Abstract
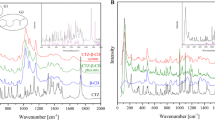
The inclusion complexes of uniconazole [(E)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-lyl)-1-penten-3-ol, UCZ] with two cyclodextrin derivatives, hydroxypropyl-β-cyclodextrin (HP-β-CD) and methylated-β-cyclodextrin (Me-β-CD), were prepared and characterized by 1H NMR and FT-IR. The phase solubility of UCZ and HP-β-CD, UCZ and Me-β-CD, which displays the ability of CDs complexation and solubilization, was studied in aqueous solutions and phosphate buffer solutions (PBS) with different property pH values (6.2, 7.2, 8.0). The solubility results indicated that the pH of PBS showed more enhancement on the interaction of HP-β-CD and UCZ than Me-β-CD with the increasing pH value, and the optimal pH value for complexation of UCZ and HP-β-CD, UCZ and Me-β-CD was at 8.0 and at 7.2, respectively. These were also determined by UCZ release behavior and dissolution studies of the complexes in solid state.







Similar content being viewed by others
References
Zhang, M., Duan, L., Tian, X., He, Z., Li, J., Wang, B., Li, Z.: Uniconazole-induced tolerance of soybean to water deficit stress in relation to changes in photosynthesis, hormones and antioxidant system. J. Plant Physiol. 164, 709–717 (2007)
Duan, L., Guan, C., Li, J., Eneji, A.E., Li, Z., Zhai, Z.: Compensative effects of chemical regulation with uniconazole on physiological damages caused by water deficiency during the grain filling stage of wheat. J. Agron. Crop Sci. 194, 9–14 (2008)
Cha-um, S., Puthea, O., Kirdmanee, C.: An effective in vitro acclimatization using uniconazole treatments and ex-vitro adaptation of phalaenopsis orchid. Sci. Hortic. 121, 468–473 (2009)
Ambrogi, V., Famiani, F., Perioli, L., Marmottinic, F., Di Cunzoloa, I., Rossia, C.: Effect of MCM-41 on the dissolution rate of the poorly soluble plant growth regulator, the indole-3-butyric acid. Microporous Mesoporous Mater. 96, 177–183 (2006)
Rika, K., Terumi, S., Toshiyuki, K.: Metabolism of uniconazole-P in water-sediment systems under illumination. Environ. Toxicol. Chem. 25, 310–316 (2006)
Szejtli, J.: Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 98, 1743–1754 (1998)
Tsai, Y., Tsai, H.H., Wu, C.P., Tsai, F.J.: Preparation, characterisation and activity of the inclusion complex of paeonol with β-cyclodextrin. Food Chem. 120, 837–841 (2010)
Wu, H.H., Liang, H., Yuan, Q.P., Wang, T.X., Yan, X.: Preparation and stability investigation of the inclusion complex of sulforaphane with hydroxypropyl-β-cyclodextrin. Carbohydr. Polym. 82, 613–617 (2010)
Szerman, N., Schroh, I., Rossi, A.L., Rosso, A.M., Krymkiewicz, N., Ferrarotti, S.A.: Cyclodextrin production by cyclodextrin glycosyltransferase from Bacillus circulans DF 9R. Bioresour. Technol. 98, 2886–2891 (2007)
Uekama, K., Hirayama, F., Irie, T.: Cyclodextrin drug carrier systems. Chem. Rev. 98, 2045–2076 (1998)
Mielcarek, J., Czernielewska, A., Czarczyńska, B.: Inclusion complexes of felodipine and amlodipine with methyl-β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 54, 17–21 (2006)
Servais, A.C., Fillet, M., Chiap, P., Dewé, W., Hubert, P., Crommen, J.: Influence of the nature of the electrolyte on the chiral separation of basic compounds in nonaqueous capillary electrophoresis using heptakis (2,3-di-O-methyl-6-O-sulfo)-β-cyclodextrin. J. Chromatogr. A 1068, 143–150 (2005)
Hanna, K., Brauer, C., Germain, P.: Solubilization of the neutral and charged forms of 2,4,6-trichlorophenol by β-cyclodextrin, methyl-β-cyclodextrin and hydroxypropyl-β-cyclodextrin in water. J. Hazard. Mater. 100, 109–116 (2003)
Özkana, Y., Ataya, T., Dïkmena, N., Işimera, A., Aboul-Enein, H.Y.: Improvement of water solubility and in vitro dissolution rate of gliclazide by complexation with β-cyclodextrin. Pharm. Acta Helv. 74, 365–370 (2000)
Higuchi, T., Connors, K.A.: Phase-solubility techniques. Adv. Anal. Chem. Instrum. 4, 117–212 (1965)
Piletska, E.V., Turner, N.W., Turner, A.P.F., Piletsky, S.A.: Controlled release of the herbicide simazine from computationally designed molecularly imprinted polymers. J. Control. Release 108, 132–139 (2005)
Gladys, G., Claudia, G., Marcela, L.: The effect of pH and triethanolamine on sulfisoxazole complexation with hydroxypropyl-β-cyclodextrin. Eur. J. Pharm. Sci. 20, 285–293 (2003)
Zhu, X.F., Duan, L.S., Tan, W.M., Li, Z.H., Tian, X.L., Wang, B.M.: The inclusion complex of uniconazole-β-cyclodextrin: preparation, characteristic and bioactivity. Chin. J. Pest. Sci. 12, 61–66 (2010)
Schneider, H.J., Hacket, F., Rudiger, V.: NMR studies of cyclodextrins and cyclodextrin complexes. Chem. Rev. 98, 1755–1785 (1998)
Hedges, A.R.: Industrial applications of cyclodextrins. Chem. Rev. 98, 2035–2044 (1998)
Ma, H.L., Wu, J.J., Liang, W.J., Chao, J.B.: Study on the association phenomenon of cyclodextrin to porphyrin J-aggregates by NMR spectroscopy. J. Incl. Phenom. Macrocycl. Chem. 58, 221–226 (2007)
Grillo, R., Melo, N.F.S., Fraceto, L.F.: Study of the interaction between hydroxymethylnitrofurazone and 2-hydroxypropyl-β-cyclodextrin. J. Pharm. Biomed. Anal. 47, 295–302 (2008)
Heise, H.M., Kuckuk, R., Bereck, A., Riegel, D.: Infrared spectroscopy and Raman spectroscopy of cyclodextrin derivatives and their ferrocene inclusion complexes. Vib. Spectrosc. 53, 19–23 (2010)
Liu, L.X., Zhu, S.Y.: Preparation and characterization of inclusion complexes of prazosin hydrochloride with β-cyclodextrin and hydroxypropyl-β-cyclodextrin. J. Pharm. Biomed. Anal. 40, 122–127 (2006)
Jun, S.W., Kim, M.S., Kim, J.S., Park, H.J., Lee, S., Woo, J.S., Hwang, S.J.: Preparation and characterization of simvastatin/hydroxypropyl-β-cyclodextrin inclusion complex using supercritical antisolvent (SAS) process. Eur. J. Pharm. Biopharm. 66, 413–421 (2007)
Ribeiro, L., Ferreira, D.C., Veiga, F.J.B.: In vitro controlled release of vinpocetine–cyclodextrin–tartaric acid multicomponent complexes from HPMC swellable tablets. J. Control. Release 103, 325–339 (2005)
Figueiras, A., Carvalho, R.A., Ribeiro, L., Labandeira, J.J.T., Veiga, F.J.B.: Solid-state characterization and dissolution profiles of the inclusion complexes of omeprazole with native and chemically modified β-cyclodextrin. Eur. J. Pharm. Biopharm. 67, 531–539 (2007)
Acknowledgments
The authors acknowledge the support of the National High Technology Research and Development Program of China (No. 2011AA10A206) and Transformation Fund for Agricultural Science and Technology Achievements of China (No. 2008GB23600451).
Author information
Authors and Affiliations
Corresponding author
Additional information
The first two authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhu, X., Tan, W., Zhou, F. et al. The effect of phosphate buffer solutions on uniconazole complexation with hydroxypropyl-β-cyclodextrin and methyl-β-cyclodextrin. J Incl Phenom Macrocycl Chem 73, 193–198 (2012). https://doi.org/10.1007/s10847-011-0042-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-011-0042-3