Abstract
Background
The lesion index (LSI) helps predict the lesion size and is widely used in ablation of various types of arrhythmias. However, the influence of the ablation settings on the lesion formation and incidence of steam pops under the same LSI value remains unclear.
Methods
Using a contact force (CF) sensing catheter (TactiCath™) in an ex vivo swine left ventricle model, RF lesions were created with a combination of various power steps (30 W, 40 W, 50 W) and CFs (10 g, 20 g, 30 g, 40 g, 50 g) under the same LSI values (5.2 and 7.0). The correlation between the lesion formation and ablation parameters was evaluated.
Results
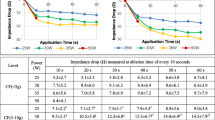
Ninety RF lesions were created under a target LSI value of 5.2, and eighty-four were developed under a target LSI value of 7.0. In the LSI 5.2 group, the resultant lesion size widely varied according to the ablation power, and a multiple regression analysis indicated that the ablation energy delivered was the best predictor of the lesion formation. To create a lesion depth > 4 mm, an ablation energy of 393 J is the best cutoff value, suggesting a possibility that ablation energy may be used as a supplemental marker that better monitors the progress of the lesion formation in an LSI 5.2 ablation. In contrast, such inconsistency was not obvious in the LSI 7.0 group. Compared with 30 W, the 50-W ablation exhibited a higher incidence of steam pops in both the LSI 5.2 and 7.0 groups.
Conclusions
The LSI-lesion size relationship was not necessarily consistent, especially for an LSI of 5.2. To avoid any unintentional, weak ablation, the ablation energy may be a useful supportive parameter (393 J as a cutoff value for a 4-mm depth) during ablation with an LSI around 5.2. Thanks to a prolonged ablation time, the LSI-lesion size relationship is consistent for an LSI of 7.0. However, it is accompanied by a high incidence of steam pops. Care should be given to the ablation settings even when the same LSI value is used.





Similar content being viewed by others
Data Availability
Available upon request.
References
McGarry TJ, Narayan SM. The anatomical basis of pulmonary vein reconnection after ablation for atrial fibrillation: wounds that never felt a scar? J Am Coll Cardiol. 2012;59(10):939–41.
Sawhney N, Anousheh R, Chen WC, Narayan S, Feld GK. Five-year outcomes after segmental pulmonary vein isolation for paroxysmal atrial fibrillation. Am J Cardiol. 2009;104(3):366–72.
Neuzil P, Reddy VY, Kautzner J, Petru J, Wichterle D, Shah D, et al. Electrical reconnection after pulmonary vein isolation is contingent on contact force during initial treatment: results from the EFFICAS I study. Circ Arrhythm Electrophysiol. 2013;6(2):327–33.
Reddy VY, Shah D, Kautzner J, Schmidt B, Saoudi N, Herrera C, et al. The relationship between contact force and clinical outcome during radiofrequency catheter ablation of atrial fibrillation in the TOCCATA study. Heart Rhythm. 2012;9(11):1789–95.
Kautzner J, Neuzil P, Lambert H, Peichl P, Petru J, Cihak R, et al. EFFICAS II: optimization of catheter contact force improves outcome of pulmonary vein isolation for paroxysmal atrial fibrillation. Europace. 2015;17(8):1229–35.
Calzolari V, De Mattia L, Indiani S, Crosato M, Furlanetto A, Licciardello C, et al. In vitro validation of the lesion size index to predict lesion width and depth after irrigated radiofrequency ablation in a porcine model. JACC Clin Electrophysiol. 2017;3(10):1126–35.
Kanamori N, Kato T, Sakagami S, Saeki T, Kato C, Kawai K, et al. Optimal lesion size index to prevent conduction gap during pulmonary vein isolation. J Cardiovasc Electrophysiol. 2018;29(12):1616–23.
Kawano D, Mori H, Kato R, Tsutsui K, Ikeda Y, Sumitomo N, et al. The optimal ablation setting for a local impedance guided catheter in an in vitro experimental model. J Cardiovasc Electrophysiol. 2021;32(8):2069–76.
Mori H, Kato R, Sumitomo N, Ikeda Y, Goto K, Tanaka S, et al. Relationship between the ablation index, lesion formation, and incidence of steam pops. J Arrhythm. 2019;35(4):636–44.
Lozano Granero C, Franco E, Matía Francés R, Hernández-Madrid A, Sánchez-Pérez I, Zamorano Gómez JL, et al. Impact of power and contact force on index-guided radiofrequency lesions in an ex vivo porcine heart model. J Interv Card Electrophysiol. 2022;63(3):687–97.
Leshem E, Zilberman I, Tschabrunn CM, Barkagan M, Contreras-Valdes FM, Govari A, et al. High-power and short-duration ablation for pulmonary vein isolation: biophysical characterization. JACC Clin Electrophysiol. 2018;4(4):467–79.
Simmers TA, de Bakker JM, Wittkampf FH, Hauer RN. Effects of heating with radiofrequency power on myocardial impulse conduction: is radiofrequency ablation exclusively thermally mediated? J Cardiovasc Electrophysiol. 1996;7(3):243–7.
Kawano D, Mori H, Tsutsui K, Fukaya H, Tanaka N, Narita M, et al. Time dependency in the radiofrequency lesion formation for a local impedance guided catheter in an ex vivo experimental model. J Arrhythm. 2022;38(6):1080–7.
Shin DG, Ahn J, Han SJ, Lim HE. Efficacy of high-power and short-duration ablation in patients with atrial fibrillation: a prospective randomized controlled trial. Europace. 2020;22(10):1495–501.
Chung MK, Refaat M, Shen WK, Kutyifa V, Cha YM, Di Biase L, et al. Atrial fibrillation: JACC council perspectives. J Am Coll Cardiol. 2020;75(14):1689–713.
Metzner A, Kuck KH, Chun JKR. What we have learned: is pulmonary vein isolation still the cornerstone of atrial fibrillation ablation? Europace. 2022;24(Supplement_2):ii8–13.
Katić J, Anić A, Brešković T, Jurišić Z. Higher than recommended lesion size index target values for pulmonary vein isolation result in better clinical outcomes in paroxysmal atrial fibrillation patients. J Interv Card Electrophysiol. 2022;64(2):463–8.
Hall B, Jeevanantham V, Simon R, Filippone J, Vorobiof G, Daubert J. Variation in left atrial transmural wall thickness at sites commonly targeted for ablation of atrial fibrillation. J Interv Card Electrophysiol. 2006;17(2):127–32.
Ho SY, Sanchez-Quintana D, Cabrera JA, Anderson RH. Anatomy of the left atrium: implications for radiofrequency ablation of atrial fibrillation. J Cardiovasc Electrophysiol. 1999;10(11):1525–33.
Tung R, Raiman M, Liao H, Zhan X, Chung FP, Nagel R, et al. Simultaneous endocardial and epicardial delineation of 3D reentrant ventricular tachycardia. J Am Coll Cardiol. 2020;75(8):884–97.
Acknowledgements
We appreciate the help in proofreading our manuscript by Mr. John Martin.
Author information
Authors and Affiliations
Contributions
HM and RK, study conception and design; NM drafted the manuscript; HS, WS, KM, and NT, data collection and data analysis; KT, YI, TA, and SN, manuscript revision; and KM, study supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Narita, M., Higuchi, S., Kawano, D. et al. Does the same lesion index mean the same efficacy and safety profile: influence of the differential power, time, and contact force on the lesion size and steam pops under the same lesion index. J Interv Card Electrophysiol 67, 147–155 (2024). https://doi.org/10.1007/s10840-023-01583-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-023-01583-z