Abstract
Agricultural intensification has led to structurally simplified landscapes with reduced and fragmented resources for farmland insects. However, studies on the effects of landscape composition on farmland insects have mainly been performed in areas dominated by open arable land and semi-natural grasslands, while studies from forest-dominated landscapes are scarce. This research examined the effects of landscape composition on hoverfly species richness and abundance in arable land in boreal forest-dominated landscapes. Hoverflies were sampled in 22 mass-flowering caraway (Carum carvi) fields in Central Finland using pan traps. The effects of landscape composition on species richness and abundance were examined for all hoverflies, and for species groups with different adult habitat preferences. Landscape composition was measured as proportions of land cover classes within two different radii. Species richness and abundances of all hoverflies, forest species and open-habitat species increased with decreasing arable land cover and/or increasing forest cover within a 500 m radius (the two land cover classes strongly negatively correlated). Wetland species were most abundant in landscapes with an intermediate cover of arable land and forest, and most species-rich in landscapes with intermediate (10%) water cover. The species richness and abundance of mixed-habitat species increased with increasing cover of transitional woodland.
Implications for insect conservation
Our results show that most hoverfies in arable land benefit from increasing surrounding forest cover even in relatively heterogeneous, forest-dominated landscapes. Preserving or increasing the area of forests and other non-arable habitats is needed to safeguard a diversity of resources for hoverflies, and associated ecosystem services in farmland.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent studies have shown alarming declines of insects in intensively used landscapes (Hallmann et al. 2017; Powney et al. 2019; Seibold et al. 2019). A key driver of the insect loss is intensified agriculture, which has led to expanded total agricultural area, reduced areas of natural and semi-natural habitats, increased field sizes, monocultures, and increased pesticide use (Stoate et al. 2009; Potts et al. 2016). The diversity loss weakens ecosystem services which agriculture depends on, threatens food production, and causes economic losses to farmers (Zhang et al. 2007; Kremen and Miles 2012; Dainese et al. 2019).
An important factor affecting insect diversity is landscape composition, i.e. the amount or proportion of different habitat types in the landscape (Stein et al. 2014; Turner and Gardner 2015). A structurally simple landscape has a dominant habitat type, which decreases the amount of other habitat types and their associated resources. For example, an increase in the area of intensively cultivated arable land decreases the amount of other habitat types in the landscape, and thus, decreases the diversity of insects (Senapathi et al. 2015; Kovács-Hostyánszki et al. 2017). The habitat loss affects species differently: specialized species with low mobility are more vulnerable than generalist and highly mobile species (Schweiger et al. 2007; Öckinger et al. 2010; Potts et al. 2010; Gaytán et al. 2020).
Besides habitat diversity, a sufficiently large area of a key habitat can be important in maintaining species diversity, and the diversity of habitat specialists in particular, due to heterogeneous resource availability within the habitat (Davidar et al. 2001; Botham et al. 2015). In agricultural landscapes, semi-natural habitats such as semi-natural grasslands and forests enhance insect diversity by providing food, and breeding and overwintering sites (Billeter et al. 2008; Sjödin et al. 2008; Meyer et al. 2009; Kovács-Hostyánszki et al. 2017; Toivonen et al. 2017; Schirmel et al. 2018). Forests also affect insects’ dispersal, since they can enhance flying conditions by reducing winds, provide dispersal routes along forest edges, or inhibit dispersal by acting as barriers (Lewis et al. 1969; Klaus et al. 2015; Öckinger et al. 2012).
Hoverflies (Syrphidae) are an important part of wild pollinator communities but remain relatively poorly known compared to bees (Doyle et al. 2020; Dunn et al. 2020). In recent years, hoverflies have been increasingly recognized as important crop pollinators (Rader et al. 2016; Dunn et al. 2020; Doyle et al. 2020; Toivonen et al. 2022). Besides pollination, hoverflies contribute to biological pest control and the decomposition of organic matter (Rotheray and Gilbert 2011; Dunn et al. 2020). Adult hoverflies mainly feed on nectar and pollen. Hoverfly larvae have diverse diets: Some species are predators, eating aphids and other soft-bodied insects, and acting as natural enemies of crop pests (Hogg et al. 2011; Raymond et al. 2014; Dunn et al. 2020). The other species are saprophagous or phytophagous as larvae. Saprophagous larvae feed on decaying organic matter, and live, for example, on deadwood, underwater on shores and in ditches, and within soil and litter. Phytophagous larvae live in plant stems and roots, but they are not substantial crop pests (Rotheray and Gilbert 2011). Due to the various habitat requirements of hoverflies, their diversity may strongly respond to land use changes.
Previous studies have shown that forests play a key role in supporting hoverfly diversity and abundance in agricultural landscapes (Sjödin et al. 2008; Meyer et al. 2009; Schirmel et al. 2018; Proesmans et al. 2019; Toivonen et al. 2022). Forests and forest edges may provide hoverflies wind-sheltered habitats (Lewis 1969) with critical larval and/or adult resources (Ricarte et al. 2011; Moquet et al. 2018; Proesmans et al. 2019; Gaytán et al. 2020). However, forest matrix may also inhibit hoverfly dispersal (Öckinger et al. 2012). The responses to forest cover differ between hoverfly species and groups (Moquet et al. 2018; Schirmel et al. 2018; Proesmans et al. 2019; Toivonen et al. 2022). However, studies on the effects of land use on hoverflies have mainly been performed in temperate and Mediterranean regions (Doyle et al. 2020), and in areas with low forest cover, where the importance of remaining forest paches is emphasized (but see Öckinger et al. 2012; Toivonen et al. 2022). So far, no study has examined how hoverfly diversity in arable land responds to landscape composition in forest-dominated agricultural landscapes.
The aim of this study was to examine the effects of landscape composition on hoverfly species richness and abundance in forest-dominated boreal agricultural landscapes. Besides total species richness and abundance, the responses of hoverfly species groups with different adult habitat preferences were separately examined. Hoverflies were collected in mass-flowering caraway (Carum carvi L., Apiaceae) fields. We expected that the total species richness and abundance of hoverflies respond positively to increasing forest cover and decreasing arable land cover, and that the hoverfly groups differ in their responses to landscape composition, reflecting the differences in their habitat preferences.
Methods
Study sites and landscape composition
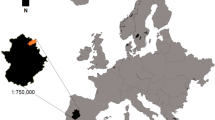
Hoverflies were collected in 22 caraway fields in Central Finland (Fig. 1A) within the coordinates of lat 61° 56′–62° 31′ and lon 25° 18′–26° 32′ (ETRS89 ~ WGS84). The study area is characterized by boreal farmland-forest mosaic landscapes where land use is dominated by forests (mainly coniferous and mixed), followed by arable land. Caraway is a common speciality crop in the area. As an umbelliferous herb, caraway has flowers with exposed nectaries that are easily accessible to various types of insects (Koul et al. 1993). Based on previous studies, caraway can be highly attractive to Diptera (Ricciardelli D’Albore 1986; Toivonen et al. 2022). The area of the study fields ranged from 1 to 9 ha. The fields had a minimum distance of 2 km between each other (and thus non-overlapping landscapes at the radius of 1 km), except for two pairs of fields, with fields situated at the distances of 750 m and 1.8 km from each other within the pair.
Landscape composition was analyzed within 500 m and 1000 m radii around the central points of the caraway fields. These radii match typical flying distances of hoverflies (Wratten et al. 2003; Rotheray et al. 2009; Rotheray and Gilbert 2011). Landscape composition was measured as the percentage cover of land cover types using ArcGIS 10.3.1 (ESRI 2014) and CORINE raster Land Cover data from 2012, with pixel size of 20 m × 20 m (Finnish Environment Institute 2012). The detailed categories of land cover types were combined into six broad land cover classes (Table 1). The four most common land cover classes—forest, arable land, transitional woodland, and water—were selected as explanatory variables, whereas two other land cover classes—articifial surfaces and wetlands—were excluded from the analyses due to their low covers. Especially wetland cover would have been an interesting explanatory variable, but 68% of the study fields had no or less than 1% wetland cover within the 500 m radius.
Hoverfly trapping and data
Hoverflies were collected in the caraway fields using white, blue and yellow pan traps to ensure efficient trapping of species attracted by different colors (Zou et al. 2012) (Online Resource Fig. 1). A total of 16 pan traps per field were placed in four groups of four traps (Fig. 1B). Each group included two white traps, one blue trap and one yellow trap. The high number of white traps was used, firstly, to compensate their poor visibility in white flowering caraway fields, which may reduce their effectiveness, and secondly, to ensure efficient trapping of hoverflies specialized in visiting caraway flowers (Goulson and Wright 1998). The distance between the adjacent traps within a group was 3 m, and the distance of the traps to the field edge was 0.5 m. Pan traps were plastic bowls (375 ml, Ø 18 cm), each of which was glued on top of a 120 cm long wooden stick. The stick was cut so that the pan trap was at the same height with the caraway flowers. The traps were filled with water, and a few drops of dishwashing detergent were added to break surface tension. Two small holes in the upper edge of the bowl prevented flooding during rainfall.
The traps were placed in the fields between the 3rd and 8th of June 2016, when caraway was starting to bloom. Before the start of the trapping, the farmers had sprayed the fields with pesticides to control Depressaria daucella moth in caraway, except for four farmers, who did not apply chemical pest control. No pesticides were used in the fields during the trapping. Insects were collected from the traps three times, always in the same order. Each trapping period lasted 6 days, the last trapping day being the 26th of June. The collected insects were preserved in ethanol. During the trapping season, caraway blooming proceeded: At the beginning of the trapping, the first flowers were open. At the end of the trapping, all flowers had opened, and the first flowers had withered.
The collected hoverflies were identified to species level, except for individuals of the genus Melanostoma and females of Sphaerophoria. Since we could not reliably identify species of these two hoverfly groups based on morphological characteristics, they were identified to genus level and treated as species in the analyses. The identification keys and nomenclature of Haarto and Kerppola (2007), and Bartsch et al. (2009a, 2009b) were used.
Additionally, to examine the differences in hoverfly species’ responses to landscape composition, hoverflies were classified into four groups based on their adult habitat preferences according to Speight (2011) (Online Resource Table 1). The groups were forest species, open-habitat species (i.e. anthropophilic, open habitat, and combination of open habitat and some other habitat sensu Speight (2011)), wetland species and mixed-habitat species. Open-habitat species can be considered as generalists because they use various resources and occupy many kinds of habitats, although they are usually found in open environments, such as fields, road verges and meadows (Branquart and Hemptinne 2000). The other three groups are more specialized to particular environments. Forest species prefer deciduous and coniferous forests, while wetland species live in aquatic environments, such as in swamps and on the edges of water bodies. The habitat preference of the mixed-habitat species is a combination of forest and wetland. Analyses on the effects of landscape composition on species richness and abundance were conducted separately for these four groups.
Hoverflies were also grouped into four larval feeding types according to Speight (2011): aphidophagous (includes zoophagous), phytophagous, saprophagous and sapro-xylophagous. Since the larval feeding types are largely associated with the adult habitat preferences, statistical analyses were not performed for the larval feeding types, but they were used to help the interpretation of the results.
Statistical analyses
Before the analyses, the hoverfly observations of the 48 catches (16 traps and three trapping periods) per field were pooled to obtain the total abundance and species richness of all hoverflies, forest species, open-habitat species, wetland species and mixed-habitat species per field. The pooling was performed because the interest was on the pollinator community of a given field, and not on the variation within a field.
The effects of landscape composition on hoverfly abundance and species richness were analyzed using regression models and an information theoretic approach. The analyses were conducted separately for the abundance and species richness of all hoverflies, forest species, open-habitat species, wetland species and mixed-habitat species, and with the landscape buffers of 500 m and 1 km radii. The four explanatory variables—arable land, forest, transitional woodland and water cover—were intercorrelated (Online Resource Table 2). Thus, four competing models were constructed by including one of the explanatory variables at a time. For species richness, generalized linear models with Poisson distribution were fitted using the function glm() of the R package stats (R Core Team 2018). For abundance, negative binomial models were fitted, due to overdispersion, using the function glm.nb() of the package MASS (Venables and Ripley 2002). For wetland species, the preliminary data inspection showed that the abundance and species richness respond to some of the explanatory variables in a hump-shaped manner. Thus, for wetland species, a total of eight candidate models were constructed: besides the four linear models, four quadratic models were fitted by including each explanatory variable and its squared term.
Akaike Information Criterion for small sample sizes (AICc) was used to identify the best models among the candidate models with different explanatory variables. Models with ∆AICc < 2 were considered equally good (Burnham and Andersson 2002). Akaike weights (wi), which indicate the probability that a particular model is the best among those considered, were also calculated for the models. The function aictab() of the package AICcmodavg (Mazerolle 2019) was applied for the AICc model selection.
The models were checked for spatial autocorrelation in residuals using Moran’s I test with the function moran.test() of the R package spdep (Bivand and Wong 2018). No spatial autocorrelation was found. One of the study fields had an exceptionally high total hoverfly abundance, as well as an exceptionally high abundance of forest species and open-habitat species (Fig. 3). Excluding the field did not qualitatively change the results, and thus, the field was included in the final analyses. All analyses were conducted with R software 3.5.0 (R Core Team 2018).
Results
A total of 2829 hovefly individuals representing 105 species were caught in the pan traps (Online Resource Table 1). One red-listed species, Temnostoma angustistriatum (NT), was caught (Hyvärinen et al. 2019). The mean species richness per field was 30 (SD = 6.9, min = 16, max = 41), and the mean abundance 129 (SD = 75.2, min = 34, max = 389).
Of the four hoverfly groups with different adult habitat preferences, open-habitat species were most abundant, whereas forest species had highest species richness (Fig. 2A–B). Wetland and mixed-habitat species were in minority (Fig. 2A–B). Three most abundant species, Xylota segnis (open-habitat species), X. ignava (forest species) and X. jakutorum (forest species), constituted 31% of all individuals. Because of the high abundance of the few Xylota species, the forest species group had a high abundance of species with sapro-xylophagous larvae, but a relatively low sapro-xylophagous species richness (Fig. 2C–D). The majority of the forest species were aphidophagous as larvae (Fig. 2D). The group of open-habitat species had both a high abundance and species richness of species with aphidophagous larvae (Fig. 2C–D). Wetland and mixed-habitat species were mostly saprophagous as larvae (Fig. 2C–D).
Total abundance (A) and species richness (B) of hoverfly groups with different adult habitat preferences in caraway fields in Finland, and the proportions of larval feeding types in the hoverfly groups, based on abundance (C) and species richness (D). a = aphidophagous, p = phytophagous, s = saprophagous, and sx = sapro-xylophagous
Hoverflies showed, on average, stronger responses to landscape composition in the 500 m than in the 1000 m radius. Thus, focus is hereafter on landscapes within the shorter radius. The results of the effects of landscape composition within the 1000 m radius are shown in the Supplementary Information (Online Resource Table 3).
The species richness and abundance of all hovefly species, open-habitat species and forest species were best predicted by arable land cover and/or forest cover (Table 2), which were strongly negatively correlated (Pearson’s r − 0.85 within the 500 m radius, Online Resource Table 2). Arable land cover and forest cover were equally good predictors (∆AICc < 2) for the species richness of all hoveflies, open-habitat species and forest species, and for the abundance of open-habitat species, while arable land cover was slightly better preditor than forest cover for the total abundance and the abundance of forest species (Table 2). The species richness and abundances decreased with increasing arable land cover and decreasing forest cover (Fig. 3, Online Resource Table 4). The decrease was steepest for forest species, which declined 89% in abundance and 56% in species richness, when arable land cover increased from 10 to 60% in the landscape (Fig. 3B).
Abundance and species richness of all hoverflies (A), forest species (B) and open-habitat species (C) in caraway fields in Finland in relation to the cover of arable land, forest, transitional woodland and water in the surrounding landscape within the 500 m radius. Lines in the scatterplots depict predicted values with 95% confidence intervals based on the models with ∆AICc < 2 (Table 2)
The abundance of wetland species was best explained by the quadratic models with arable land cover and forest cover as predictors (Table 2). The abundance was highest in landscapes with 23% arable land cover and 33% forest cover (Fig. 4, Online Resource Table 4). The species richness of wetland species was best explained by the quadratic model with water cover as a preditor (Table 2). The species richness peaked in landscapes with 10% water cover (Fig. 4, Online Resource Table 4).
Abundance and species richness of wetland species (A) and mixed-habitat species (B) in caraway fields in Finland in relation to the cover of arable land, forest, transitional woodland and water in the surrounding landscape within the 500 m radius. Lines in the scatterplots depict predicted values with 95% confidence intervals based on the models with ∆AICc < 2 (Table 2)
The abundance of mixed-habitat species was best explained by the cover of transitional woodland (Table 2). For the species richness of mixed habitat species, the models with transitional woodland cover and arable land cover were considered equally good, yet the model with arable land cover had a relatively low Akaike weight (wi = 0.18; Table 2), indicating considerable model selection uncertainty. The abundance and species richness of mixed habitat species increased with increasing transitional woodland cover (Fig. 4, Online Resource Table 4). The species richness slightly decreased with increasing arable land cover (Fig. 4, Online Resource Table 4).
Discussion
Our results showed that, even in relatively heterogeneous, forest-dominated landscapes, hoverfly diversity in arable land increased with increasing surrounding forest cover and decreasing arable land cover. Due to the high negative correlation between arable land and forest covers in our boreal study area, the effects of the two land cover classes are difficult to distinguish from each other. However, while increasing arable land cover was associated with reductions in both total abundance and species richness, the opposite effect of forest was more pronounced for species richness than for abundance. This reflects, firstly, the high species richness of hoverflies associated to forests, and the high abundance of open-habitat hoverfly species in our study. Secondly, the result indicates the heterogeneity of microhabitats and resources for hoverflies in forests (e.g. decaying wood, swamp, trees with aphids, and openings with herbaceous vegetation). The resource heterogeneity has a stronger effect on species richness than on abundance, which, in contrast, depends more on resource quantity and can be driven by a small number of species (Meyer et al. 2009). In line with studies from more southern regions (Sjödin et al. 2008; Meyer et al. 2009; Schirmel et al. 2018), our results suggests that forests are crucial in maintaining hoverfly species richness in boreal agricultural landscapes, whereas hoverfly abundance is importantly supported by other habitats besides forests, and is not guaranteed by high forest cover.
Forest species and open-habitat species responded to the surrounding landscape composition in a surprisingly similar manner, although forest species showed a steeper decline with increasing arable land cover and decreasing forest cover. Almost 70% of the individuals of forest species in our study were sapro-xylophagous, eating decaying wood in their larval stage, and being thus directly dependent on the proximity of woody habitats. However, the majority of forest species were aphidophagous as larvae, as were the majority of individuals and species belonging to the open-habitat species group. Although aphids can be occasionally abundant in crop fields, forest-dominated landscapes may provide higher amounts, and greater stability of aphid resources (Söderman et al. 2016; Stack Whitney et al. 2016; Moquet et al. 2018), as well as aphid honeydew, commonly utilized by aphidophagous hoverflies as adults (van Rijn et al. 2013). Besides larval resources, adult hoverflies may find floral resources within the forests (Ricarte et al. 2011; Proesmans et al. 2019; Gaytán et al. 2020; González et al. 2022), and in herbaceous habitats in forest edges, tracks and openings, similarly to farmland butterflies (Berg et al. 2011; Toivonen et al. 2017). In our study, data on floral resources were not collected. However, certain woody plants that are common in the area, e.g. Salix sp. and Calluna vulgaris, may provide important pollen and nectar sources for hoverflies in forests and their edges in early or late season when floral resources are generally scarce (Branquart and Hemptinne 2000). In addition, pesticide use in arable land is potentially harmful to hoverflies (Potts et al. 2010; Moens et al. 2011).
The negative effect of arable land on open-habitat species can also be partly explained by the categorization of hoverflies in our study: open-habitat species also included generalist species that occupy many kinds of habitats. For example, X. segnis and Syrphus ribesii are common in forests, besides open habitats. Meyer et al. (2009), who classified hoverfies according to their larval macrohabitats, found that the density of species associated to crops increased with increasing arable land cover in landscapes dominated by arable land. However, the hoverfly communities of Meyer et al. (2009) were dominated by one crop-associated species, Episyrphus balteatus, which is highly mobile and ubiquitous, and thus less dependent on landscape complexity than most hoverfly species. The differences in the results between the studies can thus be due to the differences in the most abundant species.
The relatively low proportional abundance of aphidophagous species in our study possibly strengthened the landscape effects on hoverfly abundance. Aphidophagous species, which belong to the subfamily Syrphinae, are generally less specialized in terms of flower visitation than other hoverfly subfamilies (Klecka et al. 2018). Since aphids occur in many habitat types, Syrphinae species can find both larval and adult feeding resources in a relatively wide range of habitats. In the study of Toivonen et al. (2022) on caraway pollination in Southern Finland, caraway flower visits by Syrphinae hoverflies did not respond to forest cover in the landscape, whereas hoverflies of the subfamily Eristalinae increased with increasing forest cover. Compared to the present study, the study of Toivonen et al. (2022) was conducted in landscapes with substantially higher arable land cover (49% vs. 29% within the 500 m radius), and Syrphinae species represented a much higher share of observed individuals (87% vs. 32%).
Mixed-habitat species showed a positive response to the cover of transitional woodland, which included forest clear-cuts, power-line corridors and other areas with bushy and herbaceous vegetation with occasional adult trees. These areas are often flower-rich, and they have been shown to provide valuable habitats for many pollinators, including butterflies, bees and wasps (Berg et al. 2011; Rubene et al. 2015; Ram et al. 2020). Compared to forest, transitional woodland had relatively low covers in the study area, and thus, an increase in its cover possibly diversified the availability of resources at the landscape scale.
The abundance or species richness of wetland species did not benefit from a high cover of any land use classes but peaked at intermediate covers. Thus, very high forest cover may also be harmful for some species, which highlights the importance of land use diversity. The species richness of wetland species was highest in the landscapes with 10% water cover. While the species are dependent on wet habitats, which they can find on the shores of lakes, ponds and streams, large continuous water areas do not provide habitats for them. This explains the hump-shaped response to water cover.
The total number of hoverfly species in our study (105 species, representing 29% of all hoverfly species in Finland; Kahanpää 2020) was high considering that the collection was restricted to one habitat type (caraway field) and early summer. The total species richness was increased by the high number of forest species, which have been scarce in previous studies conducted in landscapes dominated by arable land (see e.g. Meyer et al. 2009; Ricarte et al. 2011; Proesmans et al. 2019). Interestingly, a few species of the genus Xylota formed the majority of individuals (Online Resource Table 1), although Xylota species are thought to be hard to catch (Rotheray and Gilbert 2011). While some Xylota species feed mainly on pollen and honeydew deposited on plant leaves, X. segnis and X. jakutorum, which were abundant in our study, actively visit flowers (Haarto and Kerppola 2007). Our study strengthens the evidence that mass-flowering caraway is a highly attractive resource for hoverflies (Toivonen et al. 2022). Although our study did not measure ecosystem services provided by hoveflies, it is likely that the reported effects of landscape composition on hoverfly abundance and species richness in caraway fields have consequences to the pollination and pest control of the crop (Dunn et al. 2020; Toivonen et al. 2022).
Conclusions
Landscape composition strongly affected hoverfly diversity in boreal farmland. Even in landscapes with relatively low arable land cover and high forest cover, the majority of hoverfies visiting mass-flowering crop fields benefitted from increasing surrounding forest cover and decreasing arable land cover. Besides hoverfly species associated to forest habitats, this applied to generalist species that are common in open environments. The result highlights the role of forest as a key habitat for a wide variety of hoverflies with different adult habitat preferences and larval feeding habits. However, land use diversity is also important: the availability of wet habitats is critical for some species, and open and semi-open habitats may temporarily provide abundant resources for hoverflies. Thus, preserving or increasing the area of forests and other non-arable habitats is needed to safeguard hoverflies and associated ecosystem services in boreal farmland.
References
Bartsch H, Binkiewicz E, Rådén A, Nasibov E (2009a) Nationalnyckeln till Sveriges flora och fauna. Tvåvingar: Blomflugor: Syrphinae. Diptera: Syrphidae: Syrphinae. ArtDatabanken, SLU, Uppsala
Bartsch H, Binkiewicz E, Rådén A, Nasibov E (2009b) Nationalnyckeln till Sveriges flora och fauna. Tvåvingar: Blomflugor: Eristalinae & Microdontinae. Diptera: Syrphidae: Eristalinae & Microdontinae. ArtDatabanken, SLU, Uppsala
Berg Å, Ahrné K, Öckinger E, Svensson R, Söderström B (2011) Butterfly distribution and abundance is affected by variation in the Swedish forest-farmland landscape. Biol Conserv 144:2819–2831. https://doi.org/10.1016/j.biocon.2011.07.035
Billeter R, Liira J, Bailey D, Bugter RJF, Arens PFP, Augenstein I, Aviron S, Baudry J, Bukacek R, Burel F, Cerny M, de Blust G, de Cock R, Diekotter T, Dietz H, Dirksen J, Dormann C, Durka W, Frenzel M, Hamersky R, Hendrickx F, Herzog F, Klotz S, Koolstra B, Lausch A, Le Coeur D, Maelfait JP, Opdam P, Roubalova M, Schermann A, Schermann N, Schmidt T, Schweiger O, Smulders MJM, Speelmans M, Simova P, Verboom J, Van Wingerden WKRE, Zobel M, Edwards PJ (2008) Indicators for biodiversity in agricultural landscapes: a pan-European study. J Appl Ecol 45:141–150. https://doi.org/10.1111/j.1365-2664.2007.01393.x
Bivand RS, Wong DWS (2018) Comparing implementations of global and local indicators of spatial association. TEST 27:716–748. https://doi.org/10.1007/s11749-018-0599-x
Botham MS, Fernandez-Ploquin EC, Brereton T, Harrower CA, Roy DB, Heard MS (2015) Lepidoptera communities across an agricultural gradient: how important are habitat area and habitat diversity in supporting high diversity? J Insect Conserv 19:403–420. https://doi.org/10.1007/s10841-015-9760-y
Branquart E, Hemptinne J-L (2000) Selectivity in the exploitation of floral resources by hoverflies (Diptera: Syrphinae). Ecography 23:732–742. https://doi.org/10.1111/j.1600-0587.2000.tb00316.x
Burnham KP, Andersson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Dainese M, Martin EA, Aizen MA, Albrecht M, Bartomeus I, Bommarco R, Carvalheiro LG, Chaplin-Kramer R, Gagic V, Garibaldi LA, Ghazoul J, Grab H, Jonsson M, Karp DS, Kennedy CM, Kleijn D, Kremen C, Landis DA, Letourneau DK, Marini L, Poveda K, Rader R, Smith HG, Tscharntke T, Andersson GKS, Badenhausser I, Baensch S, Bezerra ADM, Bianchi FJJA, Boreux V, Bretagnolle V, Caballero-Lopez B, Cavigliasso P, Ćetković A, Chacoff NP, Classen A, Cusser S, da Silva e Silva FD, de Groot GA, Dudenhöffer JH, Ekroos J, Fijen T, Franck P, Freitas BM, Garratt MPD, Gratton C, Hipólito J, Holzschuh A, Hunt L, Iverson AL, Jha S, Keasar T, Kim TN, Kishinevsky M, Klatt BK, Klein AM, Krewenka KM, Krishnan S, Larsen AE, Lavigne C, Liere H, Maas B, Mallinger RE, Martinez Pachon E, Martínez-Salinas A, Meehan TD, Mitchell MGE, Molina GAR, Nesper M, Nilsson L, O’Rourke ME, Peters MK, Plećaš M, Potts SG, Ramos DL, Rosenheim JA, Rundlöf M, Rusch A, Sáez A, Scheper J, Schleuning M, Schmack JM, Sciligo AR, Seymour C, Stanley DA, Stewart R, Stout JC, Sutter L, Takada MB, Taki H, Tamburini G, Tschumi M, Viana BF, Westphal C, Willcox BK, Wratten SD, Yoshioka A, Zaragoza-Trello C, Zhang W, Zou Y, Steffan-Dewenter I (2019) A global synthesis reveals biodiversity-mediated benefits for crop production. Sci Adv 5:eaax0121. https://doi.org/10.1126/sciadv.aax0121
Davidar P, Yoganand K, Ganesh T (2001) Distribution of forest birds in the Andaman islands: importance of key habitats. J Biogeogr 28:663–671. https://doi.org/10.1046/j.1365-2699.2001.00584.x
Doyle T, Hawkes WLS, Massy R, Powney GD, Menz MHM, Wotton KR (2020) Pollination by hoverflies in the Anthropocene. Proc R Soc B 287:20200508. https://doi.org/10.1098/rspb.2020.0508
Dunn L, Lequerica M, Reid CR, Latty T (2020) Dual ecosystem services of syrphid flies (Diptera: Syrphidae): pollinators and biological control agents. Pest Manag Sci 76:1973–1979. https://doi.org/10.1002/ps.5807
Esri (2014) ArcGIS Desktop: Release 10.3.1. Environmental Systems Research Institute, Redlands
Finnish Environment Institute (2012) CORINE Land Cover 2012 dataset. https://www.avoindata.fi/data/en_GB/dataset/corine-maanpeite-2012
Gaytán Á, Ricarte A, González-Bornay G (2020) Hoverfly diversity (Diptera: Syrphidae) of Pyrenean oak woodlands in Central-Western Spain: a preliminary study with conservation outcomes. J Insect Conserv 24:163–173. https://doi.org/10.1007/s10841-019-00208-z
González E, Bianchi FJJA, Eckerter PW, Pfaff V, Weiler S, Entling MH (2022) Ecological requirements drive the variable responses of wheat pests and natural enemies to the landscape context. J Appl Ecol 59:444–456. https://doi.org/10.1111/1365-2664.14062
Goulson D, Wright N (1998) Flower constancy in the hoverflies Episyrphus balteatus (Degeer) and Syrphus ribesii (L.) (Syrphidae). Behav Ecol 9:213–219. https://doi.org/10.1093/beheco/9.3.213
Haarto A, Kerppola S (2007) Suomen kukkakärpäset ja lähialueiden lajeja (Finnish hoverflies and some species in adjacent countries). Otava, Keuruu
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, Stenmans W, Müller A, Sumser H, Hörren T, Goulson D, de Kroon H (2017) More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 12:e0185809. https://doi.org/10.1371/journal.pone.0185809
Hogg BN, Nelson EH, Mills NJ, Daane KM (2011) Floral resources enhance aphid suppression by a hoverfly. Entomol Exp Appl 141:138–144. https://doi.org/10.1111/j.1570-7458.2011.01174.x
Hyvärinen E, Juslén A, Kemppainen E, Uddström A, Liukko U-M (eds) (2019) The 2019 Red List of Finnish Species. Ympäristöministeriö & Suomen ympäristökeskus, Helsinki
Kahanpää J (2020) Diptera, true flies. In: FinBIF 2020. The FinBIF checklist of Finnish species 2019. Finnish Biodiversity Information Facility, Finnish Museum of Natural History, University of Helsinki, Helsinki
Klaus F, Bass J, Marholt L, Müller B, Klatt B, Kormann U (2015) Hedgerows have a barrier effect and channel pollinator movement in the agricultural landscape. J Landsc Ecol 8:22–31. https://doi.org/10.1515/jlecol-2015-0001
Klecka J, Hadrava J, Biella P, Akter A (2018) Flower visitation by hoverflies (Diptera: Syrphidae) in a temperate plant-pollinator network. PeerJ 6:e6025. https://doi.org/10.7717/peerj.6025
Koul P, Sharma N, Koul AK (1993) Pollination biology of Apiaceae. Curr Sci India 65:219–222. https://www.jstor.org/stable/24095119
Kovács-Hostyánszki A, Espíndola A, Vanbergen AJ, Settele J, Kremen C, Dicks LV (2017) Ecological intensification to mitigate impacts of conventional intensive land use on pollinators and pollination. Ecol Lett 20:673–689. https://doi.org/10.1111/ele.12762
Kremen C, Miles A (2012) Ecosystem services in biologically diversified versus conventional farming systems: benefits, externalitites, and trade-offs. Ecol Soc 17:1–23. https://doi.org/10.5751/ES-05035-170440
Lewis T (1969) The distribution of flying insects near a low hedgerow. J Appl Ecol 6:443–452. https://doi.org/10.2307/2401510
Mazerolle MJ (2019) AICcmodavg: Model selection and multimodel inference based on (Q)AIC(c). R package version 2.2-0. https://cran.r-project.org/package=AICcmodavg.
Meyer B, Jauker F, Steffan-Dewenter I (2009) Contrasting resource-dependent responses of hoverfly richness and density to landscape structure. Basic Appl Ecol 10:178–186. https://doi.org/10.1016/j.baae.2008.01.001
Moens J, De Clercq P, Tirry L (2011) Side effects of pesticides on the larvae of the hoverfly Episyrphus balteatus in the laboratory. Phytoparasitica 39:1–9. https://doi.org/10.1007/s12600-010-0127-3
Moquet L, Laurent E, Bacchetta R, Jacquemart A-L (2018) Conservation of hoverflies (Diptera, Syrphidae) requires complementary resources at the landscape and local scales. Insect Conserv Divers 11:72–87. https://doi.org/10.1111/icad.12245
Öckinger E, Lindborg R, Sjödin NE, Bommarco R (2012) Landscape matrix modifies richness of plants and insects in grassland fragments. Ecography 35:259–267. https://doi.org/10.1111/j.1600-0587.2011.06870.x
Öckinger E, Schweiger O, Crist TO, Debinski DM, Krauss J, Kuussaari M, Petersen JD, Pöyry J, Settele J, Summerville KS, Bommarco R (2010) Life-history traits predict species responses to habitat area and isolation: a cross-continental synthesis. Ecol Lett 13:969–979. https://doi.org/10.1111/j.1461-0248.2010.01487.x
Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, Kunin WE (2010) Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol 25:345–353. https://doi.org/10.1016/j.tree.2010.01.007
Potts SG, Imperatriz-Fonseca V, Ngo HT, Aizen MA, Biesmeijer JC, Breeze TD, Dicks LV, Garibaldi LA, Hill R, Settele J, Vanbergen AJ (2016) Safeguarding pollinators and their values to human well-being. Nature 540:220–229. https://doi.org/10.1038/nature20588
Powney GD, Carvell C, Edwards M, Morris R, Roy HE, Woodcock BA, Isaac N (2019) Widespread losses of pollinating insects in Britain. Nat Commun 10:1018. https://doi.org/10.1038/s41467-019-08974-9
Proesmans W, Bonte D, Smagghe G, Meeus I, Verheyen K (2019) Importance of forest fragments as pollinator habitat varies with season and guild. Basic Appl Ecol 34:95–107. https://doi.org/10.1016/j.baae.2018.08.004
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/.
Rader R, Bartomeus I, Garibaldi LA, Garratt MPD, Howlett BG, Winfree R, Cunningham SA, Mayfield MM, Arthur AD, Andersson GKS, Bommarco R, Brittain C, Carvalheiro LG, Chacoff NP, Entling MH, Foully B, Freitas BM, Gemmill-Herren B, Ghazoul J, Griffin SR, Gross CL, Herbertsson L, Herzog F, Hipólito J, Jaggar S, Jauker F, Klein A, Kleijn D, Krishnan S, Lemos CQ, Lindström SAM, Mandelik Y, Monteiro VM, Nelson W, Nilsson L, Pattemore DE, de Pereira ON, Pisanty G, Potts SG, Reemer M, Rundlöf M, Sheffield CS, Scheper J, Schüepp C, Smith HG, Stanley DA, Stout JC, Szentgyörgyi H, Taki H, Vergara CH, Viana BF, Woyciechowski M (2016) Non-bee insects are important contributors to global crop pollination. Proc Natl Acad Sci USA 113:146–151. https://doi.org/10.1073/pnas.1517092112
Ram D, Lindström Å, Pettersson LB, Caplat P (2020) Forest clear-cuts as habitat for farmland birds and butterflies. Forest Ecol Manag 473:118239. https://doi.org/10.1016/j.foreco.2020.118239
Raymond L, Sarthou J-P, Plantegenest M, Gauffre B, Ladet S, Vialatte A (2014) Immature hoverflies overwinter in cultivated fields and may significantly control aphid populations in autumn. Agr Ecosyst Environ 185:99–105. https://doi.org/10.1016/j.agee.2013.12.019
Ricarte A, Marcos-García ÁM, Moreno CE (2011) Assessing the effects of vegetation type on hoverfly (Diptera: Syrphidae) diversity in a Mediterranean landscape: implications for conservation. J Insect Conserv 15:865–877. https://doi.org/10.1007/s10841-011-9384-9
Ricciardelli D’Albore GC (1986) Les insectes pollinisateurs de quelques ombellifères d’intérêt agricole et condimentaire (Angelica archangelica L., Carum carvi L., Petroselinum crispum A.W. Hill., Apium graveolens L., Pimpinella anisum L., Daucus carota L., Foeniculum vulgare Miller v. azoricum Thell.). Apidologie 17:107–124. https://doi.org/10.1051/apido:19860204
Rotheray GE, Gilbert F (2011) The natural history of hoverflies. Forrest text, Tresaith
Rotheray EL, MacGowan I, Rotheray GE, Sears J, Elliott A (2009) The conservation requirements of an endangered hoverfly, Hammerschmidtia ferruginea (Diptera, Syrphidae) in the British Isles. J Insect Conserv 13:569–574. https://doi.org/10.1007/s10841-008-9204-z
Rubene D, Schroeder M, Ranius T (2015) Diversity patterns of wild bees and wasps in managed boreal forests: effects of spatial structure, local habitat and surrounding landscape. Biol Conserv 184:201–208. https://doi.org/10.1016/j.biocon.2015.01.029
Senapathi D, Carvalheiro LG, Biesmeijer JC, Dodson C-A, Evans RL, McKerchar M, Morton RD, Moss ED, Roberts SPM, Kunin WE, Potts SG (2015) The impact of over 80 years of land cover changes on bee and wasp pollinator communities in England. Proc R Soc B 282:1806. https://doi.org/10.1098/rspb.2015.0294
Schirmel J, Albrecht M, Bauer P-M, Sutter L, Pfister SC, Entling MH (2018) Landscape complexity promotes hoverflies across different types of semi-natural habitats in farmland. J Appl Ecol 55:1747–1758. https://doi.org/10.1111/1365-2664.13095
Schweiger O, Musche M, Bailey D, Billeter R, Diekötter T, Hendrickx F, Herzog F, Liira J, Maelfait J-P, Speelmans M, Dziock F (2007) Functional richness of local hoverfly communities (Diptera, Syrphidae) in response to land use across temperate Europe. Oikos 116:461–472. https://doi.org/10.1111/j.2007.0030-1299.15372.x
Seibold S, Gossner MM, Simons NK, Blüthgen N, Müller J, Ambarlı AC, Bauhus J, Fischer M, Habel JC, Linsenmair KE, Nauss T, Penone C, Prati D, Schall P, Schulze E-D, Vogt J, Wöllauer S, Weisser WW (2019) Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574:671–674. https://doi.org/10.1038/s41586-019-1684-3
Sjödin NE, Bengtsson J, Ekbom B (2008) The influence of grazing intensity and landscape composition on the diversity and abundance of flower-visiting insects. J Appl Ecol 45:763–772. https://doi.org/10.1111/j.1365-2664.2007.01443.x
Söderman AME, Ekroos J, Hedlund K, Olsson O, Smith HG (2016) Contrasting effects of field boundary management on three pollinator groups. Insect Conserv Diver 9:427–437. https://doi.org/10.1111/icad.12179
Speight MCD (2011) Species accounts of European Syrphidae (Diptera), Glasgow 2011. Syrph the Net: the database of European Syrphidae, vol. 65. Syrph the Net publications, Dublin
Stack Whitney K, Meehan TD, Kucharik CJ, Zhu J, Townsend PA, Hamilton K, Gratton C (2016) Explicit modeling of abiotic and landscape factors reveals precipitation and forests associated with aphid abundance. Ecol Appl 26:2600–2610. https://doi.org/10.1002/eap.1418
Stein A, Gerstner K, Kreft H (2014) Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecol Lett 17:866–880. https://doi.org/10.1111/ele.12277
Stoate C, Báldi A, Beja P, Boatman ND, Herzon I, van Doorn AM, de Snoo GR, Rakosy L, Ramwell C (2009) Ecological impacts of early 21st century agricultural change in Europe—a review. J Environ Manag 91:22–46. https://doi.org/10.1016/j.jenvman.2009.07.005
Toivonen M, Karimaa A-E, Herzon I, Kuussaari M (2022) Flies are important pollinators of mass-flowering caraway and respond to landscape and floral factors differently from honeybees. Agr Ecosyst Environ 323:107698. https://doi.org/10.1016/j.agee.2021.107698
Toivonen M, Peltonen A, Herzon I, Heliölä J, Leikola N, Kuussaari M (2017) High cover of forest increases the abundance of most grassland butterflies in boreal farmland. Insect Conserv Diver 10:321–330. https://doi.org/10.1111/icad.12226
Turner MG, Gardner RH (2015) Landscape ecology in theory and practice. Springer, New York
van Rijn PCJ, Kooijman J, Wäckers F (2013) The contribution of floral resources and honeydew to the performance of predatory hoverflies (Diptera: Syrphidae). Biol Control 67:32–38. https://doi.org/10.1016/j.biocontrol.2013.06.014
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Wratten SD, Bowie MH, Hickman JM, Evans AM, Sedcole JR, Tylianakis JM (2003) Field boundaries as barriers to movement of hover flies (Diptera: Syrphidae) in cultivated land. Oecologia 134:605–611. https://doi.org/10.1007/s00442-002-1128-9
Zhang W, Ricketts TH, Kremen C, Carney K, Swinton SM (2007) Ecosystem services and dis-services to agriculture. Ecol Econ 64:253–260. https://doi.org/10.1016/j.ecolecon.2007.02.024
Zou Y, Feng J, Xue D, Sang W, Axmacher JC (2012) A comparison of terrestrial arthropod sampling methods. J Resour Ecol 3:174–182. https://doi.org/10.5814/j.issn.1674-764x.2012.02.010
Acknowledgements
We are grateful to the farmers who gave permission for our field sampling. JT acknowledges funding from Emil Aaltonen Foundation, Oiva Kuusisto Foundation, Societas pro Fauna et Flora Fennica and Societas Entomologica Fennica. MT acknowledges funding from Oiva Kuusisto Foundation.
Funding
This work was supported by Emil Aaltonen Foundation, Oiva Kuusisto Foundation, Societas pro Fauna et Flora Fennica and Societas Entomologica Fennica. Open access funding provided by Finnish Environment Institute (SYKE).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. JT collected the data. JT and MT analyzed the data, wrote the manuscript text and prepared the figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.〹
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Toikkanen, J., Halme, P., Kahanpää, J. et al. Effects of landscape composition on hoverflies (Diptera: Syrphidae) in mass-flowering crop fields within forest-dominated landscapes. J Insect Conserv 26, 907–918 (2022). https://doi.org/10.1007/s10841-022-00436-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-022-00436-w