Abstract
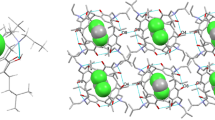
Tetrakis(l,3-dihydrobenzoxazine) calix[4]resorcinarenes 4–6 were synthesized via the Mannich reaction from the corresponding resorcin[4]arenes 1–3 with S-(−)-α-methylbenzylamine or R-(+)-α-methylbenzylamine and formaldehyde (aq.). The products were well characterized by FT-IR, 1H NMR, 13C NMR spectroscopies and single crystal X-ray diffraction analysis. Molecular structures of compounds 4, 5 and 6 showed the same R/S configuration to the starting amines. Compounds 4–6 are stabilized by a collar of intramolecular hydrogen bonding networks between the hydroxy groups and the oxygens from the benzoxazine rings. Compounds 4 and 5 could encapsulate the guest molecules of acetone and dichloromethane, respectively. The UV and 1H NMR titration experiments were performed to study the host-guest chemistry between compound 4 and small acetone molecules, indicating that compound 4 exhibited encapsulation behavior towards acetone molecules through hydrogen bonding interactions.















Similar content being viewed by others
References
Cram, D.J., Karbach, S., Kim, H.E., Knobler, C.B., Maverick, E.F., Ericson, J.L., Helgeson, R.C.: Cavitands as open molecular vessels form solvates. J. Am. Chem. Soc. 110, 2229–2237 (1988)
Yamanaka, M., Kobayashi, K.: Capsular assemblies of calix[4]resorcinarene-based cavitands. Asian J. Org. Chem. 2, 276–289 (2013)
Kobayashi, K., Yamanaka, M.: Self-assembled capsules based on tetrafunctionalized calix[4]resorcinarene cavitands. Chem. Soc. Rev. 44, 449–466 (2015)
Timmerman, P., Verboom, W., Reinhoudt, D.N.: Resorcinarenes. Tetrahedron 52, 2663–2704 (1996)
Shimizu, S., Shimada, N., Sasaki, Y.: Mannich-type reactions in water using anionic water-soluble calixarenes as recoverable and reusable catalysts. Green. Chem. 8, 608–614 (2006)
Dong, Y.B., Shi, H.Y., Yang, J., Liu, Y.Y., Ma, J.F.: Molecular dumbbell, sandwich, and paddle-wheel assembled with methylresorcin[4]arene cavitands and organooxotin clusters. Cryst. Growth Des. 15, 1546–1551 (2015)
Kim, B., Tripp, L.S., Wei, A.: Self-organization of large gold nanoparticle arrays. J. Am. Chem. Soc. 123, 7955–7956 (2001)
Wei, A., Kim, B., Pusztay, S.V., Tripp, S.L., Balasubramanian, R.: Resorcinarene-encapsulated nanoparticles: building blocks for self-assembled nanostructures. J. Incl. Phenom. Macro. 41, 83–86 (2001)
Wei, A.: Calixarene-encapsulated nanoparticles: self-assembly into functional nanomaterials. Chem. Commun. 15, 1581–1591 (2006)
Liu, J., Wei, A.: Prenucleation and coalescence of cobalt nanoclusters mediated by multivalent calixarene complexes. Chem. Commun. 28, 4254–4256 (2009)
Pei, W.-Y., Xu, G.-H., Yang, J., Wu, H., Chen, B.-L., Zhou, W., Ma, J.-F.: Versatile assembly of metal-coordinated calix[4]resorcinarene cavitands and cages through ancillary linker tuning. J. Am. Chem. Soc. 139, 7648–7656 (2017)
Coleman, A.W., Jebors, S., Shahgaldian, P., Ananchenko, G.S., Ripmeester, J.A.: Para-acylcalix[n]arenes: from molecular to macroscopic assemblies. Chem. Commun. 20, 2291–2303 (2008)
Shivanyuk, A.: Nanoencapsulation of calix[4]arene inclusion complexes. J. Am. Chem. Soc. 129, 14196–14199 (2007)
Beyeh, N.K., Pan, F., Rissanen, K.: Hierarchical ordering in ternary co-crystals of c60, n–benzyl ammonium resorcinarene bromide and solvent molecules. Cryst. Growth Des. 14, 6161–6165 (2014)
Puttreddy, R., Beyeh, N.K., Taimoory, S.M., Trant, M.D., Rissanen, J.F.: Host–guest complexes of conformationally flexible C-hexyl-2-bromoresorcinarene and aromatic N-oxides: solid-state, solution and computational studies. J. Org. Chem. 14, 1723–1733 (2018)
Kashapov, R.R., Razuvayeva, Y.S., Ziganshina, A.Y., Mukhitova, R.K., Sapunova, A.S., Voloshina, A.D., Nizameev, I.R., Marsil, K., Kadirov, M.K., Zakharova, L.Y.: Design of N–methyl–D–glucamine-based resorcin[4]arene nanoparticles for enhanced apoptosis effects. Mol. Pharm. 17, 40–49 (2020)
Chwastek, M., Szumna, A.: Higher analogues of resorcinarenes and pyrogallolarenes: bricks for supramolecular chemistry. Org. Lett. 22, 6838–6841 (2020)
Alver, C.A., Mauricio, M.: Preparation of methacrylate-based polymers modified with chiral resorcinarenes and their evaluation as sorbents in norepinephrine microextraction. Polymers 11, 1428 (2019)
Ligimol Louis, L., Alexander, V., Kumar, D.S., Senthan, S.A., Viveke, A.A.: Photoluminescence and electrochemical studies of tetranuclear ruthenium(II) polypyridyl complexes of benzimidazolyl functionalised pyrenylcalix[4]resorcinarene. Inorg. Chim. Acta 486, 245–251 (2019)
Liu, J.-L., Liu, X.-L., Jia, A.-Q., Shi, H.-T., Zhang, Q.-F.: Supramolecular structures and crystal stability of diisobutylaminomethylated calix[4]resorcinarenes. J. Incl. Phenom. Macro. 98, 49–56 (2020)
Ngodwana, L., Bout, W., Nqaba, Z., Motlokoa, T., Vatsha, B.: Methodologies for the derivatization of resorcin[4]arenes at the upper rim ortho-positions. Eur. J. Org. Chem. 2022, 1–13 (2022)
Betty, A.V.-S., Alver, C.-A., Zuly, J.R.-M., Mauricio, M.: Aminomethylated calix[4]resorcinarenes as modifying agents for glycidyl methacrylate (GMA) rigid copolymers surface. Polymers 11, 1427 (2019)
Sakowicz, A.M., Szumna, A.: Chiral water-soluble molecular capsules with amphiphilic interiors. Front. Chem. 10, 883093 (2022)
Amecke, R., Bohmer, V., Paulus, E.F., Vogt, W.: Regioselective formation of dissymmetric resorcarene derivatives with C4-symmetry. J. Am. Chem. Soc. 117, 3286–3287 (1995)
Atwood, J.L., Szumna, A.: Anion-sealed single-molecule capsules. Chem. Commun. (2003). https://doi.org/10.1039/B301511D
Lenz, K., Alexander, S., David, B.G., Julius, R.: Spin labeling monitors weak host–guest interactions. Chem. Commun. 3, 272–273 (2004)
Atwood, J.L., Szumna, A.: Hydrogen bonds seal single-molecule capsules. J. Am. Chem. Soc. 124, 10646–10647 (2002)
Sheldrick, G.M.: Sadabs: University of Göttingen. Göttingen, Germany (1996)
SMART and SAINT + for, Windows, N.T.: Version 6.02a, Bruker Analytical X-ray Instruments Inc., Madison (1998)
Sheldrick, G.M.: Shelxtl Software Reference Manual (Version 5.1). Bruker AXS Inc, Madison (1997)
Sheldrick, G.M.: A short history of SHELXTL. Acta Crystallogr. A64, 112–122 (2008)
Högberg, A.G.S.: Stereoselective synthesis and DNMR Study of two 1,8,15,22-Tetraphenyl [14]metacyclophan-3,5,10,12,17,19,24,26-octols. J. Am. Chem. Soc. 102, 6046–6050 (1980)
Moharem, T., El, G., Harry, H., Alexandra, M.Z.S.: Highly diastereoselective functionalisation of calix[4]resorcinarene derivatives and acid catalysed epimerisation reactions. Tetrahedron Lett. 36, 4905–4909 (1995)
Szumna, A.: Cyclochiral conformers of resorcin[4]arenes stabilized by hydrogen bonds. Org. Biomol. Chem. 5, 1358–1368 (2007)
Acknowledgements
This project was supported by National Natural Science Foundation of China (90922008).
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, XM., Wang, Q., Sun, M. et al. Tetrakis(benzoxazine) calix[4]resorcinarenes as hosts for small molecules. J Incl Phenom Macrocycl Chem 103, 289–299 (2023). https://doi.org/10.1007/s10847-023-01195-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-023-01195-0