Abstract
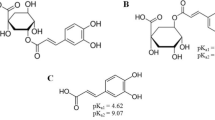
To investigate the inclusion ability of γ-cyclodextrin (γ-CD) for caffenic acid (CA). The conditions for the formation of inclusion complex and the binding constant between γ-CD and CA were determined by fluorescent and ultraviolet spectroscopic methods. The behavior of CA as a free radical scavenger before and after its inclusion was investigated. In addition, solid samples of the inclusion complex, prepared through the co-precipitation and grinding methods, were characterized via IR spectroscopy and differential scanning calorimetry. The inclusion complex was further characterized with 1H NMR spectroscopy. By using fluorescent and ultraviolet spectroscopy, the conditions for the formation of inclusion complex between γ-CD and CA were optimized and the binding constant determined. It was observed that the guest molecule behaves as a better anti-oxidant after its inclusion into γ-CD.












Similar content being viewed by others
References
Zhang, G., Shuang, S.: The new development of supramolecular chemistry of cyclodextrin in life science. J. Anal. Sci. 21(2), 200–204 (2005)
Zhang, A., Luo, F.: Study on supramolecular complexes of cyclodextrins and fenoxaprop-p-ethyl by UV spectra. Spectrosc. Spectr. Anal. 26(12), 2298–2301 (2006)
Yang, H.: A study on inclusion of β-CD and disperse dyes via UV–Vis spectrophotometry. Dyestuffs Coloration 43(2), 40–43 (2006)
Li, X.: Effects of two test methods on the test result of DPPH spectrophotometric assay. Biotechnology 17(4), 50–53 (2007)
Gao, R.: Preparation and characterization of a ferulic acid/α-cyclodextrin inclusion compound. China Surfactant Detergent Cosmetics 39(11), 94–97 (2004)
Catena, G.: The thermodynamic study on the effects of β-cyclodextrin inclusion with Anilino-naphthyl sulfonates. Anal. Chem. 8(61), 905–909 (1989)
Liu, Y.: Study of ketoprofen-hydroxypropyl-β-cyclodextrin inclusion compound. Chin. J. Mod. Appl. Pharm. 23(5), 377–379 (2006)
Yu, F., Wen, X.: Studies on equilibrium constants of DSF-β-CD_(HP) inclusion complexes and thermodynamic properties. J. Shenyang Pharm. University. 23(10), 630–632(2006)
Liu, J., Zhang, L.: Functions of cinnamic acid derivatives on DNA damage induced by Phen-Cu-VC system. Food Sci. 23(1), 49–51 (2003)
Cristiano, G., Karina, C.: Electrochemistry of caffeic acid aqueous solutions with pH 2.0–8.5. J. Braz. Chem. Soc. 13(3), 332–338 (2002)
Zhu, M., Wang, F.: The comparison of the anti-oxidizing property for caffenic acid and hydroxycinnamic acid. Sci. Technol. Food Ind. 2(9), 74–75 (2004)
Anselmi, C., Centini, M., Ricci, M., Buonocore, A., Granata, P., Tsuno, T., Facino, R.M.: Analytical characterization of a ferulic acid/γ-cyclodextrin inclusion complex. J. Pharm. Biomed. Anal. 40(4), 875–881 (2006)
Zhang, M., Li, J.: Preparation and spectral investigation of inclusion complex of caffeic acid with hydroxypropyl-β-cyclodextrin. Spectrochim. Acta A 71, 1891–1895 (2009)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, W., Chao, J., Du, R. et al. Spectroscopic studies on the inclusion behavior between caffenic acid and γ-cyclodextrin. J Incl Phenom Macrocycl Chem 71, 25–34 (2011). https://doi.org/10.1007/s10847-011-9930-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-011-9930-9