Abstract
In 2016, the annual value of all global crops dependent upon pollination was valued at $ 235–$ 577 billion (US$). Pollination service is threatened by habitat loss, pesticides, and climate change. We collected potential pollinators with blue vane traps in native grasslands, Conservation Reserve Program (CRP) lands, and cropland (land use), and their embedded playa wetlands (land type) in the Southern High Plains of Texas (SHP). We collected vegetation data that included floral cover, floral diversity, native grass cover, introduced grass cover, canopy gap, and duff cover. Our objective was to determine if individual vegetation characteristics influenced potential pollinator abundance and richness in addition to land use or land type as a whole. The top model explaining abundance was land use, land type, and floral cover. Floral cover had a positive influence on all land uses. The top model that explained insect richness was land use x floral cover, land type, canopy gap, and duff cover. Floral cover positively influenced insect richness in native grasslands but negatively influenced cropland and CRP richness. Percent of canopy gap had a slight negative influence on insect richness, and duff cover positively influenced richness. Our recommendations to land managers are to protect playas to preserve undisturbed areas and native floral resources and seed all future CRP contracts with floral rich plantings to provide for pollinators over the entire growing season.
Implications for insect conservation
Our results show that vegetation characteristics shared by all land uses such as floral cover, canopy gap and duff cover influence pollinator abundance and richness. Therefore, conserving playa wetlands and programs such as the CRP have the potential for enhancing pollinator habitat in working landscapes of the SHP of Texas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pollination service is a vital element of the world's agricultural economy. Consistent estimates of the economic contribution of pollination to the world economy can be difficult to determine, however, the 2016 Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services report estimated that the annual value of all global crops that are dependent upon pollination is $ 235–$ 577 billion (2015 US$) (Lautenbach et al. 2012; IPBES 2016). Food products are not the only commodity dependent upon this service; biofuels, fiber, and timber product economies also benefit from pollination service (IPBES 2016). Although the ecological benefit of wild pollinators to natural areas has not been calculated, it likely exceeds agricultural values (Lebuhn et al. 2013) as 85% of wild flowering plants depend upon pollination (Ollerton et al. 2011).
Researchers have long suspected population declines in insects, including invertebrate pollinators. Hallmann et al. (2017) estimated a 76% seasonal decline (April–October) in insect biomass over 27 years in nature protection areas in Germany. Additional studies support the decline of some wild bees (Goulson et al. 2008; Potts et al. 2010; Cameron et al. 2011) while questions remain regarding dipteran pollinator populations (Kearns 2001; Biesmeijer et al. 2006). There are several potential causes of wild pollinator decline including climate change (Tylianakis 2008; Hegland et al. 2009; Bartomeus et al. 2013), effects of pesticides (Goulson et al. 2015; Rundlöf et al. 2015; Woodcock et al. 2017), introduced invasive species (Potts et al. 2016), habitat loss (Cane and Tepedino 2001; National Resource Council 2007; Potts et al. 2010; Vanbergen et al. 2013) and intensification of land use for agricultural purposes (Winfree et al., 2009; Ollerton et al. 2014). These threats to pollinator communities could reduce the ecosystem services they provide, jeopardize sustainability of the agricultural economies they support (Kevan and Phillips, 2001; Garibalidi, 2009) and ultimately, adversely affect human health and well-being (Smith et al. 2015, Potts et al. 2016).
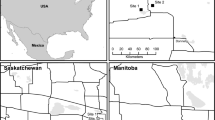
The Southern High Plains of Texas is shortgrass prairie in the north and transitions to mesquite shrub (Prosopis glandulosa) in the south (Johnson 2020). The climate is subhumid to semiarid, and the only reliable source of water is the Ogalalla Aquifer (Rathjen 2020). In the counties involved in this study, Bailey, Briscoe, Carson, Castro, Floyd, Gray, Hockley, Lubbock, and Swisher (Fig. 1), the three primary working land uses are cropland, grazing (native grasslands) (Texas A&M Natural Resources Institute 2021) and Conservation Reserve Program (CRP) (USDA Farm Service Agency 2021). Potential pollinator communities and their resulting services in this region are threatened by loss of habitat from agricultural intensification, and likely insecticides, specifically neonicitinoids. For this study, we focus on potential pollinator habitat.
In 2010, the value of US crops directly dependent upon pollination from the European honey bee (Apis mellifera) was $12.4 billion (US$) (Calderone 2012). Products indirectly dependent on honey bee pollination (e.g. vegetable seed production) were valued at $ 6.8 billion (US$) (Calderone 2012). Losey and Vaughan (2006) estimated that annual pollination service by wild bees contributed to $ 3.07 billion in US fruit and vegetable crops (US$). Although the primary crops of Texas High Plains counties (cotton, corn, wheat, and sorghum) do not require managed pollination services, cotton has shown an 18% increase in boll weight when pollinated by native bees (Cusser et al. 2016) and this service cannot be provided as efficiently by A.mellifera (Vaissière et al. 1984; Cusser et al., 2016). Beef production is also a major agricultural product in Texas, producing over $ 10 million (US$) of value in both 2013 and 2014 (USDA Economic Research Service 2020). Most of the native grasslands in this region that are not in a conservation program are grazed by domestic livestock (Smith 2003). Because native pollinators are necessary for the persistence and health of rangeland plant communities (Black et al. 2011), their service is essential to livestock production in this region (Kevan 1999). In turn, native pollinators need healthy rangelands for floral resources and nesting habitat (Black et al. 2011). Of the counties we sampled, Bailey, Floyd, Hockley, Lubbock, and Swisher have been identified as "at-risk" for loss of pollination services in a study by Koh et al. (2016) because the predicted supply of wild bees is less than what will be required to meet the demand for pollination services (Koh et al. 2016).
In addition to cropland and grazing land use, a large portion of this landscape is dedicated to conservation programs. Intensive agriculture coupled with highly erodible soils (Smith 2003) has resulted in one of the highest concentrations of CRP contracts in the country (USDA Farm Service Agency 2021). The CRP is one of the largest government-private land programs, and over 8.9 million ha are currently enrolled (USDA Farm Service Agency 2021). It is administered by the United States Department of Agriculture Farm Service Agency (USDA FSA) with technical services provided by the Natural Resources Conservation Service (NRCS). Participating landowners apply to take highly erodible or environmentally sensitive land out of production in exchange for the establishment of permanent vegetation cover for 10 to 15 years (USDA Farm Service Agency 2021). A defining landscape characteristic of the High Plains embedded in these land uses are playa wetlands, here out referred to as "playas" (Smith 2003). They are shallow, depressional, recharge wetlands characteristic of prairie and semi-arid habitats formed and maintained by wind, waves, and dissolution (Smith 2003). In this area of intensive agriculture, they are the primary source of floral biodiversity in the region (Haukos and Smith 1994).
For invertebrate pollinators, primarily wild bees, to survive and thrive, they need abundant and diverse populations of annual and perennial flowers across the entire growing season (Vaughan and Black 2008). Approximately 70% of wild bees are solitary ground nesters (Black et al. 2011) and require varying sizes of bare soil exposed to complete life history requirements (Potts et al. 2005; Gilgert and Vaughan 2011; Buckles and Harmon-Threatt 2019). Dead wood, pithy plant stems, and ground litter provide additional nesting habitat for aboveground nesting bees (Vaughan and Black 2008; Sardiñas and Kremen 2014). Additionally, many species of Bombus (bumble bees) will use abandoned rodent or bird nests (Goulson 2010; Broughton et al. 2015). Federal conservation programs in the United States like the CRP present an opportunity to expand and improve habitat resources for wild pollinators. In response to a growing concern for how a decline in pollination service could affect the agricultural economy (Morse and Calderone 2000; Steffan-Dewenter et al. 2005; Calderone 2012), the United States White House, through The National Strategy to Promote the Health of Honey Bees and other Pollinators (White House, 2016), specifically targeted the CRP to "restore or enhance 7 million acres of land for pollinators over the next 5 years through Federal actions and public/private partnerships." Playas within these land uses may also play an important role in providing habitat for potential pollinators, specifically, a rich and diverse floral resource (Haukos and Smith 1994).
Begosh et al. 2020 determined that CRP had lower insect abundance and richness than croplands and native grasslands, and there was no difference in insect abundance and richness between uplands and playas. Patch size had a negative effect on insect abundance and a positive effect on richness. An interaction of vegetation height and percent of bare ground had a positive effect on abundance in cropland and native grassland and a positive effect on insect richness in all land uses. In CRP, vegetation height negatively influenced insect abundance and percent of bare ground cover had a positive influence. These results lead us to question if other vegetation characteristics that could be more directly controlled by land management actions, influenced insect richness and abundance. With this analysis, we examined floral cover, floral diversity, canopy gap cover, and duff cover. Additionally, the data analyzed were specific to a single late-season sampling period, and insects were collected on the same day as the vegetation data rather than over the entire sampling season. The objectives of this study are to determine if potential pollinator abundance and richness is influenced by:
-
specific vegetation characteristics shared by different land uses and land types;
-
land use and land type as a whole;
-
or a combination of vegetation characteristics and land use or land type.
From our analysis, we will make recommendations to land managers how they can improve potential pollinator habitat within a working landscape.
Materials and methods
Study location and site selection
In 2013–2014, we collected insect and vegetation data from privately owned properties located in nine counties in the Southern High Plains of Texas (Bailey, Briscoe, Carson, Castro, Floyd, Gray, Hockley, Lubbock, and Swisher). We selected these sites for our study because they had been used in several previous ecosystem service research projects and the landowners had demonstrated willingness to provide long term access to their properties. These counties encompassed approximately 2,353,522 ha (US Census Bureau 2020). In 2012, 1,221,650 ha (51.91% of land use) were in agricultural crops (Texas A&M Natural Resources Institute 2021), primarily cotton, winter wheat, corn, sorghum, or soybeans (National Agricultural Statistics Service 2020). Approximately 639,649 ha (27.18% of land use) were used for grazing livestock, and 292,564 ha (12.43% of land use) in the study area were under CRP contract with over $ 26,829,099 (US$) in rental payments for the fiscal year (Farm Service Agency 2021). The mean monthly temperature range for 2013 between April and September was 12.6–25.9 °C. The minimum monthly temperature range was 2.6–18.5 °C with a maximum range of 22.5–33.8 °C and average monthly precipitation of 0.5–77.7 mm (US Climate Data 2021). For 2014, the mean monthly temperature range was 14.3–25.6 °C. The minimum monthly temperature range was 4.6–18.9 °C with a maximum range of 24–33.4 °C and average monthly precipitation of 13.7–129.8 mm (US Climate Data, 2021). The sampling area was in a drought when data collection began in 2013 that persisted until 2014 (National Drought Mitigation Center 2020).
Field methods
Each county had three sampling sites that represented each land use; cropland, CRP, and native grasslands. Embedded in each site was a playa. At each site, we sampled in the playa and in the upland. We had 27 sampling sites with two sampling units (playa and upland) at each site. For this analysis, we collected insect and vegetation data during one sampling period in 2013 and one sampling period in 2014 (September 1–14, 2013, and August 31–September 13, 2014), which is a subset of a larger data set collected over 13 sampling periods each year (Begosh et al. 2020). This provided us with an upland and a playa sample for each land use in each county both years (18 upland native grassland, 18 upland CRP, 18 upland cropland, 18 playa native grassland, 18 playa CRP, and 18 playa cropland). We collected insects and vegetation data from each land use in a county on the same day, between 0800 and 1800 CST, for consistency of weather and other environmental factors.
At each sampling site, we established six permanent sets of parallel transects, each 25 m long separated by approximately 10 m. Three were placed in the upland surrounding the playa, and three were placed in the playa. One transect in each set was used to hang the invertebrate traps, and the other was used to collect vegetation data. Transects were placed based on a random azimuth bearing from the center of the playa. Upland transects were placed at least 25 m from the edge of the playa, with the primary criteria presence of upland vegetation. We determined the playa edge using aerial maps and visual confirmation on-site based on vegetation, change in soil characteristics and slope (Luo et al. 1997).
Blue vane traps
We collected potential pollinators with Springstar™ blue vane traps (Stephen and Rao 2005, 2007). We hung them on a wooden post with a wire hanger placed in the middle of one of the transects (12.5 m). The hanging height varied so that the traps were in line with vegetation height and visible. Traps were set out each day and collected the following day as close to 24 h as possible. We poured the contents of the traps into kill jars with ethyl acetate to kill the insects, then specimens were packaged, labeled, and stored frozen. We pooled the upland and playa traps so each site had an upland sample and a playa sample that consisted of the contents of three traps. Insects were identified to species, when possible, by Northern Prairie Wildlife Research Center (NPWRC) technicians and at Oklahoma State University by a technician and graduate student. We confirmed insect identification by consulting reference collections and with input from taxonomic specialists.
Vegetation data collection
We collected floral cover, native grass cover, introduced grass cover, duff cover, and canopy gap data. We used a combination of line-point intercept and gap intercept methods (Elzinga et al. 1998; Herrick 2009). All data were collected off a 25-m tape pulled taut against the ground, anchored on each end. We categorized plants as flowering forbs and native and introduced graminoids. Graminoids included all plants in the family Poaceae and Eleocharis spp. (USDA, NRCS 2020). We defined duff as loose plant material that does not have a clear boundary between the soil layers and can be moved by wind (Herrick 2009). To measure canopy gaps, we used guidelines provided by Herrick (2009). Along the length of the 25 m transect, we recorded the beginning and end of each gap in the canopy larger than 15 cm as viewed from standing height. We recorded the measurement to the nearest centimeter. Unattached litter is not canopy cover and was included as part of the canopy gap. A canopy was defined when 50% of any 3 cm segment of the tape edge intercepts the plant canopy when viewed from above the canopy to the ground (Herrick 2009).
Statistical analysis
To determine the best predictors for insect richness, we created a set of generalized linear mixed-effect models in R version 4.0.2 (R Development Core Team 2020) using insect specimens from traps and vegetation data collected. Insects and vegetation data were collected on the same day at each sampling site so we could analyze only insects and flowers that could potentially interact. Vegetation metrics included percent of floral cover, floral Shannon diversity, percent of native grass cover, percent of introduced grass cover, percent of canopy gap, and percent of duff cover. Potential pollinator data comprised insects from the orders Hymenoptera and Diptera because they are the primary pollinators in most habitats (Larson et al. 2001; Willmer 2011; Orford et al. 2015). We standardized the vegetation data to correct for large eigenvalues. We then checked for collinearity using correlation coefficients and variance inflation factors (VIF) (Zuur et al., 2009). There were no correlation coefficients higher than 0.3, and the highest VIF was 1.5, which were under the acceptable thresholds of 0.7 and 10 respectively (Dormann et al. 2012).
To determine the best predictors for insect abundance and richness, we constructed potential models for abundance and richness (Table S1) using R package lme4 (Bates et al., 2015) and used the bblme package (Bolker and R Core Development Team 2017) to rank models by Akaike Information Criteria corrected for smalls sample size (AICc) (Burnham and Anderson, 2002). We constructed these models specifically to address land use, land type, and vegetation characteristics that might be encountered in the field and that land managers could change with specific management actions. We included at least one variable of native grass cover, introduced grass cover, percent of floral cover or floral diversity as a single variable or as an interaction with land use or land type. Duff was included in models because some wild bees use bits of plant material for nesting material (Black et al. 2014). We included canopy gap because ground-nesting native bees need access to the soil to excavate nests, and need soil with sun exposure for temperatures adequate for larval development (Vaughan and Black 2008). Dense cover may reduce nesting availability and suitability. Canopy gap did not stand alone in a model because the land managers need to know vegetation cover composition to make management decisions. We created plots using ggplot2 (Wickham 2016), interplot (Solt and Yue 2018), plyr (Wickham 2011), and dplyr (Wickham et al. 2019).
We conducted two-way ANOVAs to determine if there were significant effects of factors included in the top models for abundance and richness. When necessary, we used Tukey's post hoc analysis to determine differences between means for significant effects. We performed ANOVAs and Tukey's post hoc analysis in Minitab 19 (Minitab 19 Statistical Software 2019). The significance level was set to α = 0.05.
Results
Mixed-effects models
Land use, land type and floral cover were the best predictors of insect abundance. The top model demonstrated that percent floral cover over all land uses and both land types positively influenced insect abundance (Fig. 2, Tables S2a, S2b).
Insect richness (Tables S3a, S3b) was best predicted by an interaction of land use and percent of floral cover (Fig. 3), land type, percent of canopy gap (Fig. 4) and percent of duff cover (Fig. 5). In cropland and CRP sites, floral cover had a negative effect on insect richness. For native grasslands, floral cover had a positive effect. Uplands had higher insect richness than playas.
For all land uses, canopy gap had a slightly negative effect on insect richness (Fig. 4).
Percent of duff cover had a positive effect on insect richness (Fig. 5).
ANOVAs
There were differences in insect abundance, insect richness, and vegetation characteristics in land use and land type (Table 1, Table S4). Cropland sites had higher insect abundance than CRP and native grassland. Cropland insect richness was higher than CRP and native grassland richness was the same as cropland and CRP. Percent native grass cover was higher in native grasslands and lowest in croplands. Uplands had higher native grass cover than playas. Cropland had higher percent of canopy gap than CRP, and native grassland had similar percent of canopy gap to cropland and CRP. Uplands had higher canopy gap than playas. Cropland had higher duff cover than native grassland, and CRP had the same duff cover as cropland and native grassland. Uplands had higher duff cover than playas.
Discussion
Based on our previous study (Begosh et al. 2020) and past research on CRP and playas in the Great Plains, we predicted that vegetation characteristics associated with CRP, specifically high cover of dense, introduced grasses characteristic of CRP in the Southern High Plains (Berthelsen et al. 1989; O'Connell et al. 2012) and associated low canopy gap would have a negative influence on potential pollinator abundance and richness. We also predicted that floral cover, floral diversity, and duff cover would have a positive effect on potential pollinator abundance because these characteristics provide foraging and nesting resources for native bees (Vaughan and Black 2008; Black et al. 2011, 2014).
Not surprisingly, both abundance and richness models supported percent of floral cover as an influence on potential pollinator communities, and floral cover had a positive effect on insect abundance in all land uses (Blaauw and Isaacs 2014; Williams et al. 2015). For richness, there was an interaction of land use and floral cover with native grasslands having a positive effect on insect richness. Floral cover in cropland and CRP, however, had a negative effect on richness, which is counterintuitive. Trying to separate the environmental influences on pollinator abundance and richness can be difficult (Kennedy et al. 2013). One theory could be that higher floral cover with low floral diversity could result in lower insect richness. This is not the case in this study, however, because there was no difference in floral diversity between land uses. Another potential explanation could be the type of flowers present in each land use and bee preference. Honey bees and wild pollinators have shown differences in their use of floral resources (Urbanowicz et al. 2020) and there can also be a difference in floral preference between bee sexes (Roswell et al. 2019).
Percent of canopy gap and duff cover were additional factors that influenced insect richness, and both are likely contributors to nesting habitat. Although canopy gap may indicate lack of vegetation, it indicates open areas in the vegetation where native bees can access soil to excavate tunnels for nests (Vaughan and Black 2008). It also provides areas where sun can penetrate the vegetation cover and warm the soil for the benefit of developing bee larvae (Vaughan and Black 2008). It may also provide structural diversity in grasslands. Habitats with diverse horizontal and vertical vegetation structure may have enhanced pollination services through architectural complementarity where various pollinators work at different plant heights and sheltered areas (Blüthgen and Klein 2011). There is a threshold, however, where a high percent of canopy gap is a result of too little vegetation cover, which could have reduced foraging resources for bees and indicate poor condition of vegetation. There was a slightly negative effect of percent of canopy gap on pollinator richness. This study took place several years into a severe drought and it is likely that drought stress reduced the percent of canopy cover and negatively affected insect richness. Some species of native bees use material in duff cover as nesting material (Vaughan and Black 2008). While excessive duff or dense embedded litter can prevent access to soil for most solitary bees, the amount measured during our study had a positive effect on insect richness.
Playas are approximately 3% of the SHP land base, but they are the primary source of biodiversity in this arid landscape (Bolen et al. 1989; Haukos and Smith 1994, Smith and Haukos 2002), and could provide an important floral resource for potential pollinators. Cropland playas may have contributed to potential pollinator abundance and richness from pockets of undisturbed refugia that provided nesting and floral resources. Of the nine cropland playas we sampled, seven (77%) were unplowed and protected from cultivation by fence or an unmanaged buffer strip, and as such, were relatively undisturbed. A balance of disturbed and undisturbed areas is favorable for many pollinators, especially bees (Gilgert and Vaughan 2011). These areas likely served as important habitat for ground nesting solitary bees (Black et al. 2011, 2014).
Recommendations and conclusion
Our data provides evidence that vegetation characteristics independent of land use such as floral cover, percent of canopy gap and percent of duff cover influence potential pollinator abundance and richness in the nine counties sampled in the Southern High Plains of Texas. Land managers have the ability to manage these characteristics in their operations. Additionally, we demonstrated that playa wetlands within each land use also influence insect communities, and these landscape features can also be managed by landowners, even in an active agricultural operation.
In the SHP, cropland playas and their immediate uplands should be a focus of conservation programs and have potential to provide quality habitat for pollinators (Deguines et al. 2012). The most important action landowners can implement is to increase and enhance floral cover in their operations. Maintaining an undisturbed de facto buffered areas around playas and the floral resources within the playas could provide valuable insect foraging and nesting resources primarily through undisturbed ground, and abundant and consistent floral resources. Riparian buffer strips have been shown to support richer populations of plants and higher numbers of pollinators than adjacent intensively managed grasslands (Cole et al. 2015). In southwestern Minnesota, butterfly abundance and diversity were positively correlated with buffer strip width (Davros et al. 2006). Future CRP plantings should be designed to incorporate native grasses and a variety of native, nectar-rich flowers that bloom over the entire growing season (Isaacs et al. 2009) into the landscape with the goal to enhance overall plant and insect biodiversity rather than pollination service alone (Chateil and Porcher 2015). In a landscape where most of the native shortgrass prairie is actively grazed by cattle (Smith 2003), CRP could provide pockets of minimally disturbed habitat to serve as refugia for more specialized pollinators (Basu et al. 2016). Although landowners cannot control percent of canopy gap and percent of duff cover directly, these characteristics can be indirectly managed by maintaining patches of healthy grassland and preserving the functional integrity of playa wetlands.
Availability of data and material (data transparency)
Due to privacy issues, data is not publicly available. See corresponding author.
Code availability (software application or custom code)
Code is available from corresponding author.
References
Bartoneus I, Park MG, Gibbs J, Danforth BN, Lasko AN, Winfree R (2013) Biodiversity ensures plant–pollinator phenological synchrony against climate change. Ecol Lett 16(11):1331–1338. https://doi.org/10.1111/ele.12170
Basu P, Parui AK, Chatterjee S, Dutta A, Chakraborty P, Roberts S, Smith B (2016) Scale dependent drivers of wild bee diversity in tropical heterogeneous agricultural landscapes. Ecol Evol 6:6983–6992
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48
Begosh A, Smith LM, McMurry ST, Harris JP (2020) Influence of the Conservation Reserve Program (CRP) and playa wetlands on pollinator communities in the Southern High Plains, USA. J Environ Manag. https://doi.org/10.1016/j.jenvman.2019.109910
Berthelsen PS, Smith LM, Coffman C (1989) CRP land and game bird production in the Texas High Plains. J Soil Water Conserv 44:504–507
Biesmeijer JC, Roberts SPM, Reemer M, Ohlemuller R, Edwards M, Peeters T, Schaffers AP, Potts SG, Kleukers R, Thomas D, Settele J, Junin WE (2006) Parallel declines in pollinators and insect-pollinated plants in Britian and Netherlands. Science 313(5785):351–354
Blaauw BR, Isaacs R (2014) Larger patches of diverse floral resources increase insect pollinator density, diversity, and their pollination of native wildflowers. Basic Appl Ecol 15(8):701–711
Black SH, Shepherd M, Vaughan M (2011) Rangeland management for pollinators. Rangelands 33:9–13
Black SH, Hodges N, Vaughn M, Shepherd M (2014) Pollination in natural areas: a primer on habitat management. The Xerces Society for Invertebrate Conservation, Portland
Blüthgen N, Klein AM (2011) Functional complementarity and specialisation: the role of biodiversity in plant–pollinator interactions. Basic Appl Ecol 12:282–291
Bolen E, Smith LM, Schramm HL (1989) Playa lakes; prairie wetlands of the Southern High Plains. Bioscience 39:615–623
Bolker B, R Development Core Team (2017) bbmle: tools for general maximum likelihood estimation. R package version 1.0.20. https://CRAN.R-project.org/package=bblme
Broughton RK, Grzegorz H, Maziarz M, Smith KW, Smith L, Hinsley SA (2015) Nest-site competition between bumble bees (Bombidae), social wasps (Vespidae) and cavity-nesting birds in Britain and the Western Palearctic. Bird Study 62(3):427–437. https://doi.org/10.1080/00063657.2015.1046811
Buckles BJ, Harmon-Threatt AN (2019) Bee diversity in tallgrass prairies affected by management and its effects on above- and below-ground resources. J Appl Ecol 56:2443–2453. https://doi.org/10.1111/13652664.13479
Burnham KP, Anderson DR (2002) Model selection and inference—a practical information theoretic approach, 2nd edn. Springer, New York
Calderone NW (2012) Insect pollinated crops, insect pollinators and US agriculture: trend analysis of aggregate data for the period 1992–2009 (Insect Pollinators and US Agriculture). PLoS ONE 7:e37235
Cameron SA, Lozier JD, Strange JT, Koch JB, Cordes N, Solter LF, Griswold T (2011) Patterns of widespread decline in North American bumble bees. PNAS 108(2):662–667. https://doi.org/10.1073/pnas.1014743108
Cane J, Tepedino V (2001) Causes and extent of declines among native North American invertebrate pollinators: detection, evidence, and consequences. Conserv Ecol 5:1–1
Chateil C, Porcher E (2015) Landscape features are a better correlate of wild plant pollination than agricultural practices in an intensive cropping system. Agric Ecosys Environ 201:51–57
Cole LJ, Brocklehurst S, Robertson D, Harrison W, McCracken DI (2015) Riparian buffer strips: their role in the conservation of insect pollinators in intensive grassland systems. Agric Ecosyst Environ 211:207–220
Cusser S, Neff JL, Jha S (2016) Natural land cover drives pollinator abundance and richness, leading to reductions in pollen limitation in cotton agroecosystems. Agric Ecosyst Environ 226:33–42
Davros N, Debinski D, Reeder K, Hohman W (2006) Butterflies and Continuous Conservation Reserve Program Filter Strips: landscape considerations. Wildl Soc Bull 34:936–943
Deguines N, Julliard R, de Flores M, Fontaine C (2012) The whereabouts of flower visitors: contrasting land-use preferences revealed by a country-wide survey based on citizen science (Land-Use Affinity of Flower Visitors). PLoS ONE 7:e45822
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, García Marquéz JR, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, McLean C, Osborne PE, Reinking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2012) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 35:001–020. https://doi.org/10.1111/j.1600-0587.2012.07348.x
Economic Research Service (ERS), U.S. Department of Agriculture (USDA) (2020). Accessed Mar 2020
Elzinga CL, Salzer DW, Willoughby JW (1998) Measuring & monitoring plant populations. Denver, C): U.S. Dept. of the Interior, Bureau of Land Management.
Garibalidi LA, Aizen M, Cunningham S, Klein AM (2009) Pollinator shortage and global crop yield: Looking at the whole spectrum of pollinator dependency. Commun Integr Biol 2(1):37–39
Gilgert W, Vaughan M (2011) The value of pollinators and pollinator habitat to rangelands: connections among pollinators, insects, plant communities, fish, and wildlife. Rangelands 33:14–19
Goulson D (2010) Bumble bees: behaviour, ecology and conservation, 2nd edn. Oxford University Press, Oxford
Goulson D, Lye GC, Darvill B (2008) Decline and conservation of bumble bees. Annu Rev Entomol 53:191–208. https://doi.org/10.1146/annurev.ento.53.103106.093454
Goulson D, Nicholls E, Botias C, Rotheray EL (2015) Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science. https://doi.org/10.1126/science.1255957
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, Stenmans W, Müller A, Sumser H, Hörren T, Goulson D, de Kroon H (2017) More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 12(10):e0185809. https://doi.org/10.1371/journal.pone.018
Haukos DA, Smith LM (1994) The importance of playa wetlands to biodiversity of the Southern High Plains. Landsc Urban Plan 28:83–98
Hegland SJ, Nielsen A, Lázaro A, Bjerknes A, Totland Ø (2009) How does climate warming affect plant-pollinator interactions? Ecol Lett 12:184–195
Herrick JE (2009) Monitoring manual for grassland, shrubland and savanna ecosystems. USDA-ARS Jornada Experimental Range, Las Cruces
IPBES (2016) The assessment Ecosystem Services on pollinators, pollination and food production. In: Potts SG, Imperatriz-Fonseca VL, Ngo HT (eds) Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services, Bonn, Germany
Isaacs R, Tuell J, Fiedler A, Gardiner M, Landis D (2009) Maximizing arthropod-mediated ecosystem services in agricultural landscapes: the role of native plants. Front Ecol Environ 7(4):196–203. https://doi.org/10.1890/080035
Johnson EH (2020) Handbook of Texas online, "High Plains". http://www.tshaonline.org/handbook/online/articles/ryh01. Accessed 06 Apr 2020
Kearns CA (2001) North American dipteran pollinators: assessing their value and conservation status. Ecol Soc 5:5
Kennedy CM, Lonsdorf E, Neel MC, Williams NM, Ricketts TH, Winfree R, Bommarco R, Brittain C, Burley AL, Cariveau D, Carvalheiro LG, Chacoff NP, Cunningham SA, Danforth BN, Dudenhöffer JH, Elle E, Gaines HR, Garibaldi LA, Gratton C, Holzschuh A, Isaacs R, Javorek SK, Jha S, Klein AM, Krewenka K, Mandelik Y, Mayfield MM, Morandin L, Neame LA, Otieno M, Park M, Potts SG, Rundlöf M, Saez A, Steffan-Dewenter I, Taki H, Viana BF, Westphal C, Wilson JK, Greenleaf SS, Kremen C (2013) A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol Lett 16:584–599
Kevan PG (1999) Pollinators as bioindicators of the state of the environment: species activity and diversity. Agri Ecosyst Environ 74(1–3):373–393. https://doi.org/10.1016/S0167-8809(99)00044-4
Kevan P, Phillips T (2001) The economic impacts of pollinator declines: an approach to assessing the consequences. Conserv Ecol 5:8–8
Koh I, Lonsdorf EV, Williams NM, Brittain C, Isaacs R, Gibbs J, Ricketts TH (2016) Modeling the status, trends, and impacts of wild bee abundance in the United States. Proc Natl Acad Sci USA 113:140
Larson BMH, Kevan P, Inouye D (2001) Flies and flowers: taxonomic diversity of anthophiles and pollinators. Can Entomol 133:439–465
Lautenbach S, Seppelt R, Liebscher J, Dormann CF (2012) Spatial and temporal trends of global pollination benefit. PLoS ONE 7(4):e35954. https://doi.org/10.1371/journal.pone.0035954
LeBuhn G, Droege S, Connor EF, Gemmill-Herren B, Potts SG, Minckley RL, Griswold T, Jean R, Kula E, Roubik DW, Cane J, Wright KW, Frankie G, Parker F (2013) Detecting insect pollinator declines on regional and globalscales. Conserv Biol 27(1):113–120
Losey JE, Vaughan M (2006) The economic value of ecological services provided by insects. Bioscience 56:311–323
Luo HR, Smith LM, Allen BL, Haukos DA (1997) Effects of sedimentation on playa wetland volume. Ecol Appl 7:247–252
Minitab 19 Statistical Software (2019) Minitab 19 Statistical Software. State College, PA
Morse R, Calderone N (2000) The value of honey bees as pollinators of U.S. crops in 2000. Bee Culture, 128:P1.
National Agricultural Statistics Service (2020) Texas county estimates. In: USDA's National Agricultural Statistics Service Southern Plains Regional Field Office, editor
National Drought Mitigation Center (2020) U.S. Drought Monitor: Texas. U.S. Drought Monitor Map Archive. University of Nebraska, Lincoln, NE
National Research Council (2007) Status of pollinators in North America. National Academies Press, Washington, DC
O’Connell JL, Johnson LA, Smith LM, McMurry ST, Haukos DA (2012) Influence of land-use and conservation programs on wetland plant communities of the semiarid United States Great Plains. Biol Conserv 146:108–115
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120(3):321–326
Ollerton J, Erenler H, Edwards M, Crockett R (2014) Extinctions of aculeate pollinators in Britain and the role of large-scale agricultural changes. Science 346(6215):1360–1362
Orford KA, Vaughan IP, Memmott J (2015) The forgotten flies: the importance of non-syrphid Diptera as pollinators. Proc Biol Sci 282:20142934
Potts SG, Vulliamy B, Roberts S, O’Toole C, Dafni A, Ne’eman G, Willmer P (2005) Role of nesting resources in organising diverse bee communities in a Mediterranean landscape. Ecol Entomol 30:78–85. https://doi.org/10.1111/j.0307-6946.2005.00662.x
Potts SG, Biesmeijer J, Kremen C, Neumann P, Schweiger O, Kunin WE (2010) Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol 25:345–353
Potts SG, Imperatriz-Fonseca V, Ngo HT, Aizen MA, Biesmeijer JC, Breeze TD, Dicks LV, Garibaldi LA, Hill R, Settele J, Vanbergen AJ (2016) Safeguarding pollinators and their values to human well-being. Nature 540:220–229. https://doi.org/10.1038/nature20588
R Development Core Team (2020) R: a language and environment for statistical computing. R Foundations for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Rathjen FW (2020) Handbook of Texas online, "Panhandle". http://www.tshaonline.org/handbook/online/articles/ryp01. Accessed 5 Apr 2020
Roswell M, Dushoff J, Winfree R (2019) Male and female bees show large differences in floral preference. PLoS ONE 14(4):e0214909. https://doi.org/10.1371/journal.pone.0214909
Rundlöf M, Andersson GKS, Bommarco R, Fries I, Hederström V, Herbertsson L, Jonsson O, Klatt BK, Pedersen TR, Yourstone J, Smith HG (2015) Seed coating with a neonicotinoid insecticide negatively affects wild bees. Nature 521(7550):77–80
Sardiñas H, Kremen C (2014) Evaluating nesting microhabitat for ground-nesting bees using emergence traps. Basic Appl Ecol 15(2):161–168
Smith LM (2003) Playas of the Great Plains, 1st edn. University of Texas Press, Austin
Smith LM, Haukos D (2002) Floral diversity in relation to playa wetland area, watershed, and disturbance. Conserv Biol 16:964–974
Smith MR, Singh GM, Mozaffarian D, Myers SS (2015) Effects of decreases of animal pollinators on human nutrition and global health: a modelling analysis. Lancet 386:1964–1972
Solt F, Yue H (2018) Interplot: plot the effects of variables in interaction terms. R package version 0.2.1. https://CRAN.R-project.org/package=interplot
Steffan-Dewenter I, Potts SG, Packer L (2005) Pollinator diversity and crop pollination services are at risk. Trends Ecol Evol 20:651–652
Stephen WP, Rao S (2005) Unscented color traps for non-Apis bees (Hymenoptera: Apiformes). J Kans Entomol Soc 78:373–380
Stephen WP, Rao S (2007) Sampling native bees in proximity to a highly competitive food resource (Hymenoptera: Apiformes). J Kans Entomol Soc 80:369–376
Texas A&M Natural Resources Institute (2021) Texas Land Trends: a database of compiled and analyzed values for working lands in Texas. College Station, TX, USA. http://txlandtrends.org
Texas Association of Counties (2021) County Information Program. https://imis.county.org/iMIS/CountyInformationProgram/TexasMapCIP.aspx
The White House (2016) National strategy to promote the health of honey bees and other pollinators: report of the pollinator health task force. The White House, Washington DC
Tylianakis JM, Didham RK, Bascompte J, Wardle DA (2008) Global change and species interactions in terrestrial ecosystems. Ecol Lett 11:1351–1363
Urbanowicz C, Muniz PA, McArt SH (2020) Honeybees and wild pollinators differ in their preferences for and use of introduced floral resources. Ecol Evol 10(3):6741–6751. https://doi.org/10.1002/ece3.6417
US Census Bureau (2020). https://data.census.gov/
US Climate Data (2021) https://www.usclimatedata.com/climate/texas/united-states/3213. Accessed 12 Mar 2021
USDA Farm Service Agency (2021) Conservation reserve statistics. https://www.fsa.usda.gov/programs-and-services/conservation-programs/reports-and-statistics/conservation-reserve-program-statistics/index
USDA, NRCS (2020) The PLANTS database (http://plants.usda.gov). National Plant Data Team, Greensboro, NC 27401-4901 USA. Accessed 14 June 2020
Vaissière BE, Moffett JO, Loper GM (1984) Honey bees as pollinators for hybrid cotton seed production on the Texas High Plains. J Agron 76(6):867–1022
Vanbergen AJ, Baude M, Biesmeijer JC, Britton NF, Brown MJF, Bryden J, Budge GE, Bull JC, Carvell C, Challinor AJ, Connolly CN, Evans DJ, Feil EJ, Garratt MP, Greco MK, Heard MS, Jansen VAA, Keeling MJ, KuninWE MGC, Memmott J, Murray JT, Nicolson SW, Osborne JL, Paxton RJ, Pirk CWW, Polce C, Potts SG, Priest NK, Raine NE, Roberts S, Ryabov EV, Shafir S, Shirley MDF, Simpson SJ, Stevenson PC, Stone GN, Termansen M, Wright GA (2013) Threats to an ecosystem service: pressures on pollinators. Front Ecol Environ 11(5):251–259
Vaughan M, Black SH (2008) Native pollinators: how to protect and enhance habitat for native bees. Native Plant J 9(2):80–91
Wickham H (2011) The split-apply-combine strategy for data analysis. J Stat Softw 40(1):1–29
Wickham H (2016) Elegant graphics for data analysis. Springer, New York
Wickham H, François R, Henry L, Müller K (2019) Dplyr: A Grammar of Data Manipulation. R package version 0.8.3. http://CRAN.R-project/package=dplyr.
Williams NM, Ward KL, Pope N, Isaacs R, Willson J, May EA, Ellis J, Daniels J, Pence A, Ulmann K, Peters J (2015) Native wildflower plantings support wild bee abundance and diversity in agricultural landscapes across the United States. Ecol Appl 25(8):2119–2131
Willmer P (2011) Chapter 1. Why pollination is interesting. In: Willmer P (ed) Pollination and floral ecology. Princeton University Press, Princeton, pp 3–10
Winfree R, Aguilar R, Vázquez DP, Lebuhn G, Aizen MA (2009) A meta-analysis of bees’ responses to anthropogenic disturbance. Ecology 90:2068–2076
Woodcock BA, Bullock JM, Shore RF, Heard MS, Pereira MG, Redhead J, Ridding L, Dean H, Sleep D, Henrys P, Peyton J, Hulmes S, Hulmes L, Sárospataki M, Saure C, Edwards M, Genersch E, Knäbe S, Pywell RF (2017) Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science 356(6345):1393–1395. https://doi.org/10.1126/science.aaa1190
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed-effects models ad extensions in ecology with R. Springer, New York
Acknowledgements
We are grateful to Skip Hyberg and the United States Department of Agriculture Farm Service Agency and CEAP Wetlands for project funding. We are especially appreciative of Lisa Overall's assistance with vane trap specimen insect identification and Sam O'Dell with the Northern Prairie Wildlife Research Center for sweep net specimens. Jonathan Harris provided valuable feedback with statistical analysis. Additionally, thanks to Joshua Dunlap and Lauren Price for collecting insect specimens. Buffalo Lake National Wildlife Refuge and Muleshoe National Wildlife Refuge provided housing and support in the field. Finally, we owe a tremendous thank you to the private landowners of Texas who provided access to their property to collect specimens. Without their generosity, this study would not have been possible.
Funding
This study was funded by the United States Department of Agriculture Farm Service Agency (Grant Number G00008468). The study sponsors did not play any role in the study design, the collection, analysis or interpretation of data, writing of the report or decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
AB: Writing: Original draft preparation, investigation, data collection, statistical analysis. LMS: Conceptualization, methodology, writing: review and editing, fund acquisition. STM: Conceptualization, methodology, writing: review and editing, fund acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Consent
Not applicable.
Ethics approval
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Begosh, A., Smith, L.M. & McMurry, S.T. Major land use and vegetation influences on potential pollinator communities in the High Plains of Texas. J Insect Conserv 26, 231–241 (2022). https://doi.org/10.1007/s10841-022-00380-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-022-00380-9