-5