Abstract
Insect declines have become pronounced in prairie ecosystems, particularly in areas of agricultural intensification. Non-target pesticide exposure has been raised as a key concern for prairie remnant health. Over seven years, we screened grass and soil samples for the presence and quantities of hundreds of pesticides from five prairies in Minnesota and South Dakota that are designated as Critical Habitat for two globally endangered butterflies: Poweshiek skipperling and Dakota skipper. We documented dozens of pesticides across all sites and years on their larval host grasses. Interiors of prairies were not less likely to have detectable pesticides than along prairie-agriculture edges. Broad-spectrum organophosphate and pyrethroid insecticides were common in late season. Chlorpyrifos quantities were higher at sites where both endangered species have been extirpated. Neonicotinoid insecticides, which have been frequently attributed to declines in insect pollinators, did not present a significant exposure signal in these prairie habitats. Few pesticides were detected in soil or early season grass samples. The prevalence and quantities of pesticides we observed are likely underestimates, and additional research is needed on the landscape of risk at small and large scales. Implications for Conservation: Protected prairies are not immune to the risk of pesticide exposure, which may impact efforts to re-establish lost populations of imperiled prairie insects, including endangered butterflies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Declines in insect populations have received significant attention (Wepprich et al. 2019; Wagner 2020; Sánchez-Bayo and Wyckhuys 2021), but deciphering their scope and causal factors is complicated (Didham et al. 2020). Such declines appear to be especially pronounced in remnant grassland ecosystems, particularly in areas of high agricultural intensification (Swengel and Swengel 2015; Habel et al. 2019; Seibold et al. 2019; Raven and Wagner 2021). The increasingly fractured remnants of North America’s endangered tallgrass prairies (Samson and Knopf 1996; Ricketts et al. 1999; Lark et al. 2019) are under stressors from many factors, including invasive species, incompatible management schemes, isolation, and climate change. However, perhaps more than any other factor, non-target exposure to agricultural pesticides has been suggested as a key driver in the declines of grassland dependent species (Mineau et al. 2005; Gibbs et al. 2009; Gibbons et al. 2015; Forister et al. 2016; Sánchez-Bayo and Wyckhuys 2019).
Two historically widespread butterflies endemic to central North American prairies, the Poweshiek skipperling (Oarisma poweshiek Parker: Hesperiidae) and Dakota skipper (Hesperia dacotae Skinner: Hesperiidae), have declined dramatically in recent decades (Royer and Marrone 1992a, b; Swengel et al. 2011). The Poweshiek skipperling, extirpated from > 99% of its historic known locations (Belitz et al. 2018), is listed as Endangered in the United States (US Fish and Wildlife Service 2014) and Canada (COSEWIC 2014a) and Critically Endangered on the IUCN Red List (Royer 2020), ranking among the most imperiled species in the world. Similarly, the Dakota skipper has been extirpated from at least 76% of all historic locations, and is now categorized as Threatened in the United States (US Fish and Wildlife Service 2014), Endangered in Canada (COSEWIC 2014b), and globally Endangered by the IUCN (Royer 2019).
The widespread extirpation of these and other prairie dependent butterflies coincides with large-scale changes in applications of several agricultural insecticides in the north-central United States during the mid-2000s. Of particular note is the essentially concurrent (1) introduction of the new class of neonicotinoid insecticides, which are now nearly universally applied as a seed coat to corn, and (2) invasion of the economically damaging soybean aphid (Aphis glycines Matsumura) in 2000 (Ragsdale et al. 2011) against which the broad spectrum organophosphate (particularly chlorpyrifos) and pyrethroid insecticides became the primary means of control in the early 2000s. These two classes of pesticides produce different exposure routes. Neonicotinoids are generally applied proactively as systemic seed coatings and may be transported via dust during planting early in the growing season (Tapparo et al. 2012) onto non-target plants or incorporated into soils or waters (Main et al. 2014, 2015; Hladik et al. 2014; Jones et al. 2014; Bonmatin et al. 2015; Williams and Sweetman 2019). Neonicotinoids are now the world’s most widely applied class of insecticides and have been widely posited as a primary driver of declines of butterflies, other pollinators, and indeed broad swaths of wildlife (Mason et al. 2013; Goulson 2013; Fairbrother et al. 2014; Gibbons et al. 2015; Pecenka and Lundgren 2015; Gilburn et al. 2015; Forister et al. 2016; Basley and Goulson 2018; Olaya-Arenas and Kaplan 2019). Conversely, organophosphates and pyrethroids are contact insecticides that are typically applied reactively later in the summer in response to pest pressures via aerial spraying and thus may be blown or absorbed into rain (Foreman et al. 2000; Mackay et al. 2014) that falls onto non-target areas.
Given the correlated declines of imperiled prairie butterflies and changes in pesticide applications, we document the presence and quantities of pesticides and their residues on putative larval host grasses of Dakota skipper and Poweshiek skipperling from prairie remnants formally designated as Critical Habitat for them in Minnesota and South Dakota between 2014 and 2020. We sought to assess how their composition and quantities varied across these sites and across years, if their occurrence and quantities would be lower within prairie interiors than along associated agricultural margins, how pesticide prevalence related to population and extirpation status, and how this composition might affect conservation practices.
Methods
Sample collections
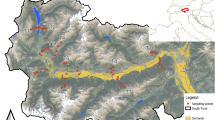
We collected samples from four prairie remnants (“Sites”) in Minnesota (“Clay”, “Pope”, “Lincoln”, and “Pipestone”) and one in South Dakota (“Day”) (Fig. 1). All of the Minnesota sites are designated as Critical Habitat for both Poweshiek skipperling and Dakota skipper (US Fish and Wildlife Service 2015). Dakota skipper remains extant at two of the sites (Day and Clay) but is extirpated from the remaining three (Pope, Pipestone, and Lincoln). Poweshiek skipperling historically occurred at all sites but is now extirpated from the region. These sites are also home to other species of conservation concern, including regal fritillary (Argynnis idalia Drury: Nymphalidae). The exact site names and specific GPS locations of sample points are censored here due to the presence of federally protected species at some sites.
From 2014 to 2020, we collected samples from between two to nine points per prairie site. Using aerial imagery in a GIS, we partitioned each prairie into 10 × 10 m grid cells, and classified cell Locations as either “edge” (within 10 m of an agricultural field) or “interior” (≥ 100 m from any edge). We then used a random number generator to select interior and edge cell coordinates for sampling. Given our interest in understanding the potential exposure to federally protected butterflies and the high-quality prairies they depend upon, we eliminated randomly selected interior locations that did not comprise suitable habitat. Fewer edge samples were collected in some years due to budgetary constraints.
Within each selected cell, we clipped > 5 g from a single, randomly selected little bluestem (Schizachyrium scoparium (Michx.) Nash: Poaceae) or clipped from a cluster of immediately adjacent little bluestems if biomass on the initially selected plant was low. If little bluestem was not available in sufficient quantities within a grid cell, we instead sampled from big bluestem (Andropogon gerardii Vitman). These grasses are indicative of intact native prairies and are among the host plants for Poweshiek skipperling, Dakota skipper, and other imperiled prairie skippers (Dana 1991; Royer et al. 2008; Rigney 2013; Seidle et al. 2018; Nordmeyer et al. 2021; Henault and Westwood 2022). We also collected paired > 25 g of soil beneath subsets of grass samples in 2014, 2015, and 2016, sieving the soil through a 1.27 mm mesh to homogenize particulate sizes for analysis. We cleaned collection supplies (scissors, collection bowl, hand shovel, mesh sieve) with 100% acetone in the field between samples. Clean nitrile gloves were changed between each sample collection. All samples were double-bagged in 1 quart plastic zip-loc bags, immediately placed on dry ice in the field, and then transferred to a -20oC freezer at the Minnesota Zoo until overnight shipment to the laboratory for analysis.
Sampling occurred in two seasons: (1) shortly after the planting season in early June 2015 and mid-May 2016, and (2) late summer from mid-August to early September in 2014 through 2020. Samples were generally collected on one date within each season for each site. These sampling periods were selected to coincide with the likelihood for the applications of different classes of pesticides. Of particular interest were the potential occurrence of neonicotinoid insecticides early in the season following seed-coated crop plantings, and the later summer aerial applications of broad-spectrum insecticides targeting soybean aphid and other agricultural pests. Due to high per sample testing cost and minimal detections in early season samples (see below), we elected not to continue early season sampling after 2016. Sites generally were sampled without information on when or where spraying may have occurred relative to sampling. However, we had the opportunity for a single time series sample collection when an airplane-applied spray event was observed adjacent to the Pipestone site within an hour of the cessation of the collection of the first set of samples in 2014. A second set of samples was then collected the following day from many of the same cells to quantify “Before” and “After” changes in pesticides levels.
Pesticides screening
Samples were screened for pesticide residues at the U.S. Department of Agriculture Agricultural Marketing Service’s National Sciences Laboratories (“NSL”; Gastonia, NC) using the QuEChERS extraction method (AOAC 2007.01; Anastassiades et al. 2003). The NSL performed all extractions from the provided substrates (either grass or soil) and then conducted Liquid Chromatography tandem mass spectrometry to determine the quantities of pesticides and some of their residues in the samples in reference to certified analyte standards for each compound. The NSL conducted all independent quality assurance and quality control standards and re-analyzed some samples as needed to satisfy control standards.
The identities, quantities, and minimum Levels of Detection (LOD) of a total of 233 pesticides and their residues were reported for each sample, with some variation in the number of analytes between years (2014 and 2015: 174; 2016: 178; 2017 and 2018: 199; 2019 and 2020: 193) (Appendix 1). LODs varied across compounds but were internally consistent for all samples within a year. Individual compounds within a sample were reported as either (1) “Not Detected” (i.e., below their LOD), (2) “Trace” (above their LOD but lower than a level that could be quantified), or (3) quantified on a parts per billion (ppb) basis (i.e. ng/g of homogenized sample material). We treated chemicals categorized as “Not Detected” as zeroes and considered a compound to be “Present” in a sample if it was either reported as “Trace” or if amounts could be specifically quantified. Extraction protocols and some instrumentation at the NSL were upgraded between the 2016 and 2017 samples, resulting in generally lower LODs and some additional analyzed pesticides, so 2017–2020 data are likely to be more quantitatively robust. We prepared and submitted blind replicates of four samples in 2014 (Appendix 2: Su8, Su10, Su135, Su127) to verify consistency; all replicate samples varied in observed quantities only slightly.
Temporal and spatial variation
We summarize descriptive information about variation in pesticide occurrence, composition, and quantities between seasons (early vs. late) and substrates (grass vs. soil). We also tested how the number of insecticide types and the quantities of some of these insecticides varied in late season grass samples across sites and years, and if the high-quality habitat interior locations of those prairie sites had lower insecticide occurrences and quantities than locations along prairie-agricultural edges. We excluded herbicides and fungicides from these statistical tests given (1) the relative rarity of herbicides in late season samples, and (2) the technical differences in fungicide detection between the 2014–2016 and 2017–2020 samples.
We tested the ability of the fixed effects of sites, years, and location nested within site, as well as the interaction between site and year, to explain variation in the number of detected insecticides on grasses in late summer with an Analysis of Deviance specifying a Poisson distribution in R using the glm package (R Core Team 2023; Bates et al. 2015). We similarly tested how the quantities (ppb) of chlorpyrifos and cyhalothrin on late season grasses differed with these same main effects and interactions with lm Analysis of Variance in R. Other compounds were too rarely documented to facilitate analyses. For these quantity analyses, we included data from after the known 2014 Pipestone spray event (test significance did not change if the pre-spray data were instead included). Since LOD is always less than or equal to the Level of Quantification (LOQ), we estimated the ppb for trace samples to be equal to the associated LOD. We truncated the dataset for the chlorpyrifos and cyhalothrin quantity tests to include only those points where these insecticides were detected to avoid overdispersion. The remaining quantities were log10 transformed for analysis to improve normality. We also excluded from the chlorpyrifos quantity analysis a single sample (Su86; 2290 ppb, Appendix 2) from Pope in 2017 that was highly leveraged even following log transformation; significance values were similar with and without this datapoint. We tested for statistically significant groups between the main effects of site and year for all analyses using post hoc Tukey tests.
Results
We detected eight insecticides, three herbicides, 10 fungicides, and three other compounds from 226 samples, across all years, seasons, sites, and substrates. All data are presented in Appendices 2 (late season grass samples), 3 (early season grass samples), and 4 (paired soil samples), and summarized graphically in Fig. 2. Pesticides were prevalent on grasses, particularly in late season, but were rare in the paired soil samples (only 5 of 46 samples). Soil samples were significantly less likely to have at least one detectable pesticide than their paired grass samples (χ21,92 = 21.45, p < .0001). This was true for both early season (χ21,60 = 9.93, p = .0016) and late season (χ21,32 = 12.70, p = .0004) sampling periods. Four pesticides (chlorpyrifos, clothianidin, atrazine, and tebuconazole) were detected in the soil samples. Notably, these single samples of clothianidin and tebuconazole are the only detections of these pesticides in the entire dataset.
Pesticides were prevalent on grasses in late season but rare in the early season. Spanning seven insecticides, three herbicides, and nine fungicides, 121 of 142 samples of the August and September grass samples from 2014 to 2020 contained at least one pesticide. In contrast, only a single pesticide (the herbicide atrazine) was detected grass samples in 21 of 38 samples during the two early season periods in May 2015 and June 2016.
Late season grass insecticides
Chlorpyrifos was by far the most frequently detected pesticide in the entire dataset, in 108 of the 142 (76.1%) late season grass samples. This organophosphate insecticide was detected in every year of the study and in every sample from 2018 to 2020. Most chlorpyrifos quantities ranged between 1 and 50 ppb (78 of the 88 quantifiable samples; global average: 49.9 ppb, median: 11.6), but a single sample contained of 2290 ppb (Appendix 2). Two other insecticides, cyhalothrin and bifenthrin, were each present in about a quarter of all late season samples (27.5% and 23.9%, respectively). The four other late summer insecticides (carbofuran, cypermethrin, diflubenzuron, esfenvalerate) were generally rare. Notably, all usage tolerances of carbofuran were revoked in the United States in 2009 (U.S. Environmental Protection Agency 2009), so the four observations at Pope in 2017 represent a prohibited application(s).
Late season fungicides
At least one of the 10 fungicides was detected in 64 of the 142 late season grass samples. Azoxystrobin was the most frequent, in 46 samples. Notably, however, all fungicide-positive samples were collected from 2017 to 2020 (n = 75), while fungicides were not detected in any of the late season samples (n = 67) from 2014 to 2016. As noted above, this temporal variation is likely due to upgrades in extraction and analysis protocols at the NSL. Thus, the fungicide data from the 2017–2020 samples are more likely representative of annual deposition patterns than the 2014–2016 data.
Late season herbicides
Herbicides were rare on grasses in late summer, with generally only Trace detections of atrazine, hexazinone, and metolachor. Most summer herbicide detections were in 2019, which was an extremely wet year that likely prompted additional or later herbicide applications within and adjacent to agricultural fields.
Other compounds
Three compounds were detected for which there are no known local agricultural applications: diphenylamine, thymol, and DEET. Diphenylamine was nearly ubiquitous in Trace or low quantities (1–22 ppb) in 2017 and 2020, but there is no apparent regional agricultural usage, and no diphenylamine was sold in Minnesota in any year for which there is documentation (2018–2020) (Minnesota Department of Agriculture https://www.mda.state.mn.us/agricultural-pesticide-sales-use-reports-statewide/). Thymol was found in a single sample at Pope in 2017. Typically used as a miticide within European honey bee (Aphis mellifera Linneaus: Apidae) hives and as an animal repellent, thymol is also a naturally occurring compound in some native plants (such as Monarda fistulosa Linnaeus: Lamiaceae). DEET was detected in three samples. Applied to clothing or skin as an insect deterrent, this compound was never used by the researchers that collected our samples in the field, so the detections are likely the result of other people who had applied DEET and coincidentally walked through our sampling locations.
Before and after a known spray
Immediately following completion of sampling at Pipestone on August 19, 2014, an airplane was observed spraying a soybean field ~ 350 m northwest of the nearest previously sampled point. Light northwest prevailing winds during and immediately following the late afternoon spray at the nearest weather station (Pipestone, MN; ~19 km SW of the Pipestone site; https://mesonet.agron.iastate.edu/) would be expected to blow aerosolized droplets containing pesticides from the spray in the direction of the Pipestone site and the previously sampled points (Su1-Su7, Appendix 2). Before and after the spray, only the three primary insecticides applied against soybean aphids (chlorpyrifos, bifenthrin, and cyhalothrin) were detected across the Pipestone site. Chlorpyrifos and cyhalothrin quantities always rose after the spray (Su8-Su12, Appendix 2). Mean chlorpyrifos quantities rose from 19.3 ppb (range 9.5–21.1) to 135.7 ppb (range 51.9–278.0). Similarly, mean cyhalothrin quantities rose from 4.0 ppb (range 2.7–7.2) to 24.8 ppb (range 8.4–83.3). In contrast, bifenthrin mean quantities fell from 31.3 ppb (range 8.3–74.5) to 24.8 ppb (range 9.5–68.9) the day after the spray. It therefore seems likely that the aerial spray was a mix of chlorpyrifos and cyhalothrin. The presence of bifenthrin in the samples was likely the result of a previous spray event or events.
Temporal and spatial variation
Insecticides were detected in late season at all sites, but the number of insecticide types varied across sites and years (Fig. 2). Site and Year interacted significantly, largely driven by increases in the number of observed insecticides at Day and Pope between 2014 and 2015 with contrasting concurrent declines at Pipestone and Clay (Appendix 5 A). The number of insecticides observed increased across all sites between 2016 and 2019 and then decreased 2020. Averaged across years, fewer insecticide types were found at Clay than at Pipestone and Day (Fig. 3A). Notably, the last known extant Dakota skipper population in Minnesota is at Clay. Fewer pesticides were detected in 2016 than in 2019, averaged across all sites (Fig. 3B). The number of insecticides detected along prairie-agricultural edges did not differ from the number detected within prairie interior locations, as indicated by a non-significant interaction between Site and nested Location (Table 1).
Similar to the pattern with the number of insecticide types noted above, Site and Year interacted significantly for both chlorpyrifos and cyhalothrin quantities, but in a manner that still allows for some interpretation of main effects (Table 1, Appendix 5B and 5 C). Crop types, pest loads, climatic effects, and associated pesticide application rates are not expected to be uniform within years across the large geographic extent of this study, and overall patterns of average exposure at a site across years are important. In terms of main effects, chlorpyrifos quantities varied significantly across both sites and years when present, while cyhalothrin quantities only varied across years (Fig. 4). Quantities of both compounds did not differ between prairie interiors and agricultural edge locations (as evidenced by non-significant interactions between Site and the nested Location for each compound). It is notable that in a post hoc Tukey contrast, chlorpyrifos quantities were significantly lower at the two sites that retain extant Dakota skipper populations (Day and Clay) than at those where Dakota skippers have been recently extirpated (Pope, Lincoln, and Pipestone).
Discussion
To our knowledge, this study represents the longest time series monitoring of pesticide occurrence across the upland portions of multiple prairie remnants that are of significant conservation interest. Most non-target prairie deposition studies have focused on wetland systems (ex: Donald et al. 1999; Messing et al. 2011; Belden et al. 2012; Main et al. 2015; Mimbs et al. 2016; McMurry et al. 2016; Evelsizer and Skopec 2018). Relatively few terrestrial pesticide occurrence studies have targeted specific compounds or situations (Main et al. 2020; Goebel et al. 2022; Zioga et al. 2023), with particular focus in North America on potential risks to monarchs and contamination of their host milkweeds (Pecenka and Lundgren 2015; Halsch et al. 2020; Krishnan et al. 2020; Hall et al. 2022; Grant et al. 2022).
Contrary to frequently cited concerns, we did not find a significant exposure signal from neonicotinoid insecticides, only detected in the soil of one of our spring samples, and none later in the summer. This does not indicate that neonicotinoids may not pose a seasonal risk or that they are not a risk in other prairie strata, but the exposure potential appears to be lower for the grasses and the soils in the hilly upland gravel prairies that we studied. In contrast, broad-spectrum insecticides, particularly organophosphates (chlorpyrifos) and pyrethroids (primarily cyhalothrin and bifenthrin) likely targeting soybean aphids in the second half of summer, were prevalent in their upland grass larval hosts. The landscape of soybean aphid insecticide applications is changing though due to (1) evolving resistance by soybean aphids to pyrethroids like cyhalothrin and bifenthrin with decreases in control effectiveness noted in Minnesota beginning in 2014 (Hanson et al. 2017; Koch et al. 2018; Menger et al. 2022) and (2) the revocation of all agricultural usage authorizations for chlorpyrifos in the United States in 2022 (U.S. Environmental Protection Agency 2022). This combination of pyrethroid resistance and chlorpyrifos regulation may have at least a short-term effect of reducing insecticide exposure to non-target organisms in these prairies.
Conservation implications
The rapid decline and complete extirpation of Poweshiek skipperling from the vast majority of its historic range across Minnesota, Iowa, North Dakota, and South Dakota was concurrent with changes in pesticide usage and the invasion of the soybean aphid. Sympatric Dakota skipper populations declined, and many also disappeared, shortly thereafter. These extirpation events occurred before this pesticide sampling began, so determining the exact linkage between the pesticides that we have observed, and the butterfly declines is necessarily speculative. However, it is highly noteworthy that the sites where Dakota skipper populations remain extant today had both fewer types of insecticide on average, as well as lower quantities of chlorpyrifos, than other sites where Dakota skippers have been extirpated.
Poweshiek skipperling and Dakota skipper are univoltine with the adult flight in late June and early July. Eggs are laid in July, and 1st through 3rd instar larvae of both species feed through the end of summer until winter hibernation on a wide range of prairie graminoids, including the little bluestem primarily sampled in this study. Thus, both species are still foraging as small larvae on their grass hosts during the late season aerial spraying of broad-spectrum insecticides that we have documented. They resume feeding in spring (when they would theoretically be exposed to early season pesticides) and pupate in June. Poweshiek skipperling and Dakota skipper may be at increased exposure risk relative to regal fritillary which, despite being another historically co-occurring prairie endemic of conservation concern, is still rather common at the same sites that we sampled but whose larvae do not feed until spring and may be thus more buffered from late season insecticide applications. Poweshiek skipperling may be even more vulnerable to environmental stressors, like pesticides exposure, than Dakota skipper and many other grass-feeding skippers because Poweshiek skipperling larvae do not construct shelters and remain higher on their host grass. While correlative, this larval foraging difference may therefore have contributed to the steeper and more dramatic disappearance of the once more common Poweshiek skipperling from the same prairies where Dakota skippers and other prairie butterflies remain.
These retrospective views of potential causes and effects can be cautiously useful, but of greater interest is understanding the current landscape of risk to advance conservation goals. Rare butterflies are not necessarily more likely to persist in protected sites than in unprotected areas (Warren 1993; Schlicht et al. 2009). While the formal U.S. designations of the sites from which we sampled as Critical Habitat for Poweshiek skipperling and Dakota skipper do afford increased oversight protecting key features necessary for their conservation, that oversight only relates to activities that involve a federal permit, license, or funding. Critical Habitat is a tool to guide federal agency actions to fulfill their responsibilities to protect endangered species and the ecological resources upon which they depend, but the activities of private landowners are not affected if there is no federal funding or authorization “nexus”. The results of this study suggest that some degree of non-target pesticide exposure in these Critical Habitats are common, even in their interiors, and should probably be expected in prairie remnants across much of the historic known range of these protected butterflies. Efforts should be made to minimize risks when possible, basing them on the best available evidence to develop protection and recovery plans (e.g., Montgomery et al. 2009).
Unlike more vagile species (Ries and Debinski 2001), Poweshiek skipperling and Dakota skipper are unlikely to colonize significantly dispersed prairie remnants. Lack of connectivity among remnants presents significant risks to low-dispersing prairie dependent species (Leach and Givnish 1996; Attwood et al. 2008; Koper et al. 2010; Nowicki et al. 2014; Wimberly et al. 2018; Crone and Schultz 2019). The U.S. Fish and Wildlife Service identifies that the re-establishment of dozens of “healthy” populations for each species via reintroductions is needed to satisfy downlisting criteria (U.S. Fish and Wildlife Service 2021, 2022), a foundational effort that the Minnesota Zoo began initiating in 2017. A healthy population is formally defined as “one that is demographically, genetically, and physically robust and occupies large areas of high-quality remnant prairie habitat” (U.S. Fish and Wildlife Service 2022). To be “robust”, a population must be “comprised of individuals with good body condition and with pesticide and pathogen loads that are below levels that could cause meaningful loss of reproductive capacity”. Thus, the risks associated with pesticide exposure must therefore be one of the formal factors considered when assessing locations for potential reintroductions (e.g., Delphey et al. 2017). The persistence of these low dispersal species will depend on the maintenance of large, high-quality prairies, for which pesticide load is an important metric.
Remaining questions
Gains have been made in our understanding of the prevalence and occurrence of pesticides in these remnant prairies, but three substantial questions remain about the landscape of risk at both small and large scales. First, how closely do the observed pesticide quantities and composition match their peak values at these sites? The data presented here are likely underestimates the extent of maximum exposures. With the exception of a single date, we do not know the specific timing or locations of the pesticide applications in the vicinity of the sampled prairie remnants (unlike say, Goebel et al. 2022). Pesticides like chlorpyrifos decay under natural conditions but may persist at low levels in the environment weeks after application (Kamrin 1997; Christensen et al. 2009; Das et al. 2020). Pesticide deposition is also expected to decline with distance from application location (Teske et al. 2002; Goebel et al. 2022), but chlorpyrifos and many other pesticides also are known to be transported long distances once volatilized through air (Harnly et al. 2005; Felsot et al. 2011; Giesy et al. 2014; Mackay et al. 2014). Thus, the observed quantities likely represent fractional remnants of the original amounts following initial deposition, and are best thought of as an estimate of the typical distributions of ppb on the landscape in late summer. We also do not fully understand the extent of pesticide composition and occurrence in mid-summer, between our early and late sampling periods. Pesticide applications are probably less likely to occur in other portions of the growing season, particularly during late June and July when Poweshiek skipperling and Dakota skipper are adults, but we cannot be certain that we are not missing an exposure window.
Second, what is the actual risk posed by the observed pesticide composition and their quantities? Risk is the combination of exposure and toxicity. We have documented widespread exposure to many pesticides, but whether or not their observed quantities generate mortality and/or sub-lethal effects to skippers and other prairie insects is central. There are few suitable studies with surrogate species for non-model systems (like the imperiled grass-feeding skippers) against which we can make useful comparisons on the effects of pesticides. What does exist often varies in method, exposure route (oral, contact, etc.), and/or metrics (ng/bee, kg/ha, or % of active ingredient, etc.) (US Environmental Protection Agency 1999). For chlorpyrifos, Krishnan et al. (2020) report laboratory contact LD50 toxicity of 79 µg/g (= 79,000 ppb) for first instar monarch larvae (Danaus plexippus Linneaus: Nymphalidae), and a dietary LD50 of 9.9–33 µg/g (= 9,900–33,000 ppb) of leaf material. These values are well above those observed in this study, but Krishnan et al. (2020) still observed significant mortality in companion standard field spray assays following similar aerial application protocols to what would be expected adjacent to the prairie remnants in this study. The reported contact LD50 for chlorpyrifos for honey bee is much lower though (0.059 µg/bee = 59 ppb; Arena and Sgolastra 2014), a value that was exceeded in seven samples in this dataset. Similarly, a reported honey bee contact LD50 for bifenthrin (0.01462 µg/bee = 14.6 ppb; Tomlin 2000) was exceeded 13 times in our dataset.
Experiments testing responses to a wide range of the pesticides observed are needed across multiple related species, particularly given the imperiled state of Dakota skipper and Poweshiek skipperling. Multiple forms of exposure studies, ranging from direct contact studies under artificial conditions that remove as many variables as possible to studies that capture as much natural history as possible, would provide useful inferences of the “real world” risks. Pesticides exposure studies must test both lethal and non-lethal responses to individual pesticides but also responses to the synergistic effects of exposure to multiple pesticides. It cannot be expected that each of the nearly two dozen pesticides observed in this study will individually affect prairie skippers, arthropods, and other wildlife in a vacuum. For example, Hladik et al. (2016) recorded nineteen pesticides and degradates on wild native bees. Indeed, compounding and non-additive effects of simultaneous exposures to multiple pesticides should be expected at both individual species and species assemblage levels (Barmentlo et al. 2018). As noted by Schulz et al. (2021), the total applied toxicity of pesticides to pollinators has risen in recent decades, a feature largely driven by increases in the toxicity of pesticide classes like pyrethroids and neonicotinoids and not necessarily the total mass of pesticides applied.
Finally, what landscape factors are driving this non-target deposition, and are these sampled prairies representative of others? How well do adjacent land-use patterns predict pesticide composition and quantities into prairie remnants? Are these sites unusual? A deeper exploration of site-specific and regional influences, including crop rotations, pest cycles, weather, topography, and other factors that shape deposition is warranted, within and across both years and sites. A more exhaustive regional inventory of pesticide exposures at additional prairie remnants coupled with predictive analyses of the factors that drive non-target exposure would be beneficial for conservation and risk minimization efforts, for Poweshiek skipperling, for Dakota skipper, and beyond.
References
Anastassiades M, Lehotay SJ, Štajnbaher D, Schenck FJ (2003) Fast and easy Multiresidue Method employing Acetonitrile Extraction/Partitioning and dispersive solid-phase extraction for the determination of pesticide residues in produce. J AOAC Int 86:412–431
Arena M, Sgolastra F (2014) A meta-analysis comparing the sensitivity of bees to pesticides. Ecotoxicol 24:324–334
Attwood SJ, Maron M, House APN, Zammit C (2008) Do arthropod assemblages display globally consistent responses to intensified agricultural land use and management? Glob Ecol Biogeogr 17:585–599
Barmentlo SH, Schrama M, Hunting ER et al (2018) Assessing combined impacts of agrochemicals: aquatic macroinvertebrate population responses in outdoor mesocosms. Sci Total Environ 631–632:341–347
Basley K, Goulson D (2018) Effects of Field-relevant concentrations of Clothianidin on Larval Development of the Butterfly Polyommatus Icarus (Lepidoptera, Lycaenidae). Environ Sci Technol 52:3990–3996
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting Linear mixed-effects models using lme4. J Stat Soft 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Belden JB, Hanson BR, McMurry ST et al (2012) Assessment of the effects of farming and conservation programs on pesticide deposition in high plains wetlands. Environ Sci Technol 46:3424–3432
Belitz MW, Hendrick LK, Monfils MJ et al (2018) Aggregated occurrence records of the federally endangered Poweshiek Skipperling (Oarisma poweshiek). Biodivers Data J e29081
Bonmatin J-M, Giorio C, Girolami V et al (2015) Environmental fate and exposure; neonicotinoids and fipronil. Environ Sci Pollut Res Int 22:35–67
Christensen K, Harper B, Luukinen B et al (2009) Chlorpyrifos Technical Fact sheet. National Pesticide Information Center. Retrieved 2014-07-03
COSEWIC (2014b) COSEWIC assessment and status report on the Dakota Skipper Hesperia dacotae in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. x + 61 pp
COSEWIC (2014a) COSEWIC assessment and status report on the Poweshiek Skipperling Oarisma poweshiek in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. xi + 43 pp
Crone EE, Schultz CB (2019) In: Butterflies (ed) CHAPTER 25. Movement Behavior and Minimum Patch size for Butterfly Population Persistence. University of Chicago Press, pp 561–602
Dana RP (1991) Conservation management of the prairie skippers Hesperia dacotae and Hesperia ottoe. Basic biology and threat of mortality during prescribed spring burning. Minnesota Agricultural Experiment Station. University of Minnesota
Das S, Hageman KJ, Taylor M et al (2020) Fate of the organophosphate insecticide, chlorpyrifos, in leaves, soil, and air following application. Chemosphere 243:125194
Delphey P, Runquist E, Nordmeyer C (2017) Plan for the Controlled Propagation, Augmentation, and Reintroduction of Dakota skipper (Hesperia dacotae)
Didham RK, Basset Y, Collins CM et al (2020) Interpreting insect declines: seven challenges and a way forward. Insect Conserv Divers 13:103–114
Donald DB, Syrgiannis J, Hunter F, Weiss G (1999) Agricultural pesticides threaten the ecological integrity of northern prairie wetlands. Sci Total Environ 231:173–181
Evelsizer V, Skopec M (2018) Pesticides, including neonicotinoids, in drained wetlands of Iowa’s prairie pothole region. Wetlands 38:221–232
Fairbrother A, Purdy J, Anderson T, Fell R (2014) Risks of neonicotinoid insecticides to honeybees. Environ Toxicol Chem 33:719–731
Felsot AS, Unsworth JB, Linders JBHJ et al (2011) Agrochemical spray drift; assessment and mitigation–a review. J Environ Sci Health B 46:1–23
Foreman WT, Majewski MS, Goolsby DA et al (2000) Pesticides in the atmosphere of the Mississippi River Valley, part II–air. Sci Total Environ 248:213–226
Forister ML, Cousens B, Harrison JG et al (2016) Increasing neonicotinoid use and the declining butterfly fauna of lowland California. Biol Lett 12:20160475
Gibbons D, Morrissey C, Mineau P (2015) A review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environ Sci Pollut Res Int 22:103–118
Gibbs KE, Mackey RL, Currie DJ (2009) Human land use, agriculture, pesticides and losses of imperiled species. Divers Distrib 15:242–253
Giesy JP, Solomon KR, Mackay D, Anderson J (2014) Evaluation of evidence that the organophosphorus insecticide chlorpyrifos is a potential persistent organic pollutant (POP) or persistent, bioaccumulative, and toxic (PBT). Environ Sci Europe 26:29
Gilburn AS, Bunnefeld N, Wilson JM et al (2015) Are neonicotinoid insecticides driving declines of widespread butterflies? PeerJ 3:e1402
Goebel KM, Davros NM, Andersen DE, Rice PJ (2022) Tallgrass Prairie wildlife exposure to spray drift from commonly used soybean insecticides in Midwestern USA. Sci Total Environ 818:151745
Goulson D (2013) Review: an overview of the environmental risks posed by neonicotinoid insecticides. J Appl Ecol 50:977–987
Grant TJ, Fisher KE, Krishnan N et al (2022) Monarch Butterfly Ecology, Behavior, and Vulnerabilities in North Central United States Agricultural Landscapes. Bioscience biac094
Habel JC, Ulrich W, Biburger N et al (2019) Agricultural intensification drives butterfly decline. Insect Conserv Divers. https://doi.org/10.1111/icad.12343
Hall MJ, Zhang G, O’Neal ME et al (2022) Quantifying neonicotinoid insecticide residues in milkweed and other forbs sampled from prairie strips established in maize and soybean fields. Agric Ecosyst Environ 325:107723
Halsch CA, Code A, Hoyle SM et al (2020) Pesticide contamination of Milkweeds across the Agricultural, Urban, and open spaces of Low-Elevation Northern California. Front Ecol Evol 8. https://doi.org/10.3389/fevo.2020.00162
Hanson AA, Menger-Anderson J, Silverstein C et al (2017) Evidence for soybean aphid (Hemiptera: Aphididae) Resistance to Pyrethroid insecticides in the Upper Midwestern United States. J Econ Entomol 110:2235–2246
Harnly M, McLaughlin R, Bradman A et al (2005) Correlating agricultural use of organophosphates with outdoor air concentrations: a particular concern for children. Environ Health Perspect 113:1184–1189
Henault J, Westwood R (2022) Endangered Oarisma poweshiek larvae vary their graminoid forage in Manitoba, Canada. Can Entomol 154:e49
Hladik ML, Kolpin DW, Kuivila KM (2014) Widespread occurrence of neonicotinoid insecticides in streams in a high corn and soybean producing region, USA. Environ Pollut 193:189–196
Hladik ML, Vandever M, Smalling KL (2016) Exposure of native bees foraging in an agricultural landscape to current-use pesticides. Sci Total Environ 542:469–477
Jones A, Harrington P, Turnbull G (2014) Neonicotinoid concentrations in arable soils after seed treatment applications in preceding years. Pest Manag Sci 70:1780–1784
Kamrin MA (1997) Pesticide profiles toxicity, Environmental Impact, and Fate. Lewis, Boca Raton, FL, pp 147–152
Koch RL, Hodgson EW, Knodel JJ et al (2018) Management of insecticide-resistant soybean aphids in the Upper Midwest of the United States. J Integr Pest Manag 9. https://doi.org/10.1093/jipm/pmy014
Koper N, Mozel KE, Henderson DC (2010) Recent declines in northern tall-grass prairies and effects of patch structure on community persistence. Biol Conserv 143:220–229
Krishnan N, Zhang Y, Bidne KG et al (2020) Assessing field-scale risks of Foliar Insecticide Applications to Monarch Butterfly (Danaus plexippus) Larvae. Environ Toxicol Chem 39:923–941
Lark TJ, Larson B, Schelly I et al (2019) Accelerated Conversion of native Prairie to Cropland in Minnesota. Environ Conserv 46:155–162
Leach MK, Givnish TJ (1996) Ecological determinants of species loss in Remnant prairies. Science 273:1555–1558
Mackay D, Giesy JP, Solomon KR (2014) Fate in the environment and long-range atmospheric transport of the organophosphorus insecticide, chlorpyrifos and its oxon. Rev Environ Contam Toxicol 231:35–76
Main AR, Headley JV, Peru KM et al (2014) Widespread use and frequent detection of neonicotinoid insecticides in wetlands of Canada’s Prairie Pothole Region. PLoS ONE 9:e92821
Main AR, Michel NL, Headley JV et al (2015) Ecological and Landscape drivers of Neonicotinoid Insecticide detections and concentrations in Canada’s Prairie wetlands. Environ Sci Technol 49:8367–8376
Main AR, Hladik ML, Webb EB et al (2020) Beyond neonicotinoids - wild pollinators are exposed to a range of pesticides while foraging in agroecosystems. Sci Total Environ 742:140436
Mason R, Tennekes H, Sanchez-Bayo F, Uhd Jepsen P (2013) Immune suppression by neonicotinoid insecticides at the root of global wildlife declines. J Environ Immunol Toxicol 1:3
McMurry ST, Belden JB, Smith LM et al (2016) Land use effects on pesticides in sediments of prairie pothole wetlands in North and South Dakota. Sci Total Environ 565:682–689
Menger JP, Ribeiro AV, Potter BD, Koch RL (2022) Change-point analysis of lambda-cyhalothrin efficacy against soybean aphid (Aphis Glycines Matsumura): identifying practical resistance from field efficacy trials. Pest Manag Sci 78:3638–3643
Messing PG, Farenhorst A, Waite DT et al (2011) Predicting wetland contamination from atmospheric deposition measurements of pesticides in the Canadian Prairie Pothole region. Atmos Environ 45:7227–7234
Mimbs WHIV, Cusaac JPW, Smith LM et al (2016) Occurrence of current-use fungicides and bifenthrin in Rainwater Basin wetlands. Chemosphere 159:275–281
Mineau P, Downes CM, Kirk DA et al (2005) Patterns of bird species abundance in relation to granular insecticide use in the Canadian prairies. Écoscience 12:267–278
Montgomery RA, Rubeck-Schurtz CN, Millenbah KF et al (2009) Modeling protected species habitat and assigning risk to inform regulatory decisions. Environ Manage 44:12–23
Nordmeyer CS, Runquist E, Stapleton S (2021) Invasive grass negatively affects growth and survival of an imperiled butterfly. Endanger Species Res 45:301–314
Nowicki P, Vrabec V, Binzenhöfer B et al (2014) Butterfly dispersal in inhospitable matrix: rare, risky, but long-distance. Landsc Ecol 29:401–412
Olaya-Arenas P, Kaplan I (2019) Quantifying Pesticide exposure risk for Monarch caterpillars on milkweeds Bordering Agricultural Land. Front Ecol Evol 7:223
Pecenka JR, Lundgren JG (2015) Non-target effects of clothianidin on monarch butterflies. Naturwissenschaften 102:19
R Core Team (2023) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ragsdale DW, Landis DA, Brodeur J et al (2011) Ecology and management of the soybean aphid in North America. Annu Rev Entomol 56:375–399
Raven PH, Wagner DL (2021) Agricultural intensification and climate change are rapidly decreasing insect biodiversity. Proceedings of the National Academy of Sciences 118:e2002548117
Ricketts TH, Dinerstein E, Olson DM et al (1999) Terrestrial ecoregions of North America: A Conservation Assessment. Island
Ries L, Debinski DM (2001) Butterfly responses to habitat edges in the highly fragmented prairies of Central Iowa. J Anim Ecol 70:840–852
Rigney CL (2013) Habitat characterization and biology of the threatened Dakota skipper (Hesperia dacotae) in Manitoba. Masters of Science, University of Winnipeg
Royer E (2019) Hesperia dacotae The IUCN Red List of Threatened Species. The IUCN Red List of Threatened Species 2019: e.T9968A122963341. https://dx.doi.org/10.2305/IUCN.UK.2019-2.RLTS.T9968A122963341.en. Accessed on 19 April 2023
Royer E (2020) Oarisma Powesheik (amended version of 2019 assessment). IUCN Red List Threatened Species 2020(eT122914337A166163683). https://doi.org/10.2305/IUCN.UK.2020-1.RLTS.T122914337A166163683.en. Accessed on 19 April 2023
Royer R, Marrone GM (1992a) Conservation status of the Dakota Skipper (Hesperia Dacotae) in North and South Dakota: a report to the United States Department of the Interior Fish and Wildlife Service. Minot State University, Denver, Colorado
Royer R, Marrone GM (1992b) Conservation status of the Powesheik Skipper (Oarisma Powesheik) in North and South Dakota: a report to the United States Department of the Interior Fish and Wildlife Service. Minot State University, Denver, Colorado
Royer R, McKenney R, Newton W (2008) A characterization of non-biotic environmental features of prairies hosting the Dakota skipper (Hesperia dacotae, Hesperiidae) across its remaining US range. J Lepidopterist’s Soc 62:1
Samson FB, Knopf FL (1996) Prairie Conservation: preserving North America’s most endangered ecosystem. Island
Sánchez-Bayo F, Wyckhuys KAG (2019) Worldwide decline of the entomofauna: a review of its drivers. Biol Conserv 232:8–27
Sánchez-Bayo F, Wyckhuys KAG (2021) Further evidence for a global decline of the entomofauna. Aust Entomol 60:9–26
Schlicht D, Swengel A, Swengel S (2009) Meta-analysis of survey data to assess trends of prairie butterflies in Minnesota, USA during 1979–2005. J Insect Conserv 13:429–447
Schulz R, Bub S, Petschick LL et al (2021) Applied pesticide toxicity shifts toward plants and invertebrates, even in GM crops. Science 372:81–84
Seidle KM, Lamb EG, Bedard-Haughn A, DeVink J-M (2018) Environmental associations of Hesperia dacotae (Lepidoptera: Hesperiidae) in southeastern Saskatchewan, Canada. Can Entomol 150:652–662
Swengel AB, Swengel SR (2015) Grass-skipper (Hesperiinae) trends in midwestern USA grasslands during 1988–2013. J Insect Conserv 19:279–292
Swengel SR, Schlicht D, Olsen F, Swengel AB (2011) Declines of prairie butterflies in the Midwestern USA. J Insect Conserv 15:327–339
Tapparo A, Marton D, Giorio C et al (2012) Assessment of the environmental exposure of honeybees to particulate matter containing neonicotinoid insecticides coming from corn coated seeds. Environ Sci Technol 46:2592–2599
Teske ME, Bird SL, Esterly DM et al (2002) AgDrift: a model for estimating near-field spray drift from aerial applications. Environ Toxicol Chem 21:659–671
Tomlin CDS (ed) (2000) The Pesticide Manual, 12th edition British Crop Protection Coun- cil, Farnham, Surrey,U.K
U.S. Environmental Protection Agency (1999) Reregistration Eligibility Science Chapter for Chlorpyrifos Fate and Environmental Risk Assessment Chapter, Office of Prevention, Pesticides and Toxic Substances, Office of Pesticide Programs, Environmental Fate and Effects Division, U.S. Government Printing Office: Washington, DC, 1999
U.S. Environmental Protection Agency (2022) Tolerance Revocations: Chlorpyrifos. Docket EPA-HQ-OPP-2021-0523-0001. 40 CFR Part 180. 48315–48336
U.S. Environmental Protection Agency (2009) Tolerance Revocations: Carbofuran. Docket EPA-HQ-OPP-2005-0162. 40 CFR Part 180. 23046–23094
U.S. Fish and Wildlife Service (2014) Endangered and threatened Wildlife and plants; threatened species Status for Dakota skipper and endangered species Status for Poweshiek Skipperling. Docket FWS-R3-2013-0043-4500030113. 50 CFR Part 17. 63672 – 63748
U.S. Fish and Wildlife Service (2015) Endangered and threatened Wildlife and plants; designation of critical Habitat for the Dakota skipper and poweshiek skipperling. Docket FWS-R3-2013-0043-4500030113. 50 CFR Part 17. 59248–59384
U.S. Fish and Wildlife Service (2021) Recovery Plan for Dakota skipper (Hesperia dacotae). September 2021. U.S. Fish and Wildlife Service, Great Lakes Region. Bloomington. Minnesota. 13 pages
U.S. Fish and Wildlife Service (2022) Recovery Plan for the Poweshiek Skipperling (Oarisma poweshiek). Midwest Regional Office, Bloomington, MN
Wagner DL (2020) Insect declines in the Anthropocene. Annu Rev Entomol 65:457–480
Warren MS (1993) A review of butterfly conservation in central southern Britain: I. Protection, evaluation and extinction on prime sites. Biol Conserv 64:25–35
Wepprich T, Adrion JR, Ries L et al (2019) Butterfly abundance declines over 20 years of. systematic monitoring in Ohio, USA
Williams N, Sweetman J (2019) Distribution and concentration of Neonicotinoid Insecticides on Waterfowl Production areas in West Central Minnesota. Wetlands 39:311–319
Wimberly MC, Narem DM, Bauman PJ et al (2018) Grassland connectivity in fragmented agricultural landscapes of the north-central United States. Biol Conserv 217:121–130
Zioga E, White B, Stout JC (2023) Pesticide mixtures detected in crop and non-target wild plant pollen and nectar. Sci Total Environ 879:162971
Acknowledgements
Sites were selected for analysis in consultation with the U.S. Fish and Wildlife Service, Minnesota Department of Natural Resources, and others. Collections occurred under permits and permissions from Minnesota State Parks and Trails, Minnesota Scientific and Natural Areas, The Nature Conservancy, and the Sisseton Wahpeton Oyate of the Lake Traverse Reservation. Funding was provided the Environment and Natural Resources Trust Fund as recommended by the Minnesota Legislative-Citizen Commission on Minnesota Resources (M.L. 2016, Chp. 186, Art. 2, Sect. 2, Subd. 03c1; M.L. 2019, First Special Session, Chp. 4, Art. 2, Sect. 2, Subd. 08a), the US Fish and Wildlife Service CFDA Program (Cooperative Agreement F15AC00020), and the Minnesota Zoo Foundation. Statistics were calculated in RStudio. Graphics were generated in RStudio and Affinity Designer. R. Briscoe Runquist provided statistical assistance.
Author information
Authors and Affiliations
Contributions
E.R. and C.N. conceived of the study and collected field samples. E.R. wrote the manuscript text, conducted statistical analyses, and prepared figures and tables. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors do not have competing interests or funding to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1:
Appendix 1: levels of detection for all 233 analyzed compounds across all years
Supplementary Material 2:
Appendix 2: Pesticides observed in late season grasses
Supplementary Material 3:
Appendix 3: Pesticides observed in early season grasses
Supplementary Material 4:
Appendix 4: Pesticides observed in soil samples
Supplementary Material 5:
Appendix 5: Site x Year interactions for the (A) mean number of insecticides observed, (B) log mean ppb chlorpyrifos observed, and (C) log mean ppb cyhalothrin observed
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Runquist, E., Nordmeyer, C. & Stapleton, S. Widespread annual occurrence of pesticides within designated critical habitats for endangered prairie butterflies. J Insect Conserv 28, 539–552 (2024). https://doi.org/10.1007/s10841-024-00572-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-024-00572-5