Abstract
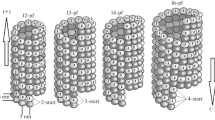
Microtubules (MTs) are fundamental structural elements in the cytoskeleton of eukaryotic cells. Their unique mechanical properties depend on the properties of the tubulin dimer, its interactions with surrounding dimers and the geometric organization within the MT. While the geometry has already been well described in experimental works, the mechanical characteristics of the dimer as well as of the individual monomers have up to date not been described. These may therefore provide new, additional insight to the microtubule tensile properties. In this paper we construct a mesoscale model of MT with a bottom-up approach. First, we evaluate the elastic constants of each of the two monomers together with the interaction force between them by means of molecular dynamics (MD) simulations carried out in an explicit water environment. Using the MD results, we model a 1 μm long MT as a cylinder constituted by interacting elastic elements and examine its properties via finite element method (FEM). The obtained results show an elastic constant value for α-tubulin of 11 N/m, while for the β-tubulin the elastic constant was measured to be 15.6 N/m. Concerning interactions between neighbouring monomers, the elastic constants along the protofilament (45 N/m for the intra-dimer interface and 18 N/m for the inter-dimer interface) are more rigid than elastic constants calculated for lateral interfaces (11 and 15 N/m). The mesoscale model provides mechanical properties of the whole MT, thus allowing the comparison with data obtained by other previous experimental and theoretical studies. We report here a Young modulus of 1.66 GPa for the MT under axial tension. In perspective our approach provides a simple tool for the analysis of MT mechanical behaviour under different conditions.










Similar content being viewed by others
References
Howard J (2001) In: Mechanics of motor proteins and the cytoskeleton. Sinauer, Sunderland, pp 119–134
Nogales E, Wolf SG, Downing KH (1998) Nature 391:199
Chrétien D, Wade RH (1991) Biol Cell 71:161
Felgner H, Frank R, Schliwa M (1996) J Cell Sci 109:509
Kurachi M, Hoshi M, Tashiro H (1997) Cell Mot Cyt 38:201
Venier P, Maggs AC, Pantaloni D (1994) J Biol Chem 269:13353
Kis A, Kasas S, Babi B, Kulik AJ, Benoît W, Briggs GAD, Schönenberger C, Catsicas S, Forró L (2002) Phys Rev Lett 89:248101
Gittes F, Mickey B, Nettleton J, Howard J (1993) J Cell Biol 120:923
Kasas S, Kis A, Riederer BM, Forró L, Dietler G, Catsicas S (2004) Chem Phys Chem 5:252
Jánosi IM, Chrétien D, Flyvbjerg H (2002) Biophys J 83:1317
VanBuren V, Cassimeris L, Odde DJ (2005) Biophys J 89:2911
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) Nucl Acid Res 28:235
Löwe J, Li H, Downing KH, Nogales E (2001) J Mol Biol 313:1045
Lindahl E, Hess B, van der Spoel D (2001) J Mol Mod 7:306
Berendsen HJC, Postma JPM, van Gunstersen WF, Hermans J (1981) In: Interaction model for water in relation to protein hydration. Reidel Publishing Company Dordrecht, Dordrecht, p 331
Berendsen HJC, Postma JPM, DiNola A, Haak JR (1984) J Chem Phys 81:3684
Nogales E, Whittaker M, Miligan RA, Downing KH (1999) Cell 96:79
Lenne PF, Raae AJ, Altmann SM, Saraste M, Hörber JKH (2000) FEBS Lett 476:124
Altmann SM, Grünberg RG, Lenne PF, Ylänne J, Raae AJ, Herbert K, Saraste M, Nilges M, Hörber JKH (2002) Struct 10:1085
Soncini M, Vesentini S, Ruffoni D, Orsi M, Deriu MA, Redaelli A (2006) Biomech Model Mechanobiol Nov 18;[Epub ahead of print]
Erickson HP, Stoffler D (1996) J Cell Biol 135:5
Acknowledgments
This research has been supported by the EST Marie Curie programme contract number MEST-CT-2004-504465 and by the Active Biomics STREP project contract number NMP4-CT-2004-516989.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deriu, M.A., Enemark, S., Soncini, M. et al. Tubulin: from atomistic structure to supramolecular mechanical properties. J Mater Sci 42, 8864–8872 (2007). https://doi.org/10.1007/s10853-007-1784-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-007-1784-6