Abstract
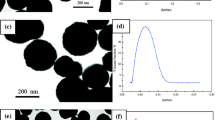
Iron oxide colloids were prepared by coprecipitation of Fe(II) and Fe(III) salts in alkaline media and stabilized by perchloric acid, oleic acid, or poly(acrylic acid). In an attempt to obtain magnetic polymer microspheres differing in size, dispersion polymerization of glycidyl methacrylate (GMA) in ethanol containing HClO4-stabilized magnetite, dispersion copolymerization of GMA and 2-hydroxyethyl methacrylate (HEMA) in toluene/2-methylpropan-1-ol mixture in the presence of oleic acid-coated magnetite, and inverse suspension copolymerization of N-isopropylacrylamide (NIPAAm) and N,N′-methylenebisacrylamide (MBAAm) in cyclohexane in the presence of poly(acrylic acid)-coated maghemite were compared. The microspheres were characterized by morphology, size, polydispersity, and some magnetic properties.




Similar content being viewed by others

References
Radisic M, Iyer RK, Murthy SK (2006) Int J Nanomed 1:3. doi:https://doi.org/10.2147/nano.2006.1.1.3
Xia N, Hunt TP, Mayers BT, Alsberg E, Whitesides GM, Westervelt RM et al (2006) Biomed Microdevices 8:299. doi:https://doi.org/10.1007/s10544-006-0033-0
Bulte JWM, Kraitchman DL (2004) NMR Biomed 17:484. doi:https://doi.org/10.1002/nbm.924
Lewin M, Carlesso N, Tung Ch H, Tang XW, Cory D, Scadden DT et al (2000) Nat Biotechnol 18:410. doi:https://doi.org/10.1038/74464
Fukumori Y, Ichikawa H (2006) Adv Powder Technol 17:1. doi:https://doi.org/10.1163/156855206775123494
Wust P, Gneveckow U, Johannsen M, Bohmer D, Henkel T, Kahmann F et al (2006) Int J Hyperthermia 22:673. doi:https://doi.org/10.1080/02656730601106037
Tennikova TB, Horák D, Švec F, Kolář J, Čoupek J, Trushin SV et al (1988) J Chromatogr A 435:357. doi:https://doi.org/10.1016/S0021-9673(01)82196-8
Lenfeld J, Švec F, Kálal J (1986) Acta Polym 37:377. doi:https://doi.org/10.1002/actp.1986.010370612
Kenawy ER, Abdel-Hay FI, El-Shanshoury AR, El Neseny MH (1998) J Control Release 50:145. doi:https://doi.org/10.1016/S0168-3659(97)00126-0
Heskins M, Guillet EJ (1968) J Macromol Sci Part A 2:1441
Shibayama M, Isono K, Okabe S, Karino T, Nagao M (2004) Macromolecules 37:2909. doi:https://doi.org/10.1021/ma0359685
Yoshida R, Sakai K, Okano T, Sakurai Y (1993) Adv Drug Deliv Rev 11:85. doi:https://doi.org/10.1016/0169-409X(93)90028-3
Castro Lopez V, Hadgraft J, Snowden MJ (2005) Int J Pharm 292:137. doi:https://doi.org/10.1016/j.ijpharm.2004.11.040
Lee CF, Wen CJ, Lin CL, Chiu WY (2004) J Polym Sci Part Polym Chem 42:3029. doi:https://doi.org/10.1002/pola.20085
Seida Y, Nakano Y (1996) J Chem Eng Jpn 29:767. doi:https://doi.org/10.1252/jcej.29.767
Lahann J, Mitragotri S, Tran TH, Kaido H, Sundaram J, Choi IS et al (2003) Science 299:371. doi:https://doi.org/10.1126/science.1078933
Ohya S, Kidoaki S, Matsuda T (2005) Biomaterials 26:3105. doi:https://doi.org/10.1016/j.biomaterials.2004.08.006
Pollert E, Knížek K, Maryško M, Závěta K, Lančok A, Boháček J et al (2006) J Magn Magn Mater 306:241. doi:https://doi.org/10.1016/j.jmmm.2006.03.069
Acknowledgements
Financial support of the Grant Agency of AS CR (grant KAN200200651) and the Academy of Sciences of the Czech Republic (grant 1QS100100553) is gratefully acknowledged. The authors thank Mrs. J. Hromádková of the Institute for electron microscopy measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horák, D., Pollert, E. & Macková, H. Properties of magnetic poly(glycidyl methacrylate) and poly(N-isopropylacrylamide) microspheres. J Mater Sci 43, 5845–5850 (2008). https://doi.org/10.1007/s10853-008-2836-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-008-2836-2