Abstract
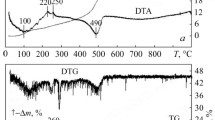
Al2O3–ZrO2 composite gel powder was prepared by sol–gel route. The gel precursor compositions were preferred to achieve yield of 5–15 mol% zirconia after calcination of respective powders. The precursor gel was characterized by Differential Thermal Analysis (DTA)/Thermo Gravimetric (TG), IR and X-ray Diffraction study (XRD). The analysis reveal the gel contained pseudoboehmite and amorphous Zr(OH)4, which was decomposed in three and two stages respectively. The phase transformation of alumina during calcination followed the sequence of pseudoboehmite → bayerite → boehmite → γ-Al2O3 → θ-Al2O3 → α-Al2O3, while that of ZrO2 follows amorphous ZrO2 → t-ZrO2 → (t + m) ZrO2. Fourier Transform Infrared Spectroscopy (FTIR) studies showed that the number of M–OH and M–O bond increases with zirconia due to a change in the cationic charge of the composite powder. Transmission Electron Microscopy (TEM) photograph of calcined powder exhibited the presence of dispersed as well as agglomerated nano sized spherical particles. SEM and Electron Probe Microscope Analysis (EPMA) confirmed the near uniform distribution of zirconia particles in the alumina matrix.








Similar content being viewed by others
References
Wefers K, Misra C (1987) Alcoa Technical Paper No. 19, Alcoa Laboratories, Pittsburgh, PA
Yarbrough WA, Roy R (1987) J Mater Res 2(4):494
Basu B (2005) Int Mater Rev 50(4):239
Basu B, Vleugels J, Van Der Biest O(2004) Mat Sci Engg A 366(2):338
Venkateswaran T, Sarkar D, Basu B (2005) J Am Ceram Soc 88(3):691
Hori S, Yoshimura M, Somiya S (1984) Am Ceram Soc. Columbus, OH, 794
Sproson DW, Messing GL (1984) J Am Ceram Soc 67(5):C92
Aksay IA, Lange FF, Davis BI (1983) J Am Ceram Soc 66(10):C190
Wu Y, Bandyopadhyay A, Bose S (2004) Mat Sci Engg A 380:349
Kagawa M, Kikuchi M, Syono YN (1983) J Am Ceram Soc 66(11):751
Caracoche MC, Rivas PC, Cervera MM, Caruso R, Benavádez E, De Sanctís O, Mintzer SR (2003) J Mater Res 18:208
Chen SG, Yin YS, Wang DP (2004) J Mol Struct 690:181
Guo GY, Chen YL (2001) J Mater Chem 11:1283
Zakharchenya RI, Vasilevskaya TN (1994) J Mater Sci 29:2806
Colomban PH (1989) J Mater Sci 24:3002
Low M, Mcpherson R (1989) J Mater Sci 24:892
Gitzen WH (1970) Am Ceram Soc Columbus, OH, 17
Lee HY, Werner R, Mordike BL (1992) J Eur Ceram Soc 10:245
Roger BB, Messing GL (1992) J Am Ceram Soc 82:825
Levin I, Brandon D (1998) J Am Ceram Soc 81(8):1995
Paglia G (2004) PhD Dissertation. Curtin Univeristy of Technology, Australia
Bhattacharyya S, Bharati S, Pratihar SK, Sinha RK, Behera RC, Ganguly RI (2003) Trans Indian Ceram Soc 62(1):18
Clearfield A, Nancollas GH, Blessing RH (1973) Ion exchange and solvent extraction. Marcel Dekker, New York, p 5
Rahaman MN (1995) Ceramic processing and sintering. Marcel Dekker, Inc, New York, p 115
Kim BN, Hiraga K, Morita K, Sakka Y (2001) Acta Mater 49:887
Sarkar D, Adak S, Cho SJ, Chu MC, Mitra NK (2007) Ceram Inter 33(2):255
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarkar, D., Mohapatra, D., Ray, S. et al. Nanostructured Al2O3–ZrO2 composite synthesized by sol–gel technique: powder processing and microstructure. J Mater Sci 42, 1847–1855 (2007). https://doi.org/10.1007/s10853-006-0737-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-006-0737-9