Abstract
Introduction
Evolutionary traits acquired in response to one type of disturbance will potentially confer resilience to other disturbances that have similar environmental impacts, even if the biota has no evolutionary history of such disturbances. In grassy ecosystems the environmental impacts of grazing have important similarities to those of fire through the removal of grass biomass, and we hypothesise that high resilience to frequent fire confers high resilience to grazing.
Aims
We test this hypothesis by investigating the resilience of highly fire-resilient ant communities to grazing in a mesic Australian savanna, which has not historically experienced such high levels of mammalian grazing.
Methods
We sampled ants using pitfall traps at Annaburroo Station in the Australian seasonal tropics using ten plot triplets, with each triplet representing no, low and heavy grazing. Grazing has had a major impact on the basal area of perennial grasses and the cover of bare ground. We considered large (> 4 mm) ants only, which tend to be particularly sensitive to disturbance.
Results
We recorded 28 species of ‘large’ ants from 14 genera. Neither ant species richness nor overall composition varied significantly with grazing, and only one of the eight most common species responded to grazing.
Discussion
Ant communities at Annaburoo Station are highly resilient to livestock grazing. The limited number of relevant studies suggest that ant communities in Australian savannas more generally have higher resilience than those in southern rangelands where fire frequency is relatively low. This supports our hypothesis that an evolutionary history of frequent fire confers resilience to grazing. Many more studies are required, but we suggest that resilience to grazing might be related more to evolutionary history in relation to fire, a more pervasive remover of plant biomass globally, than to grazing.
Implications for insect conservation
Grazing by cattle is the dominant land-use in Australian savannas and the Australian savanna ant fauna is one of the richest on Earth; our findings of high resilience is therefore good news for insect conservation. Such good news is likely to have wide applicability to other fire-prone grassy ecosystems subject to livestock grazing.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The resilience of biological communities - their ability to return to their prior state after disturbance - is fundamental to their ecological dynamics (Holling 1973). Resilience is strongly influenced by ‘ecological memory’, defined as the ability of past states or experiences to affect the contemporary response of a community to disturbance (Thompson et al. 2001; Bengtsson et al. 2003; Johnstone et al. 2016). This means that different ecosystems with different ecological memories are likely to support biological communities that differ in their resilience (Moretti et al. 2006; Moretti and Legg 2009; Laliberte et al. 2010; Peco et al. 2012).
Ecological disturbance plays a fundamental role in the dynamics of most ecosystems (Sousa 1984). Survival and regeneration strategies in relation to disturbance are responses to disturbance impact rather than to the agent of disturbance itself (Keeley et al. 2011). This implies that evolutionary traits acquired in response to one type of disturbance can confer resilience in relation to other disturbances that have similar environmental impacts, despite having no direct experience with these other disturbances (Peterson 2002; Keeley et al. 2011). For example, fire removes vegetation biomass, especially from the ground layer, and, more than fire itself, this provides the evolutionary pressure for plant and animal responses (Whelan 1995; Bond and Van Wilgen 1996). It means that adaptive traits in relation to fire should also be effective for other agents of disturbance that similarly remove plant biomass and therefore create similar environmental conditions.
In addition to fire, grazing is another major agent of disturbance in grassy ecosystems worldwide (Bond and Parr 2010) that similarly acts to remove plant biomass from the ground layer (Crawley 1983; Ash et al. 1997; Ash and Corfield 1998; Laycock 1991). Fire is obviously different from grazing in that it has little selectivity and can result in the complete removal of above-ground grass biomass. Fire also consumes grass biomass in a single pulse rather than being an ongoing pressure (Archibald and Hempson 2016). However, the effects of fire persist until grass biomass is restored, which is typically at least several months. Given that fire frequency is often very high, even annual, in grassy ecosystems (Parr et al. 2014), this means that the effects of grass consumption by fire can occur for a substantial proportion of overall time. Moreover, frequent fire results in persistent reductions in litter cover in grassy ecosystems, both through direct consumption (Cook 2012) and by reducing woody cover (Sankaran 2005). Both fire and grazing therefore act to increase openness of the ground habitat, which is a critical factor mediating faunal responses to disturbance (Andersen 2019). It can thus be hypothesized that communities with an evolutionary history of frequent fire will also have high resilience to heavy grazing, even if they have had no direct experience of it. This has important implications for biodiversity conservation given that livestock grazing is a dominant land-use in grassy ecosystems throughout the world.
With most areas burning every 2–3 years (Andersen et al. 2003; Russell-Smith et al. 1997; Russell-Smith and Yates 2007), Australian savannas are among the world’s most fire-prone biomes, and, as in other savannas, have experienced frequent fire throughout their evolutionary history (Bourlière1983; Hutley and Setterfield 2019). Consequently, the Australian savanna biota is highly resilient to frequent fire (Williams et al. 2003; Andersen et al. 2005; Andersen 2021;). The dominant land-use across Australian savannas is grazing by cattle (Australian Bureau of Agricultural and Resource Economics and Sciences 2016). It is likely that current grazing pressure by mammalian herbivores is now higher than it has ever been, given that the native megaherbivores that were driven to extinction following the arrival of humans about 50,000 years ago tended to be browsers rather than grazers (Johnson 2006), and the abundance of extant native herbivores is generally low due to low-nutrient soils, water limitation and predation (Eldridge et al. 2016; Letnic et al. 2009). We hypothesise that despite no evolutionary history of high grazing pressure, faunal groups showing high resilience to frequent fire are also highly resilient to high levels of grazing.
Here we test this hypothesis by investigating the resilience of ant communities to grazing in a mesic Australian savanna. Such ant communities show especially high resilience to frequent fire (Andersen et al. 2012, 2014) because they are dominated by arid-adapted taxa that are favoured by the open habitats that are maintained by high fire frequency (Andersen 2003, 2007; Andersen and Vasconcelos 2022). We therefore predict that these communities will also be highly resilient to grazing, more so than ant communities without an evolutionary history of frequent fire. Previous studies in Australian savannas at Lakefield Station (1,050 mm mean annual rainfall; Arcoverde et al. 2017) and the Victoria River District (< 650 mm; Hoffmann 2000) in the Northern Territory showed ants to be highly resilient to grazing, but the extent to which such resilience extends to more mesic savannas remains unknown. Disturbance might be expected to have greater impacts in areas of higher rainfall because of higher vegetation complexity (Andersen 2019). In this paper we examine ant responses to grazing in the far north of the Northern Territory, where mean annual rainfall exceeds 1,500 mm.
A global review of ant responses to livestock grazing found that the foraging abundances of typically between 25 and 50% of ant species that could be analysed statistically were significantly affected (Hoffmann 2010). We specifically predict that this figure will be markedly less than 25% in our study. We first examine the effects of grazing on the structure of ground-layer vegetation, to document the extent to which grazing affects key habitat variables for ants. We then describe the effects of grazing on ant communities, using a simplified assessment protocol that considers larger species (> 4 mm) only. The targeting of larger ant species for assessment of disturbance impacts has proven to be highly efficient and reliable, providing similar results to those of entire ant communities (Andersen et al. 2002; Arcoverde et al. 2018). Moreover, larger ant species tend to be especially sensitive to disturbance (Gibb et al. 2018; Ness et al. 2004), and so if anything we would expect to find greater impacts of grazing than is the case for entire ant communities.
Materials and methods
Study area
The study was conducted at Annaburroo Station, a 308 km2 cattle station located 100 km southeast of Darwin in Australia’s Northern Territory. The area experiences a tropical monsoonal climate, with its mean annual rainfall of 1522 mm heavily concentrated in a summer wet season from December to April (Bureau of Meteorology 2022). Mean temperature shows relatively little variation between summer (mean maximum and minimum temperatures of 36.4ºC and 25.5 ºC respectively) and winter (31.7 ºC and 19.2 ºC). The vegetation is savanna woodland dominated by evergreen species of Eucalyptus and Corymbia, with an understory dominated by C4 perennial grasses (Egan and Williams 1996).
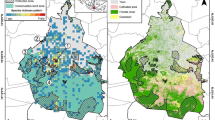
Annaburroo is an ideal location for the study of grazing impacts because it includes an extensive conservation corridor connecting two national parks, where grazing has been excluded for > 20 years. Our study design was based on cross-fence comparisons inside and outside the conservation corridor (Fig. 1). The corridor runs east-west and then north-south for about 10 km with a width of approximately 100 m, and cattle are managed differently in the paddocks on either side. For at least the past five years cattle have been held throughout the year to the south and east, but for only six months on the other side; we refer to these areas as experiencing high and low levels of grazing respectively. It was therefore possible to locate plots of no, low and high levels of grazing in very close proximity to each other, and we established ten such triplets of plots (Fig. 1) spread along the conservation corridor, with triplets separated from each other by at least 100 m.
Sampling
Ants were sampled in each of the 30 plots using 20 pitfall traps (45 mm-diameter plastic specimen containers, partly filled with ethylene glycol as a preservative), arranged in a line with 10 m spacing between traps. ‘No’ transects were located along the centre of the conservation corridor, and ‘Low’ and ‘High’ transects were located approximately 30 m from the corridor fences and thus approximately 80 m from ‘No’ transects. Traps were operated for a 48-hr period in September 2014.
Larger ants were sorted and identified following Andersen et al. (2002) using a body length guideline of > 4 mm. We specifically defined large ants as all species within the genera Lioponera, Rhytidoponera, Anochetus, Odontomachus, Leptogenys, Platythyrea, Pseudoneoponera, Camponotus, Oecophylla, Polyrhachis and Opisthopis, as well as species of the rufoniger and curtus groups of Melophorus, the rothsteini group of Monomorium, and the purpureus group of Iridomyrmex (see Andersen 2000 for these groups). Ants were sorted to species, and when possible named, following the species nomenclature of Shattuck (2000). Species that could not be named were identified to species group following Andersen (2000) and assigned code numbers (sp. 1, etc.) following those of previous studies in the Top End of the Northern Territory (Andersen et al. 2016). Voucher specimens of all species are deposited in the ant collection held at the CSIRO Laboratory in Darwin. Species abundances were recorded and capped at 100 per trap to avoid data distortions caused by a very large number of individuals from a single colony falling into one or a few traps (see Andersen 1991).
We measured ground-layer structural variables likely to be affected by grazing using 1 m × 1 m quadrats centred on each pitfall trap location (n = 20 per plot) to visually estimate the percentage basal area of perennial grasses, and the percentage cover of herbs (annual grasses and forbs), litter, and bare ground.
Analysis
We used linear regression models to analyze the relationship between grazing level and ground-layer habitat variables. Herb cover was highly correlated (R > 0.7) with the cover of perennial grasses, and so we excluded it from analysis. Linear regressions were checked for outliers, overly influential observations, homoscedasticity and normal distribution of residuals. Analyses were performed using the multcomp package in R (R Core Team 2014).
We used GLMs to analyze variation in ant abundance and species richness in relation to grazing level and environmental variables. We used Poisson error distribution for species richness and Quasi-Poisson error distribution for abundance to account for over-dispersed data (Crawley 2012). We also conducted GLMs on the individual abundances of the most common species, defined as those occurring at > 9 sites and with > 50 total individuals. For GLMs with grazing level (a categorical variable), we established “no grazing” as the basis for comparisons with other grazing levels. Models were checked for outliers, overly influential observations, homoscedasticity and normal distribution of residuals. Analyses were performed using the car and multcomp package in R (R Core Team 2014).
We used Canonical Correspondence Analyses (CCA) to evaluate variation among sites in ant species composition (based on species abundance data), and how such variation was associated with grazing level and ground-layer habitat variables (Ter Braak 1986; Legendre et al. 2011). We conducted randomization tests (10,000 randomizations) to check the statistical significance of grazing level and habitat variables (Legendre et al. 2011). The CCA and randomisation analyses were performed using the vegan package in R (R Core Team 2014).
Results
Ground-layer habitat variables
The basal area of perennial grasses (Fig. 2a) decreased with increasing grazing level, from nearly 50% under no grazing to about 25% under high grazing, and the cover of bare ground (Fig. 2b) increased from about 10–25% under no and high grazing respectively (Table 1). The cover of neither litter (Fig. 2c) nor herbs (Fig. 2d) varied in relation to grazing (Table 1).
The ant fauna
We recorded a total of 13,158 ‘large’ ants from 28 species and 14 genera (Supplementary material Appendix 1, Table A1). The richest genera were Rhytidoponera (6 species) and Camponotus, Leptogenys, Opisthopsis, and Polyrhachis (each with 3 species). The most common species were Iridomyrmex sanguineus (representing 65% of all ants, recorded from 86% of the plots), I. reburrus (12%, 25%) and Monomorium maryannae (9%, 301%); these three species together represented 86.% of all ‘large’ ants recorded.
Responses to grazing
Total ant abundance was significantly lower in the low grazing treatment than in the other treatments, which did not vary significantly from each other (Fig. 3a; Table 2). Mean ant richness showed no variation in relation to either grazing (Fig. 3b; Table 2) or to the ground-layer habitat variables influenced by grazing. However, it increased with increasing litter cover (Table 2).
Eight species were common enough for statistical analysis, and the abundance of only one (12.5%) of these varied significantly with grazing level: the abundance of Iridomyrmex sanguineus in low-grazing plots was half or less than in no and high plots (Fig. 4; Table 2). The overall abundance of the harvester ant Monomorium maryannae was far higher in the absence of grazing (Table S1), but nearly all records were from just two plots and so the differences are not statistically significant. The abundances of two of the other common species varied with habitat variables that were influenced by grazing, but they did not vary with grazing; the abundance of Melophorus sp. 59 increased with the increase of bare ground, and the abundance of Iridomyrmex reburrus decreased with increasing cover of perennial grasses (Table 2).
Overall species composition did not vary with grazing (χ² = 0.089, F = 1.109, DF = 1, p = 0.308; Fig. 5) or with any ground-layer habitat variables.
Discussion
Our study tested the hypothesis that the high resilience of ant communities to fire in Australian savannas confers high resilience to livestock grazing, because fire and grazing have many similar effects on ant habitat structure, and adaptive responses to disturbance relate to disturbance impact rather than to the agent of disturbance itself. We found strong effects of grazing on ground-layer habitat structure, substantially reducing grass basal area and increasing the cover of bare ground. Reductions in the cover of perennial grasses and increases in the cover of bare ground are typical effects of even low levels of livestock grazing globally, due to the combined effects of removal of grass biomass and trampling (Ash et al. 1997; Eldridge et al. 2011; Tongway and Ludwig 2011).
The cover of perennial grasses and bare ground are often particularly important variables influencing the structure of savanna ant communities (Parr et al. 2004; Vasconcelos et al. 2008), but, despite this, the marked effect of grazing on them in our study was not translated to major impact on ant communities. The abundance of only one species common enough for statistical analysis, Iridomyrmex sanguineus, varied significantly among grazing levels. Its abundance was lowest under low grazing and this was a driver of the same pattern in overall ant abundance. Iridomyrmex sanguineus is an ecologically dominant ant throughout monsoonal Australia and has a strong preference for open environments (Andersen 2000). It is heavily favoured by frequent fire (Andersen et al. 2012) and its abundance was promoted by grazing at Lakefield Station elsewhere in the Northern Territory (Arcoverde et al. 2016). We are unable to explain its pattern of abundance in relation to grazing in our study.
Only 12.5% of common species in our study showed a detectable response to grazing, which is markedly less than the 25–50% typically reported globally (Hoffmann 2010). We acknowledge that only eight of our 28 sampled species were common enough for statistical analysis and that the responses of these common species may not reflect those of rare species. We also acknowledge that with additional sampling we may have obtained data on a larger number of species. However, we note that neither species richness nor composition varied with grazing, and these both considered all 28 of our sampled species. Moreover, our sampling intensity was comparable to that of other studies showing far higher responsiveness among common species. For example, in a South Australian rangeland where sites were similarly surveyed by a single bout of 20 pitfall traps, 34% of common species were found to be affected by grazing (Hoffmann and James 2011).
At Lakefield Station (1046 mm mean annual rainfall) elsewhere in the Northern Territory only three (10%) out of the 30 ant species common enough for statistical analysis responded to grazing, and grazing had no significant effect on either species richness or composition (Arcoverde et al. 2017). The proportion of common species affected by grazing in the Victoria River District southwest of Lakefield was also very low (17%, out of 12 species analysed; Hoffmann 2000). Our findings at Annaburroo Station indicate that similar resilience occurs in mesic savannas. Combining results from the three studies, the abundances of only six (12%) out of the 50 species analysed were significantly affected by grazing. It therefore seems clear that ant communities in Australian savannas are particularly resilient to grazing when compared with the global average (25–50% of common species significantly affected; Hoffmann 2010).
The figure of 16% of common species significantly affected by grazing across the three savanna studies in the Northern Territory is far lower than the average of 36% from three relevant studies in central and southern Australia (Arcoverde et al. 2018; Hoffmann and James 2011; Landsberg et al. 1997), where fire is far less frequent, occurring at intervals from one to many decades (Edwards et al. 2008; Nano et al. 2012). The number of studies is too low for formal statistical analysis, but the results are consistent with the notion that ant communities of Australian savannas, where fire frequency is extremely high, are more resilient to grazing than are those from other Australian rangelands, where fire frequency is relatively low. Whatever the case, the high resilience of Australia’s savanna ant fauna to grazing is good news for biodiversity conservation given that this fauna likely contains several thousand species and is possibly the richest in the world (Andersen and Vasconcelos 2022).
High fire frequency is a feature of tropical savannas worldwide (Bourlière 1983). If high resilience to fire also confers high resilience to grazing in savannas regardless of their evolutionary experience with grazing, then this has important implications for international comparisons of grazing impacts in savannas. For example, it would be predicted that ant communities in African savannas that support high biomasses of native mammalian herbivores would not necessarily be more resilient to grazing than are ant communities in other savannas. The effect of grazing on ant communities in African savannas remains almost totally unknown (see Mauda et al. 2017) and so data are not available to test this prediction. There have been a number of studies of the effects of pastoralism on ant communities in Brazilian savannas (Pacheco and Vasconcelos 2012; Pacheco et al. 2013, 2017), and these studies indicate a high degree of resilience. However, livestock grazing in Brazilian savannas is typically on introduced pastures following land clearing, and so the data are not directly comparable quantitatively.
In conclusion, our findings of exceptionally high resilience of ant communities to grazing in Australian savannas are consistent with the hypothesis that high resilience to frequent fire confers high resilience to grazing. Many more studies are required, but we suggest that resilience to grazing globally might be related more to evolutionary history in relation to fire, a more pervasive remover of plant biomass, than to grazing.
References
Andersen AN (1991) Sampling communities of ground-foraging ants: Pitfall catches compared with quadrat counts in an Australian tropical savanna. Austral Ecol 16:273–279
Andersen AN (2000) The ants of northern Australia: a guide to the monsoonal fauna. CSIRO Publishing
Andersen AN (2003) Ant biodiversity in arid Australia: productivity, species richness and community organisation. Records of the South Australian Museum Monograph Series 7:79–92
Andersen AN (2007) Ant diversity in arid Australia: a systematic overview. Advances in ant (Hymenoptera: formicidae) systematics: homage to EO Wilson-50 years of contributions. Mem Am Entomol Inst 80:20
Andersen AN (2019) Responses of ant communities to disturbance: Five principles for understanding the disturbance dynamics of a globally dominant faunal group. J Anim Ecol 88(3):350–362
Andersen AN (2021) Faunal responses to fire in Australian tropical savannas: insights from field experiments and their lessons for conservation management. Divers Distrib 27:828–843
Andersen AN, Cook GD, Corbett LK, Douglas MM, Eager RW, Russell-Smith JE, Setterfield SA, Williams RJ, Woinarski JC (2005) Fire frequency and biodiversity conservation in Australian tropical savannas: implications from the Kapalga fire experiment. Austral Ecol 30:155–167
Andersen AN, Cook GD, Williams RJ (2003) Fire in tropical savannas: the Kapalga experiment. Springer-Verlag, New York.
Andersen AN, Hoffmann BD, Müller WJ, Griffiths AD (2002) Using ants as bioindicators in land management: simplifying assessment of ant community responses. J Appl Ecol 39:8–17
Andersen AN, Hoffmann BD, Oberprieler S (2016) Diversity and biogeography of a species-rich ant fauna of the Australian seasonal tropics. Insect Sci 25:519–526
Andersen AN, Ribbons RR, Pettit M, Parr CL (2014) Burning for biodiversity: highly resilient ant communities respond only to strongly contrasting fire regimes in Australia’s seasonal tropics. J Appl Ecol 51:1406–1413
Andersen AN, Vasconcelos HL (2022) Historical biogeography shapes ecology: contrasting savanna ant communities. J Biogeogr 49:590–599
Andersen AN, Woinarski JC, Parr CL (2012) Savanna burning for biodiversity: fire management for faunal conservation in Australian tropical savannas. Austral Ecol 37:658–667
Archibald S, Hempson GP (2016) Competing consumers: contrasting the patterns and impacts of fire and mammalian herbivory in Africa. Philos T R Soc B 371(1703):20150309
Arcoverde GB, Andersen AN, Setterfield SA (2017) Is livestock grazing compatible with biodiversity conservation? Impacts on savanna ant communities in the Australian seasonal tropics. Biodivers Conserv 26:883–897
Arcoverde GB, Andersen AN, Leal IR, Setterfield SA (2018) Habitat-contingent responses to disturbance: impacts of cattle grazing on ant communities vary with habitat complexity. - Ecol Appl 287:1808–1817
Ash AJ, Corfield JP (1998) Influence of pasture condition on plant selection patterns by cattle: its implications for vegetation change in a monsoon tallgrass rangeland. Trop Grasslands 32:178–187
Ash AJ, McIvor JG, Mott JJ, Andrew MH (1997) Building grass castles: integrating ecology and management of Australia’s tropical tallgrass rangelands. Rangel J 19:123–144
Australian Bureau of Agricultural and Resource Economics and Sciences (2016) Land use of Australia 2010–11 (Version 5 NLUM). http://www.agriculture.gov.au/abares/aclump/land-use. Accessed 20 June 2017
Bengtsson J, Angelstam P, Elmqvist T, Emanuelsson U, Folke C, Ihse M, Moberg F, Nyström M(2003) Reserves, resilience and dynamic landscapes. - AMBIO: A Journal of the Human Environment 32: 389–396
Bond WJ, Parr CL (2010) Beyond the forest edge: Ecology, diversity and conservation of the grassy biomes. Biol Conserv 143:2395–2404
Bond WJ, Van Wilgen BW (1996) Fire and Plants. Chapman & Hall, London
Bourlière F (1983) Tropical savannas, Vol. 13: Ecosystems of the world. Elsevier, Amsterdam
Bureau of Meteorology (2022) Monthly rainfall data - http://www.bom.gov.au/jsp/ncc/cdio/weatherData/av?p_nccObsCode=139&p_display_type=dataFile&p_startYear=&p_c=&p_stn_num=014041. Accessed 24 Febuary 2022
Cook GD (2012) Fire management and minesite rehabilitation in a frequently burnt tropical savanna. Austral Ecol 37(6):686–692
Crawley MJ (1983) Herbivory. The dynamics of animal–plant interactions. Blackwell Scientific Publications, Oxford
Crawley MJ (2012) The R book. John Wiley & Sons, New York
Edwards GP, Allan GE, Brock C, Duguid A, Gabrys K, Vaarzon-Morel P (2008) Fire and its management in central Australia. Rangel J 30:109–121
Egan JL, Williams RJ (1996) Lifeform distributions of woodland plant species along a moisture availability gradient in Australia’s monsoonal tropics. Aust Syst Bot 9:205–217
Eldridge DJ, Poore AG, Ruiz-Colmenero M, Letnic M, Soliveres S (2016) Ecosystem structure, function, and composition in rangelands are negatively affected by livestock grazing. Ecol Appl 26:1273–1283
Eldridge DJ, Val J, James AI (2011) Abiotic effects predominate under prolonged livestock-induced disturbance. Austral Ecol 36:367–377
Gibb H, Sanders NJ, Dunn RR, Arnan X, Vasconcelos HL, Donoso DA, Andersen AN, Silva RR, Bishop TR, Gomez C, Grossman BF (2018) Habitat disturbance selects against both small and large species across varying climates. Ecography 41:7: 1184–1193
Hoffmann BD (2000) Changes in ant species composition and community organisation along grazing gradients in semi-arid rangelands of the Northern Territory. Rangel J 22:171–189
Hoffmann BD (2010) Using ants for rangeland monitoring: global patterns in the responses of ant communities to grazing. Ecol Indic 10:105–111
Hoffmann BD, James CD (2011) Using ants to manage sustainable grazing: dynamics of ant faunas along sheep grazing gradients conform to four global patterns. Austral Ecol 36:698–708
Holling CS (1973) Resilience and stability of ecological systems. Annu Rev Ecol Syst 4:1–23
Hutley LB, Setterfield SA(2019) Savanna. In: Encyclopedia of ecology (Ed. Fath, B, 2nd ed., pp. 623–633). Elsevier, Amsterdam
Johnson C (2006) Australia’s mammal extinctions: a 50,000-year history. Cambridge University Press, Melbourne, Australia
Johnstone JF, Allen CD, Franklin JF, Frelich LE, Harvey BJ, Higuera PE, Mack MC, Meentemeyer RK, Metz MR, Perry GL, Schoennagel T (2016) Changing disturbance regimes, ecological memory, and forest resilience. Front Ecol Environ 14:369–378
Keeley JE, Pausas JG, Rundel PW, Bond WJ, Bradstock RA (2011) Fire as an evolutionary pressure shaping plant traits. Trends Plant Sci 16:406–411
Laliberte E, Wells JA, DeClerck F, Metcalfe DJ, Catterall CP, Queiroz C, Aubin I, Bonser SP, Ding Y, Fraterrigo JM, McNamara S (2010) Land-use intensification reduces functional redundancy and response diversity in plant communities. Ecol Lett 13:76–86
Landsberg J, James CD, Morton SR (1997) Assessing the effects of grazing on biodiversity in Australia’s rangelands. Australian Biologist 10:153–162
Laycock WA(1991) Stable states and thresholds of range condition on North American rangelands: a viewpoint.J Range Manage427–433
Legendre P, Oksanen J, ter Braak CJ (2011) Testing the significance of canonical axes in redundancy analysis. Methods Ecol Evol 2:269–277
Letnic M, Koch F, Gordon C, Crowther MS, Dickman CR (2009) Keystone effects of an alien top-predator stem extinctions of native mammals. P Roy Soc Lond B Bio 276:3249–3256
Mauda EV, Joseph GS, Seymour CL, Munyai TC, Foord SH (2017) Changes in landuse alter ant diversity, assemblage composition and dominant functional groups in African savannas. - Biodivers Conserv 27:947–965
Moretti M, Duelli P, Obrist MK (2006) Biodiversity and resilience of arthropod communities after fire disturbance in temperate forests. Oecologia 149:312–327
Moretti M, Legg C (2009) Combining plant and animal traits to assess community functional responses to disturbance. Ecography 32:299–309
Nano CE, Clarke PJ, Pavey CR (2012) Fire regimes in arid hummock grasslands and Acacia shrublands. In: Bradstock RA, Gill AM, Williams RJ (eds) ‘Flammable Australia. Fire Regimes, Biodiversity and Ecosystems in a Changing World’. - CSIRO Publishing, Melbourne, pp 195–214
Ness JH, Bronstein JL, Andersen AN, Holland JN (2004) Ant body size predicts dispersal distance of ant-adapted seeds: implications of small-ant invasions. Ecology 85:1244–1250
Pacheco R, Camacho GP, Frizzo TL, Vasconcelos HL (2017) Effects of land-use changes on ecosystem services: decrease in ant predation in human-dominated landscapes in central Brazil. - Entomol Exp Appl 162:302–308
Pacheco R, Vasconcelos HL (2012) Habitat diversity enhances ant diversity in a naturally heterogeneous Brazilian landscape. Biodivers Conserv 21:797–809
Pacheco R, Vasconcelos HL, Groc S, Camacho GP, Frizzo TL (2013) The importance of remnants of natural vegetation for maintaining ant diversity in Brazilian agricultural landscapes. Biodivers Conserv 22:983–997
Parr CL, Robertson HG, Biggs HC, Chown SL (2004) Response of African savanna ants to long-term fire regimes. J Appl Ecol 41:630–642
Parr CL, Lehmann CE, Bond WJ, Hoffmann WA, Andersen AN (2014) Tropical grassy biomes: misunderstood, neglected, and under threat. Trends Ecol Evol 29(4):205–213
Peco B, Carmona CP, De Pablos I, Azcárate FM (2012) Effects of grazing abandonment on functional and taxonomic diversity of Mediterranean grasslands. Agric Ecosyst Environ 152:27–32
Peterson GD (2002) Contagious disturbance, ecological memory, and the emergence of landscape pattern. Ecosystems 5:329–338
R Core Team (2014) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, - Vienna, Austria
Russell-Smith J, Ryan PG, Durieu R (1997) A LANDSAT MSS-Derived fire history of kakadu national park, monsoonal northern australial, 1980-94: Seasonal extent, frequency and patchiness. J Appl Ecol 1:748–766
Russell-Smith J, Yates CP (2007) Australian savanna fire regimes: context, scales, patchiness. Fire Ecol 3:48–63
Sankaran M, Hanan NP, Scholes RJ, Ratnam J, Augustine DJ, Cade BS, Gignoux J, Higgins SI, Le Roux X, Ludwig F, Ardo J (2005) Determinants of woody cover in African savannas. Nature 438:846–849
Shattuck S (2000) Australian ants: their biology and identification. CSIRO publishing, Melbourne
Sousa W (1984) The role of disturbance in natural communities. Ann Rev Ecol Syst 15:353–391
Ter Braak CJ (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67:1167–1179
Thompson JN, Reichman OJ, Morin PJ, Polis GA, Power ME, Sterner RW, Couch CA, Gough L, Holt R, Hooper DU, Keesing F (2001) Frontiers of Ecology: As ecological research enters a new era of collaboration, integration, and technological sophistication, four frontiers seem paramount for understanding how biological and physical processes interact over multiple spatial and temporal scales to shape the earth’s biodiversity. Bioscience 51:15–24
Tongway DJ, Ludwig JA (2011) Restoring disturbed landscapes: putting principles into practice. Island Press, Washington DC
Vasconcelos HL, Leite MF, Vilhena JM, Lima AP, Magnusson WE (2008) Ant diversity in an Amazonian savanna: relationship with vegetation structure, disturbance by fire, and dominant ants. Austral Ecol 33:221–231
Whelan RJ (1995) The ecology of fire. Cambridge University Press, Cambridge, UK
Williams RJ, Woinarski JC, Andersen AN (2003) Fire experiments in northern Australia: contributions to ecological understanding and biodiversity conservation in tropical savannas. Int J Wildland Fire 12:391–402
Acknowledgements
We thank the managers of Annaburroo Station for allowing access. The research was conducted under NT Parks and Wildlife Commission Permit No. 70403.
Funding
Funding was provided to GBA by the Brazilian program Science without Borders from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) in the form of scholarship 236918/2012-5.
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Authorship
The study was conceived and designed by Gabriela Arcoverde and Alan Andersen. Data collection and analysis were performed by Gabriela Arcoverde, who also wrote the first draft of the manuscript. All authors commented on previous versions of the manuscript and read and approved the final manuscript.
Competing interests
There are no competing interests to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Table S.1
Table S.1
Ant species list, showing total abundances at sites with different grazing intensities (No, Low and High) at Annaburroo Station, Northern Territory, Australia
Species | Grazing class | |||
|---|---|---|---|---|
No | Low | High | ||
DORYLINAE | ||||
Lioponera sp.4 (singularis gp) | 0 | 0 | 1 | |
ECTATOMMINAE | ||||
Rhytidoponera sp.9 (tenuis gp) | 7 | 14 | 27 | |
Rhytidoponera borealis | 11 | 6 | 6 | |
Rhytidoponera aurata | 0 | 0 | 1 | |
Rhytidoponera brunnea | 10 | 0 | 0 | |
PONERINAE | ||||
Anochetus paripungens | 1 | 1 | 7 | |
Odontomachus nr.turneri | 14 | 30 | 24 | |
Leptogenys exigua | 6 | 32 | 1 | |
Leptogenys nr.conigera | 1 | 0 | 0 | |
Leptogenys adlerzi | 1 | 0 | 0 | |
Platythyrea parva | 0 | 5 | 0 | |
Bothroponera sp.4 (sublaevis gp) | 2 | 0 | 0 | |
MYRMICINAE | ||||
Monomorium maryannae | 270 | 6 | 11 | |
DOLICHODERINAE | ||||
Iridomyrmex reburrus | 0 | 81 | 141 | |
Iridomyrmex sanguineus | 1325 | 481 | 1425 | |
FORMICINAE | ||||
Melophorus sp.5 (curtus gp.) | 35 | 56 | 28 | |
Melophorus sp.59 (curtus) | 70 | 59 | 123 | |
Melophorus sp.1 (rufoniger gp) | 7 | 4 | 14 | |
Oecophylla smaragdina | 8 | 16 | 4 | |
Opisthopsis haddoni | 10 | 6 | 10 | |
Opisthopsis sp.1 (haddoni gp) | 13 | 13 | 6 | |
Opisthopsis rufonigra | 5 | 2 | 2 | |
Camponotus ‘crozieri’ | 3 | 10 | 13 | |
Camponotus ‘novaehollandiae’ | 1 | 1 | 3 | |
Camponotus sp.9 (novaehollandiae gp) | 9 | 13 | 8 | |
Polyrhachis senilis | 12 | 3 | 7 | |
Polyrhachis sp.12 (obtusa gp) | 0 | 3 | 1 | |
Polyrhachis sp.9 (obtusa gp) | 1 | 0 | 0 | |
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arcoverde, G.B., Setterfield, S.A., Leal, I.R. et al. Does resilience to fire confer resilience to grazing in savanna ant communities of Northern Australia?. J Insect Conserv 27, 107–115 (2023). https://doi.org/10.1007/s10841-022-00426-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-022-00426-y