Abstract
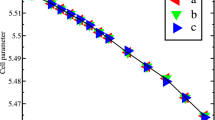
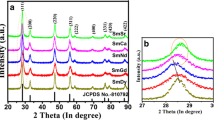
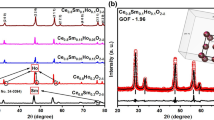
Solid oxide fuel cells (SOFCs) have been acknowledged as a possible future source for clean and efficient electric power generation. One of the most important goals in the SOFCs research is to decrease the operating temperature, which in turn will improve the stability and decrease the cost of various components enabling its widespread utilization. For realizing the aforementioned goal, it is imperative to identify suitable electrolyte materials that show enhanced conductivity in the intermediate temperature range (773–1,073 K). Sm-doped ceria (SDC) is considered a promising candidate for use as an electrolyte material for SOFC operation in intermediate temperature range due to the high oxygen ion conductivity. In this article, we present a theoretical investigation using first-principles and kinetic lattice Monte Carlo (KLMC) computations to highlight the trends in oxygen ion conductivity as a function of dopant content and temperature in SDC. Using first-principles calculations, oxygen vacancy formation and migration were examined at first, second, and third nearest neighbor positions to a Sm ion. The activation energies for oxygen vacancy migration along various pathways in SDC computed using first-principles were used as input to the KLMC model to study vacancy mediated diffusion. SDC with 20 % mole fraction of dopant content yields the maximum conductivity, which is in very good agreement with experimentally identified compositions. Rationale for increase in conductivity as a function of increase in dopant content and subsequent decrease in conductivity at higher dopant fractions in SDC is presented. This combined methodology of first-principles and KLMC computations is a useful tool for the design and identification of various ceria-based electrolyte materials used in SOFCs.




Similar content being viewed by others
References
Steele BCH, Heinzel A (2001) Nature 414:345
Inaba H, Tagawa G (1996) Solid State Ion 83:1
Trovarelli A (2002) Catalytic science series 2: catalysis by ceria and related materials. Imperial College Press, London
Mogensen M, Sammes NM, Tompsett GA (2000) Solid State Ion 129:63
Hibino T, Hashimoto A, Inoue T, Tokuno J, Yoshida S, Sano M (2000) Science 288:2031
Balazs GB, Glass RS (1995) Solid State Ion 76:155
Yahiro H, Eguchi K, Arai H (1989) Solid State Ion 36:71
Eguchi K, Setoguchi T, Inoue T, Arai H (1992) Solid State Ion 52:165
Yamashita K, Ramanujachary KV, Greenblatt M (1995) Solid State Ion 81:53
Kuharuangrong S (2007) J Power Sources 171:506
Yahiro H, Eguchi Y, Eguchi K, Arai H (1989) J Appl Electrochem 18:527
Kim DJ (1989) J Am Ceram Soc 72:1415
Dholabhai PP, Adams JB, Crozier P, Sharma R (2010) Phys Chem Chem Phys 12:7904
Dholabhai PP, Adams JB, Crozier P, Sharma R (2010) J Chem Phys 132:094104
Fu YP, Wen SW, Lu CH (2008) J Am Ceram Soc 91:127
Zha S, Xia C, Meng G (2003) J Power Sources 115:44
Huang W, Shuk P, Greenblatt M (1997) Solid State Ion 100:23
Souza ECC, Muccillo ENS (2009) ECS Trans 25:1643
Ou DR, Mori T, Ye F, Zou J, Auchterlonie G, Drennan J (2008) Phys Rev B 77:024108
Wei X, Pan W, Cheng L, Li B (2009) Solid State Ion 180:13
Andersson DA, Simak SI, Skorodumova NV, Abrikosov IA, Johansson B (2006) Proc Natl Acad Sci 103:3518
Nakayama M, Martin M (2009) Phys Chem Chem Phys 11:3241
Ismail A, Hooper J, Giorgi JB, Woo TK (2011) Phys Chem Chem Phys 13:6116
Hooper J, Ismail A, Giorgi JB, Woo TK (2010) Phys Rev B 81:224104
Hooper J, Ismail A, Giorgi JB, Woo TK (2010) Phys Chem Chem Phys 12:12969
Dholabhai PP, Anwar S, Adams JB, Crozier P, Sharma R (2011) J Solid State Chem 184:811
Dholabhai PP, Anwar S, Adams JB, Crozier PA, Sharma R (2012) Model Simul Mater Sci Eng 20:015004
Hohenberg P, Kohn W (1994) Phys Rev 136:B864
Kohn W, Sham LJ (1965) Phys Rev 140:A1133
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865
Dudarev SL, Botton GA, Savrasov SY, Humphreys CJ, Sutton AP (1998) Phys Rev B 57:1505
Bhochl PE (1994) Phys Rev B 50:17953
Kresse G, Joubert D (1999) Phys Rev B 59:1758
Kresse G, Hafner J (1993) Phys Rev B 47:558
Kresse G, Furthmuller J (1996) Phys Rev B 54:11169
Hellmann H (1937) An introduction to quantum chemistry. Deuticke, Leipzig
Feynman RP (1939) Phys Rev 56:340
Monkhorst HJ, Pack JD (1976) Phys Rev B 13:5188
Yang Z, Woo TK, Hermansson K (2006) J Chem Phys 124:224704
Jiang Y, Adams JB, Schilfgaarde MV, Sharma R, Crozier PA (2005) Appl Phys Lett 87:141917
Yang Z, Luo G, Lu Z, Woo TK, Hermansson K (2008) J Phys Condens Matter 20:035210
Yang Z, Luo G, Lu Z, Hermansson K (2007) J Chem Phys 127:074704
Skorodumova NV, Simak SI, Lundqvist BI, Abrikosov IA, Johansson B (2002) Phys Rev Lett 89:166601
Andersson DA, Simak SI, Skorodumova NV, Abrikosov IA, Johansson B (2007) Appl Phys Lett 90:031909
Shimojo F, Okazaki H (1992) J Phys Soc Jpn 61:4106
Yashima M, Kobayashi S, Yasui T (2007) Faraday Discuss 134:369
Steele BCH (2000) Solid State Ion 129:95
Dudek M, Rapacz-Kmita A, Mroczkowska M, Mosiałek M, Mordarski G (2010) Electrochim Acta 55:4387
Li B, Wei X, Pan W (2010) Inter J Hydro Energy 35:3018
Ji Y, Liu J, He T, Wang J, Su W (2005) J Alloys Compd 389:317
Omar S, Wachsman ED, Nino JC (2008) Solid State Ion 178:1890
Wang FY, Chen S, Cheng S (2004) Electrochem Comm 6:743
Acknowledgements
This article is based upon the work supported by the Department of Energy under the Grant No. DE-PS02-06ER06-17. The authors gratefully acknowledge the Fulton High Performance Computing Initiative (HPCI) at the Arizona State University for the computational resources. P.P.D thanks Shahriar Anwar, Peter A. Crozier and Renu Sharma for stimulating discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dholabhai, P.P., Adams, J.B. A blend of first-principles and kinetic lattice Monte Carlo computation to optimize samarium-doped ceria. J Mater Sci 47, 7530–7541 (2012). https://doi.org/10.1007/s10853-012-6398-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-012-6398-y